Pyrimidine_class-S.ppt
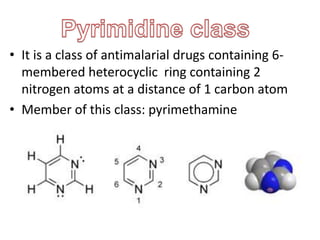
- 1. • It is a class of antimalarial drugs containing 6- membered heterocyclic ring containing 2 nitrogen atoms at a distance of 1 carbon atom • Member of this class: pyrimethamine
- 2. 2, 4 diaminopyrimidine • Which was an antifolate • The compound was studied extensively and maximum activity was noticed when a chlorophenyl group was attached at position 5 and an electron donating group (C2H5) at position 6 • Cl group should be at para position • By such modifications-pyrimethamine-has maximum activity
- 3. Structure Activity Relationship • 2 primary amino groups at position 2 and 4 when converted to 2o or 3o activity is reduced • An aromatic ring is directly attached with pyrimidine ring for maximum activity, if carbon or N is inserted between them activity is decreased C N
- 4. • If aromatic ring is replaced by hetrocyclic compound activity is decreased • Aromatic ring must contain an electron withdrawing group at p-position • Presence of e donating group at position 6 is also necessary for activity
- 5. • When 2 rings are separated by a methylene, antiplasmodial activity is decreased but antibacterial activity is increased- trimethoprim
- 6. • Pyrimethamine (25 mg) is used in combination with sulfadoxine (500 mg) and the name is Fansidar, which is used in treatment and prophylaxis of malaria. It radically cures P. falciparum but clinically cures other species • Sulfonamide competes with PABA for synthesis of dihydrofolate while pyrimethamine inhibits dihydrofolate reductase–hence sequential inhibition of purine and pyrimidine
- 7. Synthesis of pyrimethamine Cl CH2 CN CH3-CH2-CO-OC2H5 C2H5ONa Cl NC HC CO C2H5 Cl NC HC COH C2H5 CH-CH2-CH2-OH CH-CH2-CH2-O Para chlorophenyl acetonitrile Ethyl propionate propionyl p-chlorophenyl acetonitrile 3-methyl butanol Hemiketal
- 8. Cl NC C C2H5 CH-CH2-CH2-O NH2C=NH NH2 Cl NC C C2H5 NH 2C=NH NH Dehydration Rearrangement Pyrimethamine β-ethyl, β-isoamyl oxy, p-chlorophenyl acetonitrile
- 9. Pyrimethamine mechanism • Interferes with utilization of folic acid inside plasmodium by inhibiting its dihydrofolate reductase • Hence, there is no synthesis of folic acid, which in needed for the synthesis of purine and pyrimidine that are required for nucleic acid. As a result, replication of plasmodium is inhibited