Overcoming Challenges in the Management of Refractory Rheumatoid Arthritis: Expert Insight on the Clinical Potential of Novel Therapeutic Options
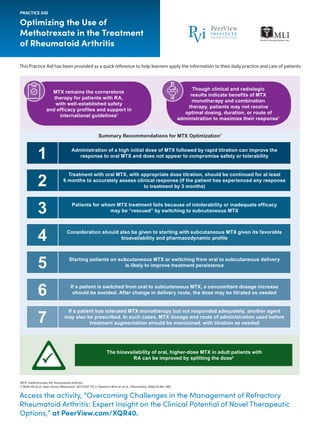
- 1. Access the activity, “Overcoming Challenges in the Management of Refractory Rheumatoid Arthritis: Expert Insight on the Clinical Potential of Novel Therapeutic Options,” at PeerView.com/XQR40. Optimizing the Use of Methotrexate in the Treatment of Rheumatoid Arthritis PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. MTX: methotrexate; RA: rheumatoid arthritis. 1. Bello AE et al. Open Access Rheumatol. 2017;9:67-79. 2. Hoekstra M et al. et al. J Rheumatol. 2006;33:481-485. MTX remains the cornerstone therapy for patients with RA, with well-established safety and efficacy profiles and support in international guidelines1 Though clinical and radiologic results indicate benefits of MTX monotherapy and combination therapy, patients may not receive optimal dosing, duration, or route of administration to maximize their response1 The bioavailability of oral, higher-dose MTX in adult patients with RA can be improved by splitting the dose2 Summary Recommendations for MTX Optimization1 Administration of a high initial dose of MTX followed by rapid titration can improve the response to oral MTX and does not appear to compromise safety or tolerability1 Treatment with oral MTX, with appropriate dose titration, should be continued for at least 6 months to accurately assess clinical response (if the patient has experienced any response to treatment by 3 months)2 Patients for whom MTX treatment fails because of intolerability or inadequate efficacy may be “rescued” by switching to subcutaneous MTX3 Consideration should also be given to starting with subcutaneous MTX given its favorable bioavailability and pharmacodynamic profile4 Starting patients on subcutaneous MTX or switching from oral to subcutaneous delivery is likely to improve treatment persistence5 If a patient is switched from oral to subcutaneous MTX, a concomitant dosage increase should be avoided. After change in delivery route, the dose may be titrated as needed6 If a patient has tolerated MTX monotherapy but not responded adequately, another agent may also be prescribed. In such cases, MTX dosage and route of administration used before treatment augmentation should be maintained, with titration as needed7
- 2. Access the activity, “Overcoming Challenges in the Management of Refractory Rheumatoid Arthritis: Expert Insight on the Clinical Potential of Novel Therapeutic Options,” at PeerView.com/XQR40. A Closer Look at Biologic and Small Molecule Targets in Rheumatoid Arthritis1 This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. APC: antigen-presenting cell; BCR: B-cell receptor; CD: cluster of differentiation; GM-CSF: granulocyte-macrophage colony-stimulating factor; IL: interleukin; JAK: Janus kinase; MHC: major histocompatibility complex; RANK: receptor activator of nuclear factor κB; RANKL: receptor activator of nuclear factor κB ligand; TCR: T-cell receptor; Tfh : T follicular helper; Th : T helper. 1. Smolen JS et al. Nat Rev Dis Primers. 2018;4:18001. PRACTICE AID IL-12 IL-23 MHC TCR T cell CD28 CD80 CD86 IFNγ IL-2 IL-21 Co-stimulation CD40L CD40 CD20 BCR B cell Anti-CD20 drugs Rituximab CD80 and CD86 inhibitors Abatacept RANK? Complement receptor Macrophage IFNγ IL-17 RANKL Fc receptor Immune complexes IL-15 IL-18 Th 1/ Th 17/ Tfh IL-6 GM-CSF Plasma cell Autoantibody RANKL Fibroblast Chondrocyte IL-6R Osteoclast TNF IL-1 IL-6 JAK inhibitors Baricitinib and tofacitinib TNF inhibitors Adalimumab, certolizumab, etanercept, golimumab, and infliximab IL-6R inhibitors Tocilizumab and sarilumab APC JAK signalling
- 3. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Disease-Modifying Antirheumatic Drugs for Rheumatoid Arthritis CD: cluster of differentiation; DMARD: disease-modifying antirheumatic drug; IL: interleukin; JAK: Janus kinase; RA: rheumatoid arthritis 1. Smolen JS et al. Nat Rev Dis Primers. 2018;4:18001. 2. Olumiant (baricitinib) Prescribing Information. http://pi.lilly.com/us/olumiant-uspi.pdf. Accessed April 5, 2019. 3. Kevzara (sarilumab) Prescribing Information. http://products.sanofi.us/kevzara/kevzara.pdf. Accessed April 5, 2019. PRACTICE AID Access the activity, “Overcoming Challenges in the Management of Refractory Rheumatoid Arthritis: Expert Insight on the Clinical Potential of Novel Therapeutic Options,” at PeerView.com/XQR40. Currently Available DMARDs for RA1 An Overview of the Newest Treatment Options for RA Synthetic DMARDs Conventional synthetic DMARDs • Unknown target: Methotrexate, sulfasalazine, chloroquine, and hydroxychloroquine • Known target: Dihydroororate dehydrogenase for leflunomide Targeted synthetic DMARDs • JAK1 and JAK 2: Baricitinib • JAK1, JAK2, and JAK3: Tofacitinib Biological DMARDs Targets of biologic originator DMARDs • Tumor necrosis factor: Adalimumab, certolizumab, etanercept, golimumab, and infliximab • IL-6 receptor: Tocilizumab and sarilumab • CD80 and CD86 (involved in T-cell costimulation): Abatacept • CD20 (expressed by B cells): Rituximab Targets of biosimilar DMARDs • Tumor necrosis factor: Adalimumab, etanercept, and infliximab Drug FDA Approval Mechanism of Action Indication Route of Administration Adverse Reactions Baricitinib2 May 2018 JAK inhibitor Adult patients with moderately to severely active RA who have had an inadequate response to one or more TNF antagonist therapies Oral Upper respiratory tract infections, nausea, herpes simplex, and herpes zoster Sarilumab3 May 2017 IL-6 receptor antagonist Adult patients with moderately to severely active RA who have had an inadequate response or intolerance to one or more DMARDs Subcutaneous Neutropenia, increased ALT, injection-site erythema, upper respiratory infections, and urinary tract infections
