Chemical_Equilibrium_Intro.ppt
- 2. Reversible Reactions • So far, we’ve treated chemical reactions as a sure thing, progressing from beginning to end. However, it’s not always that simple… • If conditions are correct, a reaction can be made to go in reverse. In theory, this is true for all reactions. Thus, all reactions are reversible. Often, reactions proceed in one direction, with a small amount of the reverse reaction occurring at the same time. Reversible reactions can be written as follows: • A double sided arrow indicates that the reaction goes in both directions
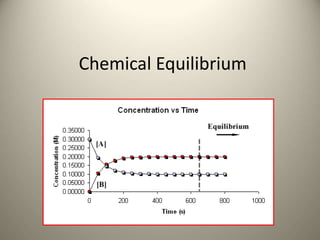
- 3. How do reactions reach Equilibrium? • Equilibrium is reached when the amount of reactants going forward equals the amount of product going backward, in other words, when there is no net change in the concentration of reactants and products. • Note that this does not mean that the reactions stop, only that the forward rate balances the reverse rate. Rateforward reaction = Ratereverse reaction
- 4. The Law of Equilibrium • When a chemical reaction is at equilibrium, there is no NET change in the concentration of all components in the reaction. This can be expressed mathematically. Consider the following reaction: • (The lower case letters are the coefficients from the balanced equations)
- 5. Equilibrium Expressions • This allows us to look at a reaction at equilibrium as an expression of a constant (the equilibrium constant, keq). • keq = products reactants • The brackets denote concentrations in Mol/L • Or keq = C c D d A a B b We only included gases in the equilibrium expression because the concentrations of solids and liquids are constant at a constant temperature
- 6. Write an Equilibrium Expression • Homogeneous: All participants in the reaction are in the same state: • Heterogeneous: Some participants are in different states.
- 7. Calculating keq What is the value for the equilibrium constant (keq) if the equilibrium concentrations are: CH4 = 0.354 mol/L CO = 0.05 mol/L C2H4O = 0.05 mol/L (.354) (.05) = .354 .05
- 8. Keq and what it means • Note that large values for keq indicate that the product is favored in this reaction (Numerator) • If the value of keq < 1, then the reactants are favored (the reaction goes toward reactants).
- 9. Practice • Calculate keq for the following reaction and determine if the products or reactants are favored and why. • [NO] = .89 M • [Br2] = .45 M • [NOBr] = .56 M • (.56)2 = .88 (.89)2 (.45) Since keq is less than 1, the reactants are favored in this reaction (slightly)