Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors
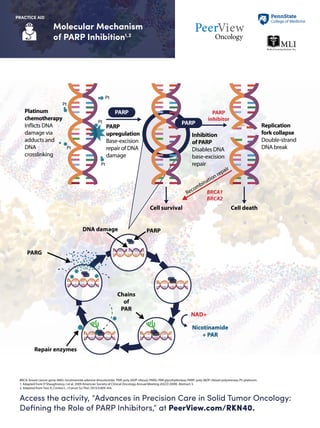
- 1. Molecular Mechanism of PARP Inhibition1,2 Access the activity, “Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors,” at PeerView.com/RKN40. PRACTICE AID BRCA: breast cancer gene; NAD: nicotinamide adenine dinucleotide; PAR: poly (ADP-ribose); PARG: PAR glycohydrolase; PARP: poly (ADP-ribose) polymerase; Pt: platinum. 1. Adapted from O’Shaughnessy J et al. 2009 American Society of Clinical Oncology Annual Meeting (ASCO 2009). Abstract 3. 2. Adapted from Toss A, Cortesi L. J Cancer Sci Ther. 2013;5:409-416. Pt Pt Pt Pt PARP PARP PARP upregulation Base-excision repair of DNA damage Platinum chemotherapy Inflicts DNA damage via adducts and DNA crosslinking Inhibition of PARP Disables DNA base-excision repair Recombination repair Replication fork collapse Double-strand DNA break Cell deathCell survival PARPDNA damage NAD+ Nicotinamide + PAR BRCA1 BRCA2 PARP inhibitor PARG Repair enzymes Chains of PAR Pt
- 2. Selected, Recruiting Phase 3 Clinical Trials of PARP Inhibitors Access the activity, “Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors,” at PeerView.com/RKN40. PRACTICE AID NCT035192301 Ovarian Cancer Pamiparib vs placebo Patients with platinum-sensitive recurrent ovarian cancer; Planned N = 216 Primary endpoint: PFS NCT03522246 (ATHENA)2 Rucaparib + nivolumab as maintenance therapy following front-line platinum-based chemotherapy Patients with newly diagnosed ovarian cancer; Planned N = 1,012 Primary endpoint: Investigator-assessed PFS NCT02855944 (ARIEL-4)3 Rucaparib vs chemotherapy Patients with BRCA-mutant, relapsed, high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer; Planned N = 345 Primary endpoint: Investigator-assessed PFS by RECIST 1.1 NCT03740165 (KEYLYNK-001)4 Carboplatin/paclitaxel + pembrolizumab and maintenance olaparib as a first-line therapy Patients with BRCAnonmutated advance EOC, fallopian tube cancer, or primary peritoneal cancer; Planned N = 620 Primary endpoint: PFS and OS NCT037376435 Durvalumab + chemotherapy + bevacizumab and maintenance durvalumab + bevacizumab + olaparib Patients with newly diagnosed, stage III or IV nonmucinous ovarian cancer; Planned N = 912 Primary endpoint: PFS NCT025022666 Olaparib + cediranib maleate vs standard chemotherapy Patients with recurrent platinum-resistant/platinum- refractory ovarian, fallopian tube, or primary peritoneal cancer; Planned N = 680 Primary endpoint: PFS and OS NCT03602859 (FIRST)7 Ovarian Cancer (Cont’d) Breast Cancer Niraparib vs standard-of-care platinum-based therapy as first-line therapy Patients with mCRPC who have not received prior chemotherapy or NHAs for mCRPC; Planned N = 720 Primary endpoint: PFS NCT03598270 (ANITA)8 Platinum-based therapy + atezolizumab + niraparib Patients with recurrent ovarian, tubal, or peritoneal cancer and a platinum-treatment interval >6 mo; Planned N = 414 Primary endpoint: PFS NCT025022669 Olaparib + platinum-based chemotherapy as neoadjuvant therapy Patients withTNBC and/or gBRCA breast cancer; Planned N = 527 Primary endpoint: N withTEAEs, pCR rate + completion rate of olaparib, and pCR at surgery after neoadjuvant therapy
- 3. Selected, Recruiting Phase 3 Clinical Trials of PARP Inhibitors Access the activity, “Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors,” at PeerView.com/RKN40. PRACTICE AID Prostate Cancer NCT03395197 (TALAPRO-2)12 Talazoparib + enzalutamide vs enzalutamide + placebo Patients with assessment of DDR mutation status; metastatic disease in bone, ECOG PS 0 or 1; Planned N = 1,037 Primary endpoint: Confirm dose of talazoparib; radiographic PFS NCT02975934 (TRITON3)11 Rucaparib vs abiraterone, enzalutamide, or docetaxel Patients with mCRPC associated with homologous recombination deficiency; Planned N = 400 Primary endpoint: Radiographic PFS NCT03732820 (PROPEL)13 Prostate Cancer (Cont’d) Gastric Cancer Olaparib or placebo + abiraterone/prednisone as first-line therapy Patients with mCRPC who have not received prior chemotherapy or NHAs for mCRPC; Planned N = 720 Primary endpoint: Radiographic PFS NCT03834519 (KEYLYNK-010)14 Olaparib + pembrolizumab vs abiraterone acetate or enzalutamide Patients with mCRPC who have failed to respond to either abiraterone acetate or enzalutamide (but not both) and to chemotherapy; Planned N = 780 Primary endpoint: OS and radiographic PFS NCT0342781415 Pamiparib (BGB-290) vs placebo Patients with previously treated advanced or inoperable gastric cancer who have responded to first-line platinum therapy; Planned N = 540 Primary endpoint: PFS NCT03748641 (MAGNITUDE)10 Niraparib + abiraterone/ prednisone in front-line disease Patients with mCRPC; Planned N = 1,000 Primary endpoint: Radiographic PFS Lung Cancer NCT03976362 (KEYLYNK-008)16 Pembrolizumab with or without maintenance olaparib as first-line therapy Patients with metastatic, squamous non–small cell lung cancer; Planned N = 735 Primary endpoint: PFS per RECIST 1.1 and OS NCT03976323 (KEYLYNK-006)17 Pembrolizumab with maintenance olaparib or pemetrexed as first-line therapy Patients with metastatic, nonsquamous non–small cell lung cancer; Planned N = 792 Primary endpoint: PFS per RECIST 1.1 and OS NCT0351608418 Niraparib as maintenance therapy following first-line platinum-based chemotherapy Patients with extensive-stage disease small cell lung cancer; Planned N = 591 Primary endpoint: BICR-assessed PFS and OS
- 4. Selected, Recruiting Phase 3 Clinical Trials of PARP Inhibitors Access the activity, “Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors,” at PeerView.com/RKN40. PRACTICE AID Ovarian Cancer Selected Active, Not RecruitingTrials of PARP Inhibitors NCT02655016 (FIRST)21 Niraparib as maintenance therapy Patients with advanced ovarian cancer following response on front-line platinum-based therapy; N = 620 NCT03642132 (JAVELIN OVARIAN PARP 100)20 Avelumab + chemotherapy followed by avelumab + talazoparib as maintenance therapy Patients with previously untreated advanced ovarian cancer; N = 79 NCT0216369422 Breast Cancer Lung Cancer Veliparib vs placebo in combination with carboplatin + paclitaxel Patients with HER2-negative, metastatic or locally advanced, unresectable, BRCA-associated breast cancer; N = 513 NCT0203227723 Veliparib + carboplatin vs standard chemotherapy + carboplatin + standard chemotherapy Patients with early stageTNBC; N = 634 NCT0226499024 Veliparib + carboplatin + paclitaxel vs investigator’s choice of standard chemotherap Patients receiving first cytotoxic chemotherapy for metastatic or advanced NSq NSCLC + who are current/former smokers; N = 595 NCT02470585 (VELIA)19 Veliparib with carboplatin + paclitaxel ± continuation maintenance veliparib Patients with previously untreated stages III or IV high-grade serous EOC, fallopian tube, or primary peritoneal cancer; N = 1,140 NCT0215298225 Temozolomide with or without veliparib Patients with newly diagnosed glioblastoma multiforme; N = 440 NCT02184195 (POLO)26 Olaparib vs placebo as maintenance monotherapy Patients with gBRCA-mutated, metastatic pancreatic cancer whose disease has not progressed on first-line platinum-based chemotherapy; N = 154 NCT02987543 (PROfound)27 Olaparib vs enzalutamide or abiraterone acetate Patients with mCRPC who have failed prior treatment with an NHA and have HRR gene mutations; N = 340 Prostate CancerPancreatic CancerGlioblastoma Ovarian Cancer Selected Active, Not RecruitingTrials of PARP Inhibitors NCT02655016 (FIRST)21 Niraparib as maintenance therapy Patients with advanced ovarian cancer following response on front-line platinum-based therapy; N = 620 NCT03642132 (JAVELIN OVARIAN PARP 100)20 Avelumab + chemotherapy followed by avelumab + talazoparib as maintenance therapy Patients with previously untreated advanced ovarian cancer; N = 79 NCT0216369422 Breast Cancer Lung Cancer Veliparib vs placebo in combination with carboplatin + paclitaxel Patients with HER2-negative, metastatic or locally advanced, unresectable, BRCA-associated breast cancer; N = 513 NCT0203227723 Veliparib + carboplatin vs standard chemotherapy + carboplatin + standard chemotherapy Patients with early stageTNBC; N = 634 NCT0226499024 Veliparib + carboplatin + paclitaxel vs investigator’s choice of standard chemotherap Patients receiving first cytotoxic chemotherapy for metastatic or advanced NSq NSCLC + who are current/former smokers; N = 595 NCT02470585 (VELIA)19 Veliparib with carboplatin + paclitaxel ± continuation maintenance veliparib Patients with previously untreated stages III or IV high-grade serous EOC, fallopian tube, or primary peritoneal cancer; N = 1,140 NCT0215298225 Temozolomide with or without veliparib Patients with newly diagnosed glioblastoma multiforme; N = 440 NCT02184195 (POLO)26 Olaparib vs placebo as maintenance monotherapy Patients with gBRCA-mutated, metastatic pancreatic cancer whose disease has not progressed on first-line platinum-based chemotherapy; N = 154 NCT02987543 (PROfound)27 Olaparib vs enzalutamide or abiraterone acetate Patients with mCRPC who have failed prior treatment with an NHA and have HRR gene mutations; N = 340 Prostate CancerPancreatic CancerGlioblastoma BRCA: breast cancer gene; CR: complete response; EOC: epithelial ovarian cancer; gBRCAm: mutated germline BRCA gene; NHA: new hormonal agent; NSCLC: non–small cell lung cancer; NSq: nonsquamous; ORR: overall response rate; PFS: progression-free survival. 1. https://clinicaltrials.gov/ct2/show/NCT03519230. Accessed September 26, 2019. 2. https://clinicaltrials.gov/ct2/show/NCT03522246. Accessed September 26, 2019. 3. https://clinicaltrials.gov/ct2/show/ NCT02855944. Accessed September 26, 2019. 4. https://clinicaltrials.gov/ct2/show/NCT03740165. Accessed September 26, 2019. 5. https://clinicaltrials.gov/ct2/show/NCT03737643. Accessed September 26, 2019. 6. https://clinicaltrials.gov/ct2/show/NCT02502266. Accessed September 26, 2019. 7. https://clinicaltrials.gov/ct2/show/NCT03602859. Accessed September 26, 2019. 8. https://clinicaltrials.gov/ct2/show/NCT03598270. Accessed September 26, 2019. 9. https://clinicaltrials.gov/ct2/show/NCT03150576. Accessed September 26, 2019. 10. https://clinicaltrials.gov/ct2/show/ NCT03748641. Accessed September 26, 2019. 11. https://clinicaltrials.gov/ct2/show/NCT02975934. Accessed September 26, 2019. 12. https://clinicaltrials.gov/ct2/show/NCT03395197. Accessed September 26, 2019. 13. https://clinicaltrials.gov/ct2/show/NCT03732820. Accessed September 26, 2019. 14. https://clinicaltrials.gov/ct2/show/NCT03834519. Accessed September 26, 2019. 15. https://clinicaltrials.gov/ct2/show/NCT03427814. Accessed September 26, 2019. 16. https://clinicaltrials.gov/ct2/show/NCT03976362. Accessed September 26, 2019. 17. https://clinicaltrials.gov/ct2/show/ NCT03976323. Accessed September 26, 2019. 18. https://clinicaltrials.gov/ct2/show/NCT03516084. Accessed September 26, 2019. 19. https://clinicaltrials.gov/ct2/show/NCT02470585. Accessed October 8, 2019. 20. https://clinicaltrials.gov/ct2/show/NCT03642132. Accessed October 8, 2019. 21. https://clinicaltrials.gov/ct2/show/NCT02655016. Accessed October 8, 2019. 22. https://clinicaltrials.gov/ct2/ show/NCT02163694. Accessed October 8, 2019. 23. https://clinicaltrials.gov/ct2/show/NCT02032277. Accessed October 8, 2019. 24. https://clinicaltrials.gov/ct2/show/NCT02264990. Accessed October 8, 2019. 25. https://clinicaltrials.gov/ct2/show/NCT02152982. Accessed October 8, 2019. 26. https://clinicaltrials.gov/ct2/show/NCT02184195. Accessed October 8, 2019. 27. https://clinicaltrials.gov/ct2/ show/NCT02987543. Accessed October 8, 2019.
- 5. FDA-Approved PARP Inhibitors for the Treatment of Solid Tumors Access the activity, “Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors,” at PeerView.com/RKN40. PRACTICE AID AML: acute myeloid leukemia; ALT: alanine transaminase; AST: aspartate aminotransferase; BRCA: breast cancer gene; CR: complete response; gBRCAm: mutated germline BRCA gene; HER2: human epidermal growth factor receptor 2; MDS: myelodysplastic syndromes; PARP: poly(ADP)-ribose polymerase; PR: partial response; sBRCAm: mutated somatic BRCA gene; tx: treatment. a Patients with hormone receptor (HR)-positive breast cancer should have been treated with a prior endocrine therapy or be considered inappropriate for endocrine therapy. 1. https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/InVitroDiagnostics/ucm301431.htm. Accessed September 23, 2019. Agent Niraparib Indicated in patients with advanced ovarian cancer who have been treated with ≥3 prior chemotherapy regimens and whose cancer is associated with homologous recombination deficiency (HRD)–positive status HRD status testing using myChoice® CDx No companion diagnostic No companion diagnostic BRCA1/2 testing using BRACAnalysis CDx® or next-gen sequencing using FoundationOne CDx® BRCA1/2 testing using BRACAnalysis CDx® or next-gen sequencing using FoundationOne CDx® BRCA1/2 testing using BRACAnalysis CDx® or next-gen sequencing using FoundationFocus™ CDx BRCA or FoundationOne CDx® BRCA1/2 testing using BRACAnalysis CDx® BRCA1/2 testing using BRACAnalysis CDx® Indicated as maintenance tx in pts with gBRCAm or sBRCAm advanced ovarian cancer following response to first-line platinum-based chemotherapy Indicated in pts with gBRCAm or sBRCAm advanced ovarian cancer previously treated with ≥2 chemotherapies Indicated as maintenance tx in pts with recurrent ovarian cancer, achieving CR or PR to platinum-based chemotherapy Indicated in pts with gBRCAm, HER2-negative, metastatic breast cancer who have been treated with chemotherapy in the neoadjuvant, adjuvant, or metastatic settinga Indicated as maintenance tx in pts with recurrent ovarian cancer, achieving CR or PR to platinum-based chemotherapy Indicated in gBRCAm advanced ovarian cancer previously treated with ≥3 chemotherapies Indicated in pts with gBRCAm, HER2-negative, locally advanced or metastatic breast cancer Common AEs Hematologic: anemia, thrombocytopenia, neutropenia, lymphocytopenia Gastrointestinal: nausea, vomiting, diarrhea, constipation, abdominal pain, dysgeusia, decreased appetite Constitutional: fatigue, asthenia, dyspnea Laboratory abnormalities: elevated creatinine, ALT, AST, and cholesterol Serious AEs Bone marrow suppression and MDS/AML Lung-related: pneumonitis Cardiovascular effects: hypertension and hypertensive crisis Embryo-fetal toxicity Olaparib Rucaparib Talazoparib Spectrum of Adverse Events With PARP Inhibitors Indications Companion Diagnostic No companion diagnostic Indicated as maintenance tx in recurrent epithelial ovarian cancer who are in a complete or partial response to platinum-based chemotherapy
