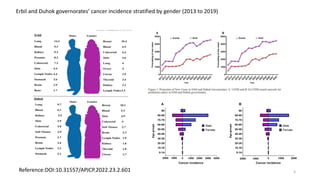
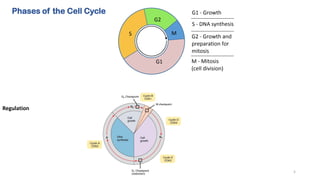
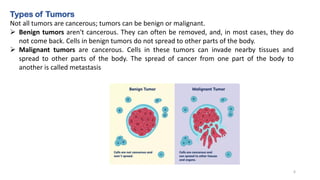
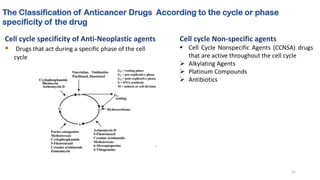
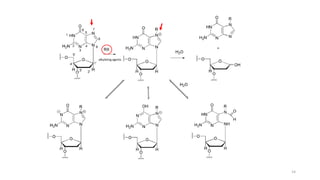
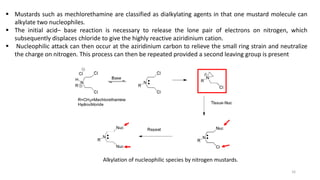
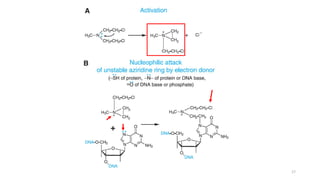
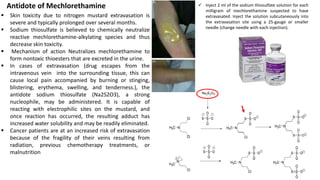
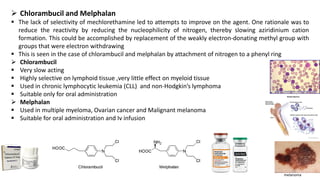
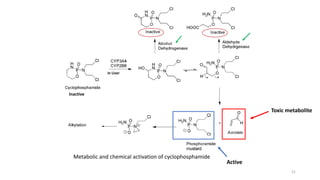
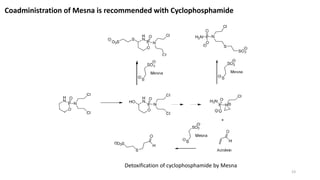
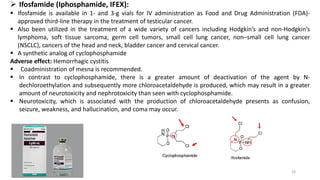
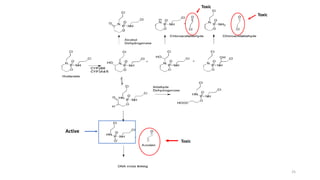
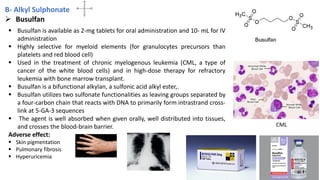
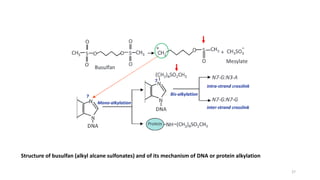
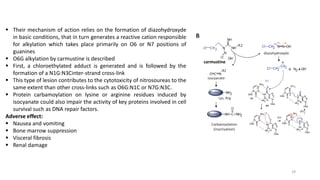
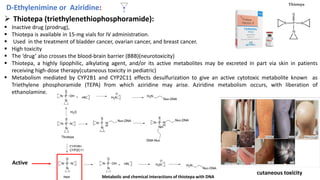
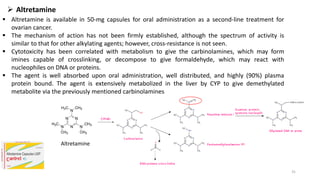
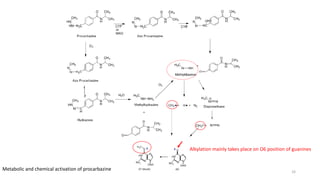
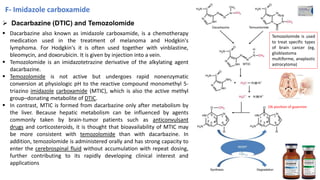
This document provides an overview of anti-cancer drugs, beginning with an introduction to cancer and statistics on cancer cases in Iraq. It then discusses the classification of anti-cancer drugs according to their chemical structure and mechanisms of action. Specifically, it covers alkylating agents including nitrogen mustards, alkyl sulphonates, nitrosoureas, and aziridines. It provides details on the structures, mechanisms of action, uses, and side effects of common alkylating agents like cyclophosphamide, ifosfamide, busulfan, carmustine, and thiotepa. The document focuses on describing the chemical properties and metabolic pathways that allow these drugs to damage cancer cell DNA and induce cell death.