How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape.
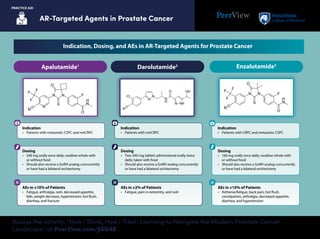
- 1. AR-Targeted Agents in Prostate Cancer PRACTICE AID Access the activity, “How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape” at PeerView.com/JGU40 Indication • Patients with metastatic CSPC and nmCRPC Dosing • 240 mg orally once daily; swallow whole with or without food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy AEs in ≥10% of Patients • Fatigue, arthralgia, rash, decreased appetite, falls, weight decrease, hypertension, hot flush, diarrhea, and fracture Indication • Patients with CRPC and metastatic CSPC AEs in ≥10% of Patients • Asthenia/fatigue, back pain, hot flush, constipation, arthralgia, decreased appetite, diarrhea, and hypertension Dosing • 160 mg orally once daily; swallow whole with or without food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy Apalutamide1 Enzalutamide3 Darolutamide2 Indication • Patients with nmCRPC Dosing • Two 300-mg tablets administered orally twice daily; taken with food • Should also receive a GnRH analog concurrently or have had a bilateral orchiectomy AEs in ≥2% of Patients • Fatigue, pain in extremity, and rash Indication, Dosing, and AEs in AR-Targeted Agents for Prostate Cancer
- 2. AR-Targeted Agents in Prostate Cancer PRACTICE AID a Final analysis after 254 deaths (15.5% darolutamide and 19.1% placebo). b OS not reached in apalutamide or placebo groups as of May 15, 2019. ADT: androgen deprivation therapy; AE: adverse event; AR: androgen receptor; CRPC: castration-resistant prostate cancer; CSPC: castration-sensitive prostate cancer; CT: computed tomography; ECOG PS: Eastern Cooperative Oncology Group Performance Status; GnRH: gonadotropin- releasing hormone; HSPC: hormone-sensitive prostate cancer; MFS: metastasis-free survival; mHSPC: metastatic hormone-sensitive prostate cancer; nmCRPC: nonmetastatic castration-resistant prostate cancer; PSA: prostate-specific antigen; PSADT: prostate-specific antigen doubling time; rPFS: radiographic progression-free survival. 1. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/ERLEADA-pi.pdf. 2. http://labeling.bayerhealthcare.com/html/products/pi/Nubeqa_PI.pdf. 3. https://www.astellas.us/docs/12A005-ENZ-WPI.PDF. 4. Small EJ et al. Ann Oncol. 2019;30:1813- 1820. 5. Smith MR et al. N Engl J Med. 2018;378:1408-1418. 6. Hussain M et al. N Engl J Med. 2018;378:2465-2474. 7. Fizazi K et al. American Society of Clinical Oncology 2019 Genitourinary Cancers Symposium (ASCO GU 2019). Abstract 140. 8. Small EJ et al. 2020 ASCO Virtual Scientific Program (ASCO 2020). Abstract 5516. 9. Fizazi K et al. ASCO 2020. Abstract 5514. 10. Sternberg CN et al. ASCO 2020. Abstract 5515. 11. Sternberg CN et al. N Engl J Med. 2020;382:2197-2206. 12. Chi KN et al. N Engl J Med. 2019;381:13-24. 13. https://clinicaltrials.gov/ct2/show/NCT02489318. 14. https://clinicaltrials.gov/ct2/show/NCT02799602. 15. https://clinicaltrials.gov/ct2/show/NCT02677896. 16. Armstrong AJ et al. ASCO GU 2019. Abstract 687. 17. https://www.ascopost.com/issues/april-10-2019-supplement-conference-highlights-gugi-2019/interim-analysis-of-the- arches-trial. Access the activity, “How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape” at PeerView.com/JGU40 • nmCRPC • Rising PSA despite castrate testosterone level (≤50 ng/dL) • PSADT ≤10 mo Apalutamide 40.5 mo Placebo 16.2 mo 1° endpoint: MFS Median OS, mo vs MFS Apalutamide 73.9 mo Placebo 59.9 mo mOS SPARTAN4,5,8 Apalutamide + ADT Placebo + ADT • nmCRPC • Rising PSA despite castrate testosterone level (≤50 ng/dL) • Baseline PSA ≥2 ng/mL; PSADT ≤10 mo Enzalutamide 36.6 mo Placebo 14.7 mo 1° endpoint: MFS Median OS, mo vs MFS Enzalutamide 67.0 mo Placebo 56.3 mo mOS PROSPER6,10,11 Enzalutamide + ADT Placebo + ADT • nmCRPC • Castrate level of serum testosterone (<50 ng/dL) • Baseline PSA ≥2 ng/mL; PSADT ≤10 mo 1° endpoint: MFS Median OS, moa vs Darolutamide 40.4 mo Placebo 18.4 mo Darolutamide NR Placebo NR MFS mOS ARAMIS7,9 Darolutamide + ADT Placebo + ADT AR-Targeted Agents in Nonmetastatic CRPC • mCSPC • Newly diagnosed or previously treated • ECOG PS 0 or 1 Apalutamide Not Reached Enzalutamide Not Reached Placebo 21.1 mo 1° endpoint: Median rPFS and OSb vs rPFS TITAN12,13 Apalutamide + ADT Placebo + ADT • mCSPC (confirmed by bone scan, CT, or MRI) • ECOG PS 0 or 1 Placebo 19.45 mo 1° endpoint: rPFS vs rPFS ARCHES15-17 Enzalutamide + ADT Placebo + ADT • mCSPC • Newly diagnosed • ECOG PS 0 or 1 1° endpoint: OS vs ARASENS14 Darolutamide + ADT + docetaxel Placebo + ADT + docetaxel AR-Targeted Agents in Metastatic CSPC Ongoing; primary completion date: August 2022
- 3. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape” at PeerView.com/JGU40 Galahad (NCT02854436); Phase 2 RESULTS • mCRPC previously treated with ≥1 line of taxane-based chemo; received ≥1 line of AR-targeted therapy • DDR anomalies • Planned N = 301 N = 81 (46 with BRCA1/2 and 35 with non-BRCA1/2) ORR 41% in BRCA1/2; 9% in non-BRCA1/2 Composite response rate 63% in BRCA1/2; 17% in BRCA1/2 PSA response rate 50% in BRCA1/2; 3% in non-BRCA1/2 Niraparib 1° endpoint: ORR Selected Ongoing Trials of PARP Inhibitors1-8 TOPARP-B (NCT01682772); Multi-stage phase 2 design RESULTS • mCRPC; ongoing ADT or prior bilateral orchiectomy • Previously treated with 1 or 2 lines of taxane-based chemo and/or AR-directed therapy • N = 98 RR; evaluable N = 98 Endpoints: ORR 54% in 400 mg, 37% in 300 mg olaparib cohort Median PFS: 5.4 mo Primary endpoint per gene subgroup: BRCA1/2: 80% PALB2: 57% Olaparib 1° endpoint: RR ATM: 37% CDK12: 25% Olaparib + abiraterone Placebo + abiraterone vs 1° endpoint: rPFS NCT01972217; Phase 2 RESULTS • mCRPC • ECOG PS 0 or 1 • ≤2 prior lines of chemo • Planned N = 158 rPFS; evaluable N = 142 Olaparib: 13.8 mo Placebo: 8.2 mo HR = 0.65; P = .034 PROfound (NCT02987543); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • Previously treated with AR-targeted therapy • N = 387 RESULTS Olaparib: median rPFS 7.39 mo Physician's choice: rPFS 3.55 mo HR = 0.34 (95% CI, 0.25-0.47); P < .0001 1° endpoint: rPFSvs Enzalutamide or abiraterone Olaparib FDA Approved May 20207,a TRITON2 (NCT02952534); Phase 2 • mCRPC; progression on AR-directed therapy and1 prior taxane; HRR gene aberration • No prior PARP inhibitor, mitoxantrone, cyclophosphamide, or platinum-based chemo • Planned N = 360 RESULTS ORR BRCA1/2 pts, n = 57; 43.9% PSA response BRCA1/2 pts, n = 57; 59.6% Rucaparib 1° endpoints: ORR, PSA response FDA Approved May 20208,b Recruiting
- 4. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape” at PeerView.com/JGU40 Talazoparib + enzalutamide vs 1° endpoints: dose, rPFS Placebo + enzalutamide Selected Ongoing Trials of PARP Inhibitors1 (Cont’d) PROpel (NCT03732820); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • ECOG PS 0 or 1 • Assessment of HRR gene aberrations • Planned N = 720 vs 1° endpoint: rPFS Placebo + abiraterone Olaparib + abiraterone vs 1° endpoint: rPFS Placebo + abiraterone Niraparib + abiraterone MAGNITUDE (NCT03748641); Phase 3 • mCRPC; ongoing ADT or prior bilateral orchiectomy • Planned N = 1,000 TRITON3 (NCT02975934); Phase 3 • mCRPC previously treated with 1 next-generation AR-targeted therapy • Deleterious mutation in BRCA1/2 or ATM • Planned N = 400 vs 1° endpoint: rPFS Abiraterone or enzalutamide or docetaxel vs Avelumab + bempegaldesleukin + talazoparib or enzalutamide Rucaparib Avelumab + bempegaldesleukin Recruiting Recruiting Recruiting Recruiting Recruiting Rucaparib 1° endpoint: ORR 1° endpoints: confirmed OR, PSA response, DLTs LODESTAR (NCT04171700); Phase 2, open-label • Unresectable, locally advanced, or metastatic solid tumor and relapsed/progressive disease • At least 1 prior line of therapy extending OS or SOC therapy for advanced disease • mCRPC with BRCA1/2 mutations • ECOG PS 0 or 1 • Planned N = 220 (with solid tumors) Avelumab + bempegaldesleukin (NKTR-214) + talazoparib or enzalutamide (NCT04052204); Phase 2, open-label • mCRPC • ECOG PS 0 or 1 • Planned N = 160 • Cohort A: deleterious mutations in BRACA1/2 • Cohort B: non-BRACA1/2 mutations Recruiting Talazoparib 1° endpoint: ORR TALAPRO-1 (NCT03148795); Phase 2, open-label • mCRPC; metastatic disease in bone • Assessment of DDR mutation status • ECOG PS 0 to 2 • Planned N = 100 TALAPRO-2 (NCT03395197); Phase 3 • mCRPC; metastatic disease in bone • Assessment of DDR mutation status • ECOG PS 0 or 1 • Planned N = 1,037 RESULTS Confirmed ORR:9,c 28.0% Composite response:9 51.2%
- 5. Novel Approaches in Advanced Prostate Cancer PRACTICE AID Access the activity, “How I Think, How I Treat: Learning to Navigate the Modern Prostate Cancer Landscape” at PeerView.com/JGU40 a In May 2020, the FDA approved olaparib for adult patients with deleterious/suspected deleterious germline/somatic HRR gene-mutated mCRPC following progression on enzalutamide of abiraterone. b In May 2020, the FDA approved rucaparib for patients with deleterious germline/ somatic BRCA mutation-associated mCRPC previously treated with AR-directed therapy and a taxane-based chemotherapy. c In patients who received talazoparib for ≥16 weeks. ADT: androgen deprivation therapy; AR: androgen receptor; DDR: DNA damage repair; DLT: dose-limiting toxicity; ECOG PS: Eastern Cooperative Oncology Group Performance Status; HRD: homologous recombination deficiency; HRR: homologous recombination repair; mCRPC: metastatic castration-resistant prostate cancer; OR: overall response; ORR: objective response rate; PARP: poly (ADP-ribose) polymerase; PD-1: programmed cell death protein 1; PD-L1: programmed death ligand 1; PSA: prostate-specific antigen; rPFS: radiographic progression-free survival ; RR: response rate. 1. https://clinicaltrials.gov. 2. Mateo J et al. 2019 American Society of Clinical Oncology Annual Meeting (ASCO 2019). Abstract 5005. 3. Clarke N et al. Lancet Oncol. 2018;19:975-986. 4. de Bono J et al. N Engl J Med. 2020;382:2091-2102. 5. Abida W et al. ESMO 2019. Abstract 846PD. 6. Smith MR et al. American Society of Clinical Oncology 2019 Genitourinary Cancers Symposium (ASCO GU 2019). Abstract 202. 7. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-olaparib-hrr-gene-mutated-metastatic-castration-resistant-prostate-cancer. 8. https://www.fda.gov/drugs/fda-grants-accelerated-approval-rucaparib-brca-mutated-metastatic-castration-resistant-prostate. 9. De Bono JS et al.2020 ASCO Virtual Scientific Program (ASCO 2020). Abstract 5566. 10. Karzai F et al. J Immunother Cancer. 2018;6:141. 11. Yu EY et al. J Clin Oncol. 2020;38 (suppl; abstr 5544). 12. Sridhar SS et al. J Clin Oncol. 2020;38 (suppl; abstr 5550). 13. Conter HJ et al. J Clin Oncol. 2020;38 (suppl; abstr 5545). Selected Ongoing Trials of PARP Inhibitors Combined With PD-1/PD-L1 Inhibitors1,7-10 JAVELIN PARP MEDLEY (NCT03330405); Phase 2 • Locally advanced or mCRPC • Primary or metastatic tumor biopsy • ECOG PS 0 or 1 • Planned N = 242 CheckMate -9KD (NCT03338790); Phase 2 • mCRPC; ongoing ADT • Plasma and fresh or archival tumor tissue • ECOG PS 0-1 Recruiting 1° endpoints: DLTs, OR 1° endpoints: safety, ORR, composite RR • HRD status (must be available before tx arm assignment) • Planned N = 330 1° endpoints: ORR, PSA response Avelumab + talazoparib Nivolumab + rucaparib or docetaxel or enzalutamide KEYNOTE-365 (NCT02861573); Phase 1b/211-13 • mCRPC; ongoing ADT • Tissue biopsy from site not previously irradiated • Planned N = 400 QUEST (NCT03431350); Phase 1/2 • mCRPC • DDR gene anomalies • Prior novel AR-targeted therapy • Planned N = 80 NCT02484404; Phase 2 (prostate cohort) • mCRPC; ongoing ADT or prior bilateral orchiectomy • ECOG PS 0 or 1; previously treated with enzalutamide and/or abiraterone • Planned N = 384 RecruitingRecruiting Recruiting Niraparib + cetrelimab RESULTS PSA response Pembro + olaparib, n = 84; 9% Pembro + docetaxel, n = 104; 28% Pembro + enzalutamide, n = 102; 22% 1° endpoint: PSA response Pembrolizumab + olaparib or docetaxel/prednisone or enzalutamide or abiraterone 1° endpoints: dose, safety Durvalumab + olaparib RESULTS rPFS; evaluable N = 17 Durvalumab + olaparib: 16.1 mo 12-month rPFS: 51.5% 9/17 (53%) patients had a radiographic and/or PSA response
