Rituximab Treats Refractory CD20 Positive Cellular Rejection in Living Kidney Transplant
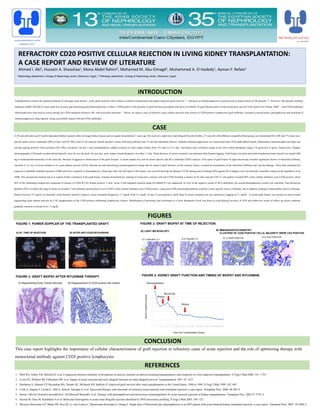
- 1. REFRACTORY CD20 POSITIVE CELLULAR REJECTION IN LIVING KIDNEY TRANSPLANTATION: A CASE REPORT AND REVIEW OF LITERATURE Ahmed I. Akl1, Hussein A. Sheashaa1, Mona Abdel Rahim2, Mohamed M. Abu-Elmagd1, Muhammed A. El Hadedy1, Ayman F. Refaie1 1 Nephrology department, Urology & Nephrology center, Mansoura, Egypt, 2 Pathology department, Urology & Nephrology center, Mansoura, Egypt. CASE INTRODUCTION FIGURES CONCLUSION Transplantation remains the optimal treatment of end-stage renal disease1. Acute graft rejection (AR) remains a common complication and impact long-term graft survival 2,3. Advances in immunosuppressive protocols led to improvement in AR episodes 3–6. However, AR specially antibody mediated (AMR) still able to cause graft loss in some and shortening graft functioning time in others. CD20 positive cells presence in graft has been associated with poor reversibility of graft function and a trend toward poor survival of the graft even without AMR 7. Anti-CD20 antibodies (Rituximab) have been used as rescue therapy for CD20 mediated refractory AR, with successful outcomes 7. Herein, we report a case of refractory acute cellular rejection with clusters of CD20 positive lymphocytes graft infiltrates, resistant to steroid pulses, plasmapheresis and switching of immunosuppressive drug regimen, being successfully treated with anti-CD20 antibodies. A 39-year-old-male type II insulin dependent diabetic patient suffer end stage kidney disease and on regular hemodialysis 3 years ago. He received a right iliac renal allograft from his brother, 27-year-old with different compatible blood group, one mismatched HLA DR and 7% donor non- specific panel reactive antibodies (PRA) Class I and 0% PRA Class II. He enjoyed smooth operative course with total ischemia time 55 min and immediate diuresis. Induction Immunosuppression was monoclonal anti-CD25 alpha (Basiliximab). Maintenance immunosuppression plan was steroids sparing protocol with tacrolimus (FK 506), everolimus. On day 2 post transplantation, sudden cessation of urine output volume from 10 L/day to 3.6 L/day. Tacrolimus and everolimus trough levels were within therapeutic range (7.8 ng/ml & 6.4 ng/ml; respectively). Doppler ultrasonography of the graft revealed absent diastolic wave all over the graft. On next day, urine output volume dropped to less than 1 L/day. Sloop decrease of serum creatinine was stationary then became lagging. Graft biopsy was done and methyl-prednisone pulse steroid was started (500 mg of methylprednisolone/day) in the same day. Because of aggressive deterioration of the graft function. A serum sample was sent for donor specific anti-HLA antibodies (DSA) analysis. First report of graft biopsy by light microscopy revealed significant clusters of interstitial infiltrate, vasculitis (v-1), foci of severe tubulitis (t-3), acute tubular necrosis (ATN). Steroids use and intensifying immunosuppressive drugs did not improve graft function; on the contrary, biopsy revealed an accentuation of the interstitial infiltrates and vascular damage. These facts prompted the suspicion of antibody mediated rejection (AMR) and led to initiation of plasmapheresis. Some days later, the full report of the biopsy was received showing the absence of C4d staining and circulating DSA against HLA antigens were not detected, reasonably ruling out the hypothesis of an AMR. This unexpected situation led us to request further evaluation of the graft biopsy. Immuno-histochemistry staining of consecutive sections with anti-CD20 (formally a marker for B cells) and anti-CD3 (T cell marker) revealed 90% of the cellular infiltrates were CD20 positive, about 80% of the infiltrating lymphocytes composed of clusters of CD20 & CD3 double positive T cells. Acute T-cell-mediated rejection grade IIA (Banff 07) was diagnosed. In view of the negative results of HLA antibodies, the second plasmapheresis session was cancelled. Anti-thymocyte globulin (ATG) would be the drug of choice in resistant T cell mediated rejection but in view of 90% of the cellular infiltrates were CD20 positive, using anti-CD20 monoclonal antibody would be a more specific choice of therapy, and in addition, keeping in hand another choice of therapy. Patient received 375 mg/m2 of rituximab. Graft function started to improve serum creatinine dropped to 2.7 mg/dL from 4.1 mg/dl. At day 12 post-operative, daily drop of serum creatinine was not satisfactory, lagging at 2.1 mg/dl. A second graft biopsy was carried out and revealed regenerating acute tubular necrosis by L/M, disappearance of the CD20 positive infiltrating lymphocytes clusters. Modification of tacrolimus and everolimus to a lower therapeutic levels was done to avoid delayed recovery of ATN and within two weeks of follow up serum creatinine gradually returned to normal level 1.2 mg/dL. FIGURE 1: POWER DOPPLER OF THE TRANSPLANTED GRAFT. A) AT TIME OF REJECTION B) AFTER ANTI-CD20 (RITUXIMAB) A.1) Vasculitis, V-1 A.2) Tubulitis, t-3 FIGURE 2: GRAFT BIOPSY AT TIME OF REJECTION. A) LIGHT MICROSCOPY. B.1) CD20 POSITIVE B) IMMUNOHISTOCHEMISTRY: CLUSTERS OF CD20 POSITIVE CELLS, MAJORITY WERE CD3 POSITIVE. B.2) CD3 POSITIVE FIGURE 3: GRAFT BIOPSY AFTER RITUXIMAB THERAPY. B) Disappearance of CD20 positive cell clusters.A) Regenerating Acute Tubular Necrosis. 0 1 2 3 4 5 6 7 8 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 SerumCreatinine(mg/dl) Time Post Transplantation (Days) Rituximab Biopsy Biopsy Transplantation FIGURE 4: KIDNEY GRAFT FUNCTION AND TIMING OF BIOPSY AND RITUXIMAB. This case report highlights the importance of cellular characterization of graft rejection in refractory cases of acute rejection and the role of optimizing therapy with monoclonal antibody against CD20 positive lymphocytes. UROLOGY&NEPHROLOGY CENTER MANSOURA, EGYPT REFERENCES 1. Wolf RA, Ashby VB, Milford EL et al. Comparison between mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplantation. N Engl J Med 2000: 341: 1725 2. Cosio FG, Pelletier RP, Falkenhain ME et al. Impact of acute rejection and early allograft function on renal allograft survival. Transplantation 1997: 63: 1611 3. Hariharan S, Johnson CP, Bresnahan BA, Taranto SE, McIntosh MJ, Stablein D. Improved graft survival after renal transplantation in the United States, 1988 to 1996. N Engl J Med 1999: 342: 605 4. Celik A, Saglam F, Cavdar C, Sifil A, Atila K, Sarioglu S, et al. Successful therapy with rituximab of refractory acute humoral renal transplant rejection: a case report. Transplant Proc. 2008; 40:302–4. 5. Iberno ´nM,Gil-VernetS,CarreraM,Sero ´nD,MoresoF,BestardO, et al. Therapy with plasmapheresis and intravenous immunoglobulin for acute humoral rejection in kidney transplantation. Transplant Proc. 2005;37:3743–5. 6. Sarwal M, Chua M, Kambham N et al. Molecular heterogenity in acute renal allograft rejection identified by DNA microarray profiling. N Engl J Med 2003: 349: 125. 7. Moscoso-Solorzano GT, Baltar JM, Seco M, Lo ´pez-Larrea C, Mastroianni-Kirsztajn G, Ortega F. Single dose of Rituximab plus plasmapheresis in an HIV patient with acute humoral kidney transplant rejection: a case report. Transplant Proc. 2007; 39:3460–2.
