Composing Personalized HCC Treatment Strategies: Insights on Harmonizing Patient Care With a Multidisciplinary Ensemble
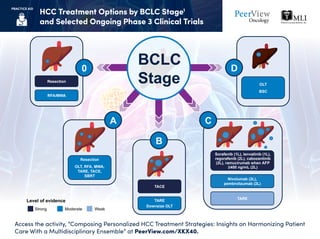
- 1. HCC Treatment Options by BCLC Stage1 and Selected Ongoing Phase 3 Clinical Trials Access the activity, “Composing Personalized HCC Treatment Strategies: Insights on Harmonizing Patient Care With a Multidisciplinary Ensemble” at PeerView.com/XKX40. PRACTICE AID BCLC StageResection OLT BSC Resection OLT, RFA, MWA, TARE, TACE, SBRT TARE Nivolumab (2L), pembrolizumab (2L) Sorafenib (1L), lenvatinib (1L), regorafenib (2L), cabozantinib (2L), ramucirumab when AFP ≥400 ng/mL (2L) TACE TARE Downsize OLT Level of evidence Strong Moderate Weak 0 A B C D RFA/MWA
- 2. AFP: alpha-fetoprotein; BCLC: Barcelona Clinic Liver Cancer; BSC: best supportive care; FFLP: freedom from local progression; HCC: hepatocellular carcinoma; MWA: microwave ablation; OLT: orthotopic liver transplantation; OS: overall survival; PFS: progression-free survival; RFA: radiofrequency ablation; RFS: recurrence-free survival; SBRT: stereotactic body radiation therapy; TACE: transarterial chemoembolization; TARE: transarterial radioembolization. 1. Marrero JA et al. Hepatology. 2018;68:723-750. HCC Treatment Options by BCLC Stage1 and Selected Ongoing Phase 3 Clinical Trials Access the activity, “Composing Personalized HCC Treatment Strategies: Insights on Harmonizing Patient Care With a Multidisciplinary Ensemble” at PeerView.com/XKX40. PRACTICE AID Ongoing Trials of First-Line Systemic Therapy in Intermediate/Advanced HCC Ongoing Trials in Resected or Locally Advanced HCC • Residual or recurrent disease after TACE • Primary endpoint: FFLP • Estimated N = 160 NCT02762266 SBRT TACEvs • Patients with high risk of recurrence after resection • Primary endpoint: RFS • Estimated N = 530 CheckMate -9DX (NCT03383458) • Locoregional not amenable to curative therapy • Primary endpoint: PFS • Estimated N = 600 EMERALD-1 (NCT03778957) Durvalumab ± bevacizumab + TACE Placebo + TACEvs • Complete radiological response after surgical resection or local ablation • Primary endpoints: RFS and OS • Estimated N = 950 KEYNOTE-937 (NCT03867084) Pembrolizumab (adjuvant) PlacebovsNivolumab Placebovs • Primary endpoint: OS • Estimated N = 1,310 HIMALAYA (NCT03298451) Durvalumab ± tremelimumab Sorafenibvs • Primary endpoint: OS • Estimated N = 368 NCT01730937 • Primary endpoints: PFS and OS • Estimated N = 480 IMbrave150 (NCT03434379) Atezolizumab + bevacizumab Sorafenibvs • Primary endpoints: PFS and OS • Estimated N = 740 COSMIC-312 (NCT03755791) Cabozantinib ± atezolizumab Sorafenibvs SBRT + sorafenib Sorafenibvs • Primary endpoints: PFS and OS • Estimated N = 750 LEAP-002 (NCT03713593) Pembrolizumab + lenvatinib Placebo + lenvatinib vs • Patients with high risk of recurrence after resection • Primary endpoint: RFS • Estimated N = 888 EMERALD-2 (NCT03847428) Durvalumab ± bevacizumab Placebovs • Primary endpoint: OS • Estimated N = 1,084 CheckMate -9DW (NCT04039607) Nivolumab + ipilimumab Sorafenib or lenvatinib vs
- 3. a Phase 3 level of evidence for all listed agents. b Real-world effectiveness data in extended populations, including in patients with Child–Pugh B cirrhosis, are available. AE: adverse event; AFP: alpha-fetoprotein; ECOG: Eastern Cooperative Oncology Group; HCC: hepatocellular carcinoma; HFSR: hand-foot skin reaction; TKI: tyrosine kinase inhibitor. 1. Adapted from slides presented by Amit Singal, MD, MS. 2. Llovet JM et al. NEnglJMed. 2008;359:378-390. 3. Kudo M et al. Lancet. 2018;391:1163-1173. 4. Bruix J et al. Lancet. 2017 Jan 7;389:56-66. 5. Abou-Alfa GK et al. NEnglJMed. 2018 Jul 5;379:54-63. 6. Zhu AX et al.LancetOncol. 2019;20:282-296. Considerations for Selecting and Sequencing Targeted Therapy for HCC1,a Access the activity, “Composing Personalized HCC Treatment Strategies: Insights on Harmonizing Patient Care With a Multidisciplinary Ensemble” at PeerView.com/XKX40. PRACTICE AID Drug • Child–Pugh A cirrhosis, ECOG 0-1, unresectable HCC with no prior systemic therapy • Excluded patients with >50% liver involvement, main portal vein tumor thrombus, and bile duct invasion • Non-inferior OS compared with sorafenib • Improved objective responses and time to progression compared with sorafenib Lenvatinib • Orally once daily • Can be taken with or without food • Increased hypertension, proteinuria, and anorexia • Tolerated sorafenib but with radiographic progression • Improved OS Regorafenib • Orally daily for 3 weeks with a 1-week holiday • Similar to AE profiles of other TKIs • Improved survival compared with placebob • Child–Pugh A cirrhosis, ECOG 0-1, unresectable HCC with no prior systemic therapy • Orally twice daily • Should be taken 1-2 hours removed from food • Increased HFSR • Intolerant to sorafenib or with radiographic progression • Could have received an additional line of systemic therapy • Improved OS Cabozantinib • Orally once daily • Similar to AE profiles of other TKIs • Intolerant to sorafenib or with radiographic progression • Patients with AFP ≥400 ng/mL • Improved OS Ramucirumab • IV infusion every 2 weeks • Well tolerated with low rates of dose reductions or discontinuations Sorafenib Inclusion Criteria Efficacy AE Profile Dosing First-LineSetting2,3 Second-LineSetting4-6
