Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook
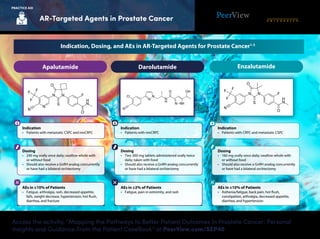
- 1. Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 AR-Targeted Agents in Prostate Cancer PRACTICE AID Indication • Patients with metastatic CSPC and nmCRPC Dosing • 240 mg orally once daily; swallow whole with or without food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy AEs in ≥10% of Patients • Fatigue, arthralgia, rash, decreased appetite, falls, weight decrease, hypertension, hot flush, diarrhea, and fracture Indication • Patients with CRPC and metastatic CSPC AEs in ≥10% of Patients • Asthenia/fatigue, back pain, hot flush, constipation, arthralgia, decreased appetite, diarrhea, and hypertension Dosing • 160 mg orally once daily; swallow whole with or without food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy Apalutamide EnzalutamideDarolutamide Indication • Patients with nmCRPC Dosing • Two 300-mg tablets administered orally twice daily; taken with food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy AEs in ≥2% of Patients • Fatigue, pain in extremity, and rash Indication, Dosing, and AEs in AR-Targeted Agents for Prostate Cancer1-3
- 2. Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 AR-Targeted Agents in Prostate Cancer PRACTICE AID a OS not reached in apalutamide or placebo groups as of May 15, 2019. ADT: androgen deprivation therapy; AE: adverse event; AR: androgen receptor; CRPC: castration-resistant prostate cancer; CSPC: castration-sensitive prostate cancer; CT: computed tomography; ECOG PS: Eastern Cooperative Oncology Group Performance Status; GnRH: gonadotropin- releasing hormone; HSPC: hormone-sensitive prostate cancer; MFS: metastasis-free survival; mHSPC: metastatic hormone-sensitive prostate cancer; nmCRPC: nonmetastatic castration-resistant prostate cancer; PSA: prostate-specific antigen; PSADT: prostate-specific antigen doubling time; rPFS: radiographic progression-free survival. 1. Erleada (apalutamide) Prescribing Information. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/ERLEADA-pi.pdf. Accessed January 28, 2020. 2. Xtandi (enzalutamide) Prescribing Information. https://www.astellas.us/docs/12A005-ENZ- WPI.PDF. Accessed January 28, 2020. 3. Nubeqa (darolutamide) Prescribing Information. http://labeling.bayerhealthcare.com/html/products/pi/Nubeqa_PI.pdf. Accessed January 28, 2020. 4. Small EJ et al. Ann Oncol. 2019;30:1813-1820. 5. Smith MR et al. N Engl J Med. 2018;378:1408- 1418. 6. Hussain M et al. N Engl J Med. 2018;378:2465-2474. 7. Fizazi K et al. American Society of Clinical Oncology 2019 Genitourinary Cancers Symposium (ASCO GU 2019). Abstract 140. 8. Chi KN et al. 2019 American Society of Clinical Oncology Annual Meeting (ASCO 2019). Abstract 5006. 9. https://clinicaltrials.gov/ct2/show/NCT02489318. Accessed January 28, 2020. 10. https://clinicaltrials.gov/ct2/show/NCT02677896. Accessed January 28, 2020. 11. Armstrong AJ et al. ASCO GU 2019. Abstract 687. 12. https://www.ascopost.com/issues/april-10-2019-supplement- conference-highlights-gugi-2019/interim-analysis-of-the-arches-trial/. Accessed January 28, 2020. 13. https://clinicaltrials.gov/ct2/show/NCT02799602. Accessed January 28, 2020. • nmCRPC • Rising PSA despite castrate testosterone level (≤50 ng/dL) • PSADT ≤10 mo Apalutamide 40.5 mo Placebo 16.2 mo 1° endpoint: MFS vs MFS SPARTAN Apalutamide + ADT Placebo + ADT • nmCRPC • Rising PSA despite castrate testosterone level (≤50 ng/dL) • Baseline PSA ≥2 ng/mL; PSADT ≤10 mo Enzalutamide 36.6 mo Placebo 14.7 mo 1° endpoint: MFS vs MFS PROSPER Enzalutamide + ADT Placebo + ADT • nmCRPC • Castrate level of serum testosterone (<50 ng/dL) • Baseline PSA ≥2 ng/mL; PSADT ≤10 mo Darolutamide 40.4 mo Placebo 18.4 mo 1° endpoint: MFS vs MFS ARAMIS Darolutamide + ADT Placebo + ADT AR-Targeted Agents in Nonmetastatic CRPC4-7 • mHSPC • Newly diagnosed or previously treated • ECOG PS 0 or 1 Apalutamide Not Reached Enzalutamide Not Reached Placebo 21.1 mo 1° endpoint: Median rPFS and OSa vs rPFS TITAN Apalutamide + ADT Placebo + ADT • mHSPC (confirmed by bone scan, CT, or MRI) • ECOG PS 0 or 1 Placebo 19.45 mo 1° endpoint: rPFS vs rPFS ARCHES Enzalutamide + ADT Placebo + ADT • mHSPC • Newly diagnosed • ECOG PS 0 or 1 1° endpoint: OS vs ARASENS Darolutamide + ADT + docetaxel Placebo + ADT + docetaxel AR-Targeted Agents in Metastatic HSPC8-13 Ongoing; primary completion date: August 2022
- 3. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 TOPARP-B (NCT01682772); Multi-stage phase 2 design RESULTS • mCRPC; ongoing ADT or prior bilateral orchiectomy • Previously treated with 1 or 2 lines of taxane-based chemo and/or AR-directed therapy • N = 98 PROfound (NCT02987543); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • Previously treated with AR-targeted therapy • N = 387 RR; evaluable N = 98 Endpoints: ORR 54% in 400 mg, 37% in 300 mg olaparib cohort Median PFS: 5.4 mo Primary endpoint per gene subgroup: BRCA1/2: 80% PALB2: 57% RESULTS Olaparib: median rPFS 7.39 mo Physician's choice: rPFS 3.55 mo HR = 0.34 (95% CI, 0.25-0.47); P < .0001 Olaparib 1° endpoint: RR 1° endpoint: rPFS Olaparib + abiraterone Placebo + abiraterone vs vs 1° endpoint: rPFS Galahad (NCT02854436); Phase 2 RESULTS RESULTS • mCRPC previously treated with ≥1 line of taxane-based chemo; received ≥1 line of AR-targeted therapy • DDR anomalies • Planned N = 301 N = 81 (46 with BRCA1/2 and 35 with non-BRCA1/2) ORR 41% in BRCA1/2; 9% in non-BRCA1/2 Composite response rate 63% in BRCA1/2; 17% in BRCA1/2 PSA response rate 50% in BRCA1/2; 3% in non-BRCA1/2 Niraparib 1° endpoint: ORR TRITON2 (NCT02952534); Phase 2 • mCRPC; progression on AR-directed therapy and 1 prior taxane; HRR gene aberration • No prior PARP inhibitor, mitoxantrone, cyclophosphamide, or platinum-based chemo • Planned N = 360 ORR BRCA1/2 pts, n = 69: 47.5% PSA response BRCA1/2 pts, n = 69: 53.6% Rucaparib 1° endpoints: ORR, PSA response Selected Ongoing Trials of PARP Inhibitors1-6 ATM: 37% CDK12: 25% NCT01972217; Phase 2 RESULTS • mCRPC • ECOG PS 0 or 1 • ≤2 prior lines of chemo • Planned N = 158 rPFS; evaluable N = 142 Olaparib: 13.8 mo Placebo: 8.2 mo HR = 0.65; P = .034 Enzalutamide or abiraterone Olaparib
- 4. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 Talazoparib + enzalutamide vs 1° endpoints: dose, rPFS Placebo + enzalutamide Selected Ongoing Trials of PARP Inhibitors1 (Cont’d) PROpel (NCT03732820); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • ECOG PS 0 or 1 • Assessment of HRR gene aberrations • Planned N = 720 vs 1° endpoint: rPFS Placebo + abiraterone Olaparib + abiraterone vs 1° endpoint: rPFS Placebo + abiraterone Olaparib + abiraterone MAGNITUDE (NCT03748641); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • Planned N = 1,000 TRITON3 (NCT02975934); Phase 3 • mCRPC previously treated with 1 next-generation AR-targeted therapy • Deleterious mutation in BRCA1/2 or ATM • Planned N = 400 vs 1° endpoint: rPFS Abiraterone or enzalutamide or docetaxel Rucaparib Avelumab + bempegaldesleukin Recruiting Recruiting Recruiting Recruiting Recruiting Rucaparib 1° endpoint: ORR 1° endpoints: confirmed OR, PSA response, DLTs LODESTAR (NCT04171700); Phase 2, open-label • Unresectable, locally advanced, or metastatic solid tumor and relapsed/progressive disease • At least 1 prior line of therapy extending OS or SOC therapy for advanced disease • mCRPC with BRCA1/2 mutations • ECOG PS 0 or 1 • Planned N = 220 (with solid tumors) Avelumab + bempegaldesleukin (NKTR-214) + talazoparib or enzalutamide (NCT04052204); Phase 2, open-label • mCRPC • ECOG PS 0 or 1 • Planned N = 160 • Cohort A: deleterious mutations in BRACA1/2 • Cohort B: non-BRACA1/2 mutations • Combination B: mCRPC with DDR defect: + talazoparib • Combination C: + enzalutamide Recruiting Recruiting Talazoparib 1° endpoint: ORR TALAPRO-1 (NCT03148795); Phase 2, open-label • mCRPC; metastatic disease in bone • Assessment of DDR mutation status • ECOG PS 0 to 2 • Planned N = 100 TALAPRO-2 (NCT03395197); Phase 3 • mCRPC; metastatic disease in bone • Assessment of DDR mutation status • ECOG PS 0 or 1 • Planned N = 1,037
- 5. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 Selected Ongoing Trials of PARP Inhibitors Combined With PD-1/PD-L1 Inhibitors1,7-10 JAVELIN PARP MEDLEY (NCT03330405); Phase 2 • Locally advanced or mCRPC • Primary or metastatic tumor biopsy • ECOG PS 0 or 1 • Planned N = 242 CheckMate -9KD (NCT03338790); Phase 2 • mCRPC; ongoing ADT • Plasma and fresh or archival tumor tissue • ECOG PS 0-1 CheckMate -7DX (NCT04100018); Phase 3 • Stage IV prostate cancer • ECOG PS 0-1 • Ongoing ADT • Chemotherapy-naïve and received 1-2 prior second-generation hormonal therapies Recruiting Recruiting Recruiting 1° endpoints: DLTs, OR 1° endpoints: safety, ORR, composite RR • HRD status (must be available before tx arm assignment) • Planned N = 330 1° endpoints: ORR, PSA response • Planned N = 984 1° endpoints: rPFS, OS Nivolumab or placebo + docetaxel in CRPC Avelumab + talazoparib Nivolumab + rucaparib or docetaxel or enzalutamide KEYNOTE-365 (NCT02861573); Phase 1b/2 • mCRPC; ongoing ADT • Tissue biopsy from site not previously irradiated • Planned N = 400 QUEST (NCT03431350); Phase 1/2 • mCRPC • DDR gene anomalies • Prior novel AR-targeted therapy • Planned N = 80 NCT02484404; Phase 2 (prostate cohort) • mCRPC; ongoing ADT or prior bilateral orchiectomy • ECOG PS 0 or 1; previously treated with enzalutamide and/or abiraterone • Planned N = 384 Recruiting Niraparib + cetrelimab RESULTS PSA response Pembro + olaparib, n = 41: 12% Pembro + docetaxel, n = 72: 31% Pembro + enzalutamide, n = 69: 27% 1° endpoint: PSA response Pembrolizumab + olaparib or docetaxel/prednisone or enzalutamide or abiraterone 1° endpoints: dose, safety Durvalumab + olaparib RESULTS rPFS; evaluable N = 17 Durvalumab + olaparib: 16.1 mo 12-month rPFS: 51.5% 9/17 (53%) patients had a radiographic and/or PSA response ADT: androgen deprivation therapy; AR: androgen receptor; DDR: DNA damage repair; DLT: dose-limiting toxicity; ECOG PS: Eastern Cooperative Oncology Group Performance Status; HRD: homologous recombination deficiency; HRR: homologous recombination repair; mCRPC: metastatic castration-resistant prostate cancer; OR: overall response; ORR: objective response rate; PARP: poly (ADP-ribose) polymerase; PD-1: programmed cell death protein 1; PD-L1: programmed death ligand 1; PSA: prostate-specific antigen; rPFS: radiographic progression-free survival ; RR: response rate. 1. https://clinicaltrials.gov. Accessed January 28, 2020. 2. Mateo J et al. 2019 American Society of Clinical Oncology Annual Meeting (ASCO 2019). Abstract 5005. 3. Clarke N et al. Lancet Oncol. 2018;19:975-986. 4. Hussain M et al. European Society for Medical Oncology Congress 2019 (ESMO 2019). Abstract LBA12_PR. 5. Abida W et al. ESMO 2019. Abstract 846PD. 6. Smith MR et al. American Society of Clinical Oncology 2019 Genitourinary Cancers Symposium (ASCO GU 2019). Abstract 202. 7. Yu EY et al. J Clin Oncol. 37;2019. Abstract 5027. 8. Massard C et al. J Clin Oncol. 37;2019. Abstract 5029. 9. Fong P et al. J Clin Oncol. 37;2019. Abstract 5010. 10. Karzai F et al. J Immunother Cancer. 2018;6:141.
- 6. Improving Patient Outcomes in Prostate Cancer: A Patient CaseBook for Physicians Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 PRACTICE AID Case 1 63-Year-Old ManWith nmCRPC • pT3bN0 Gleason 4+3 prostate cancer s/p prostatectomy (2013) and salvage radiation therapy (2015) • Has been on salvage ADT since 2016 – Pretreatment PSA: 12.7 ng/mL; PSA nadir: <0.10 ng/mL • Latest PSA: 2.7 ng/mL; PSADT: 5 months • Bone scan and abdominal-pelvic CT with no detectable metastases Diagnosis: Nonmetastatic castration-resistant prostate cancer Next Steps: Start one of the novel AR inhibitors (apalutamide, enzalutamide, or darolutamide) Case 2 59-Year-Old ManWith CRPC • pT3aN1 prostate cancer, Gleason 4+5 • Post-operative PSA elevated at 2.3 ng/mL • Has been on salvage ADT for the last 5 years – PSA nadir: <0.10 ng/mL • Latest PSA: 5.7 ng/mL; PSADT: 4.9 months • Bone scan and abdominal-pelvic CT with no detectable metastases • PSMA PET-CT with multiple subcentimeter PSMA-avid nodes (para-aortic, mediastinal, and left supraclavicular) Diagnosis: Nonmetastatic castration-resistant prostate cancer (Bone scan and abdominal pelvic CT showed no detectable metastases, so by the definitions of the trials he would be nmCRPC) Next Steps: Start one of the novel AR inhibitors (apalutamide, enzalutamide, or darolutamide). SPARTAN, PROSPER, and ARAMIS trials actually provide the best evidence for treating patients with CRPC and metastases that are detectable only by sensitive next-generation imaging modalities
- 7. Improving Patient Outcomes in Prostate Cancer: A Patient CaseBook for Physicians Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 PRACTICE AID Case 3 69-Year-Old ManWith mCSPC • Presents with urinary obstructive symptoms • Past medical history: Diabetes for 15 years, well controlled on metformin • Family history: Father and brother with history of prostate cancer • Examination: Enlarged nodular prostate on rectal exam • Labs: Elevated PSA (25 ng/mL) • Prostate biopsy: Adenocarcinoma of prostate, Gleason 4+4 • CT abdomen and pelvis: Enlargement of multiple retroperitoneal lymph nodes • Bone scan: 2 osteoblastic lesions (T4, T9 vertebral body lesions) Diagnosis: Metastatic castration-sensitive prostate cancer (low volume/low risk) For Refill times Date What would you recommend for this patient? • Treatment: Bicalutamide 50 mg daily x 2 weeks and leuprolide 22.5 mg IM started the same day. Pre-authorization for abiraterone + prednisone started • 1-month follow-up: Abiraterone 1,000 mg + prednisone 5 mg daily started • 2-month follow-up: Mild fatigue, right upper quadrant discomfort • Vitals: BP 155/95 mmHg • Labs: Elevated liver enzymes (ALT: 550 units/L; AST: 500 units/L) Diagnosis: Liver toxicity and hypertension due to abiraterone Next Steps: Abiraterone discontinued; apalutamdide 240 mg daily started
- 8. Improving Patient Outcomes in Prostate Cancer: A Patient CaseBook for Physicians Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 PRACTICE AID Case 4 65-Year-Old ManWith mCSPC • Presents to ED with right hip/right lower quadrant pain • Past medical history: Hypertension, diabetes, currently not on any medications • Exam: Decreased movement in the right hip • CT scan of the abdomen and pelvis: Impending pathologic fracture in the right femur shaft; multiple sclerotic bone lesion in the right and left femur; retroperitoneal lymadenopathy • Bone scan: Widespread osteoblastic bone metastasis (>25 bone lesions) • Serum PSA level: 550 ng/mL • Prostate biopsy: Prostate adenocarcinoma, Gleason score 5+4 Diagnosis: Metastatic castration-sensitive prostate cancer (high volume/ high risk) For Refill times Date What would you recommend for this patient? Treatment: Bicalutamide 50 mg daily for 4 weeks, leuprolide 22.5 mg IM 2 weeks into bicalutamide. Docetaxel is started on the day of completion of bicalutamide every 3 weeks x 6. Leuprolide is continued every 3 months 6-month follow-up: Mild fatigue; tingling and numbness in fingertips and toes (mild sensory neuropathy); PSA: 0.4 ng/mL; patient asks about the role of adding enzalutamide or apalutamide to treatment
- 9. Improving Patient Outcomes in Prostate Cancer: A Patient CaseBook for Physicians Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 PRACTICE AID Case 5 58-Year-Old ManWith mCRPC • Presents for second opinion with widespread mCRPC • Received all FDA-approved agents that prolong survival for mCRPC; still has an ECOG PS of 1 • Hoping for clinical trial options • Upon taking a family history, you find out he has a significant family history of cancer, with 3 women on his mother’s side who died at a young age from breast cancer Patient: Prostate Cancer Family History of Breast Cancer Died car accident Died car accident Died car accident Died car accident Prostate cancer Dx 58 Prostate cancer Dx 58 Died (35 y) breast cancer Died (33 y) breast cancer Died breast cancer
- 10. Improving Patient Outcomes in Prostate Cancer: A Patient CaseBook for Physicians Access the activity, “Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook” at PeerView.com/SEP40 PRACTICE AID Recommended Screening for Risk Reduction Died car accident Referred patient's daughter to breast/ovarian prevention clinic, had RRO Referred patient's nephews for prostate cancer screening Prostate cancer Dx 58 Died car accident Died (35 y) breast cancer Died (33 y) breast cancer Died breast cancer For Refill times Date What should you consider now? Germline DNA repair gene testing Microsatellite instability testing Somatic tumor testing with next-generation sequencing Referral to a genetic counselor ADT: androgen deprivation therapy; AR: androgen receptor; CRPC: castration-resistant prostate cancer; ECOG PS: Eastern Cooperative Oncology Group Performance Status; ED: emergency department; mCRPC: metastatic castration-resistant prostate cancer; mCSPC: metastatic castration-sensitive prostate cancer; nmCRPC: nonmetastatic castration-resistant prostate cancer; PSA: prostate-specific antigen; PSADT: prostate-specific antigen doubling time; PSMA: prostate-specific membrane antigen.
