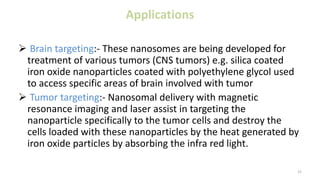
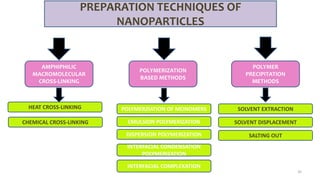
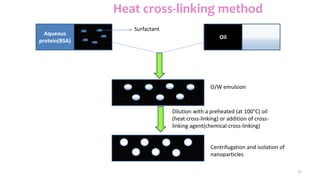
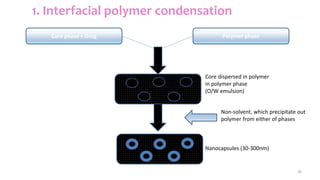
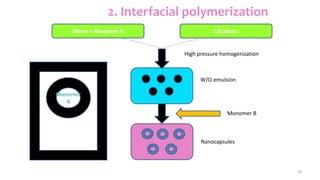
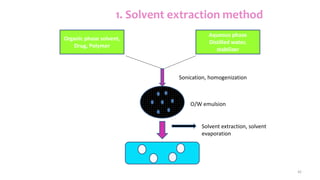
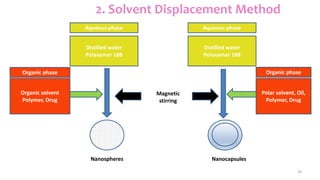
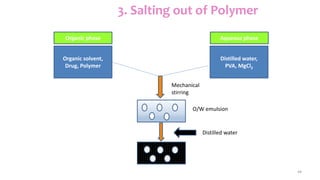
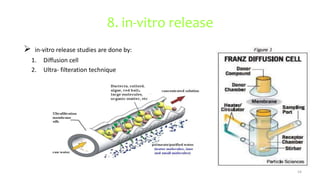
This document discusses various types of nanoparticles including their properties, preparation, and applications. It describes carbon-based nanoparticles like carbon nanotubes, solid-lipid nanoparticles, silicon-based nanoparticles, liposomes, nanosomes, and niosomes. Nanoparticles have sizes between 1-100 nanometers and unique optical, magnetic, mechanical, and thermal properties dependent on their size and structure. They are useful for drug and gene delivery, cancer therapy, and other medical applications due to properties like cell specificity and reduced toxicity.