Mahra Nourbakhsh's presentation, hepatitis C virus #2
- 2. HCV circulation in blood
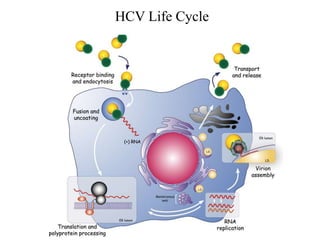
- 3. HCV Virion Produced in infected patient • Heterogeneous density (1.006-1.25 g/ml). • Infectivity is inversely correlated to the density of particles. (Andre P et al., 2002) • The majority of viral RNA is associated with density<1.06g/ml. (Andre P et al., 2002) • The particles are irregular in shape and contain core, E1,E2 (HCV) and ApoB, ApoE and ApoC1 (VLDL derivatives). Chang, K.S. et al., 2007; Nielsen, S.U. et al. 2006; Meunier, J.C. et al., 2008 HCV+Lipoprotein LVPs
- 4. HCV Virion Produced in cell culture • Higher density compare to HCV derived from patients (1.1 vs. 1.06 g/ml). • Transfer of HCVcc to chimpanzee ---> viral particles with a lower density, similar to that of patient-derived LVP---> again back to Huh7.5 cells --->Viral particles with a higher density similar to HCVcc. (Lindenbach, B.D. et al. (2006) • The particles contain core, E1,E2 (HCV) and ApoE and ApoC1 but not ApoB. (Chang, K.S. et al., 2007; Merz, A. et al., 2010; Meunier, J.C. et al., 2008)
- 5. LVP and HCVcc From: Bartenschlager et al., 2011
- 6. HCV Assembly: Core Protein J. McLauchlan / Biochimica et Biophysica Acta 1791 (2009) 552–559 Core can be divided to two domains D1, D2 and signal peptide. Core is released from HCV polyprotein by SP and SPP and translocates to cytosolic lipid droplet (cLD) D1 is hydrophilic positively charge (attachment to negative PO4 backbone of HCV RNA) and faced to cytosol. D2 is hydrophobic and anchors D1 to a lipid membrane (cLD) .
- 7. HCV Assembly: Replication Complex, Core & cLD From: Bartenschlager et al., 2011
- 8. Co-translational lipidation of ApoB100 the main protein component of VLDL by MTP and formation of poorly lipidated pre- VLDL. Formation of luminal lipid droplets from cytosolic lipid droplets by possibly MTP. They may contain ApoE. Fusion of luminal lipid droplets with pre-VLDL and formation of mature fully lipidated VLDL. Taken from Ohsaki Y et al.,2009 VLDLAssembly Pathway
- 9. From: Bartenschlager et al., 2011 VLDL Assembly and HCV Release Inhibition of MTP, reduces the HCV egress. (Huang et al. 2007) SiRNA down regulation of ApoB reduces the secretion of HCV. (Huang et al. 2007) Cells were treated with brefeldin A, produced high-density HCV particles which were degraded. (Gastaminza et al. 2008) Dwon-reglation of ApoE reduces the production of infectious HCV particles (Cheng et al. 2007)
- 10. Hepatitis C virus cell entry: cellular receptors and role of lipoproteins.
- 11. Tetraspanin CD81 • 4 TMD, intracellular N- and C-terminal, small and large extracellular loops (SEL, LEL). • An HCV E2 binding region maps to the LEL of CD81, species – specific. (Pileriet al.,1998, Flint et al., 2006) • Inhibition of HCV infection by mAbs directed against CD81 , a soluble form of E2 (sE2) and down regulation of CD81 expression using siRNA. (Bartosch et al., 2003; Wakita et al.2005; Meuleman et al. 2008; Zhang et al., 2004) • Activation of GTP-ases and the actin-dependent relocalization of the E2/CD81 complex to cell–cell contact areas with tight junction proteins occludin and CLDN-1 (Brazzoli et al.,2008) SEL LEL
- 12. Scavenger Receptor Class B Type I (SR-BI) • 2 TMD, single extracellular loops. • A receptor for various classes of Lipoproteins; HDL, LDL, VLDL, LDLox. Important role in reverse cholesterol transport by CE uptake from HDL. • HCV E2 binds to SR-BI, species –specific. In this case SR-BI co- operatively interacts with CD81 in HCV cell entry. (Scarselli et al., 2002; Zeisel et al., 2007) • Inhibition of HCV infection by mAbs directed against SR-BI . The expression levels of SR-BI can modulate HCVcc infectivity. (Grove et al., 2007; Catanese et al., 2007) • Enhancement of SR-BI dependent HCV entry by HDL, and inhibition by VLDL, LDLox.. (Bartosch et al.,2005; Maillard et al.,2006; Von Hahn et al.,2006; Lavie et.al, 2006)
- 13. Tight Junction Proteins: Claudin 1 (CLDN-1) • 4 TMD, intracellular loop N- and C-terminal, 2 extracellular loops (EL1 & EL2). • EL1 seems to be involve in HCV entry but still no evidence for a direct interaction between CLDN1 and the HCV virus. (Evans et al., 2007) • Inhibition of HCV entry to permissive cells (Huh7.5) by silencing CLDN-1. (Evans et al., 2007) • Enhancing susceptibility of HCV infection in non-permissive cells (293T) by CLDN-1 expression. (Evans et al., 2007) EL1
- 14. Tight Junction Proteins: Occludin • 4 TMD, intracellular loop N- and C-terminal, 2 extracellular loops (EL1 & EL2). • Critical role for HCV entry. Interacts directly with HCV E2 (colocalization). (Liu et al., 2009; Benedicto et al., 2008). • Inhibition of HCV entry to permissive cells by SiRNA silencing of Occludin. (Liu et al., 2009) • Human occludin renders murine cells infectable with HCVpp. (Ploss et al., 2009) Possibility for introducing immuno-competent murine HCV model EL1 EL2
- 15. LDL Rceptor (LDL-R) • Cell surface receptor recognizes apoB100 of LDL and apoE of LDL/Chylomicron remnant. It has been proposed to be involved in HCV entry. (Agnello et al., 1999; Monazahian et al., 1999) • Pros: 1. Association of HCV with lipid particles. 2. Inhibition of HCV (derived from plasma) entry (Cellular viral RNA) by mAb against ApoB. (Molina et al.,2007) 3. Correlation between accumulation of cellular HCV RNA and LDL-R mRNA level. (Molina et al.,2007) • Cons: 1. The role of LDL-R in HCVcc has not yet been demonstrated 2. Lipoprotein lipase inhibits HCV cell entry while it should enhance it. (Andreo et al., 2007)
- 16. Other Receptors: Glycosaminoglycans (GAGs) • Primary, low affinity receptor for HCV entry. Binds to sE2 but not E1- E2 heterodimer (?!).GAGs also is able to attach to lipoproteins. (Callens et al., 2005) • Heparan Sulphate (GAGs) inhibition by heparinase (degradation) or heparin (competition) reduces HCV entry. (Barth et al., 2006) Other Receptors: DC-SIGN, L-SIGN • DC-SIGN is expressed in Kuppfer cells, DC and lymphocytes.L-SIGN in sinusoidal cells. • Capture receptors, tissue tropism acts. (Cormier et al., 2004)
- 17. Summary • HCV particles circulates in patient’s sera in association with lipoproteins in particular VLDL (LVPs) and contains ApoB, E and C1. • Several receptors for HCV entry are also involved in lipoprotein reuptake/metabolism; SR-BI, LDL- R, GAGs. • Lipid droplets act as a scaffold for primary HCV assembly. • VLDL assembly and secretion pathway is essential for HCV secretion.