Alzheimer's Research Review Paper
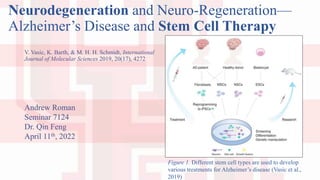
- 1. Neurodegeneration and Neuro-Regeneration— Alzheimer’s Disease and Stem Cell Therapy V. Vasic, K. Barth, & M. H. H. Schmidt, International Journal of Molecular Sciences 2019, 20(17), 4272 Andrew Roman Seminar 7124 Dr. Qin Feng April 11th, 2022 Figure 1. Different stem cell types are used to develop various treatments for Alzheimer’s disease (Vasic et al., 2019)
- 2. Key Points • Neurodegeneration leads to dementia • Multiple diseases cause dementia • Brain self-repairs • Therapeutic stimulation of adult neurogenesis • Stem cell therapy (Vasic et al., 2019)
- 3. Alzheimer’s disease • Short-term memory loss • Reduction in brain volume • Daily life impaired Figure 2. Comparison of normal brain with Alzheimer’s disease patient (Mattson, 2004). (Vasic et al., 2019)
- 4. Alzheimer’s Disease Continued • Death of neurons • Loss of synapses • Amyloid-β accumulation • Neurofibrillary tangles of tau protein (Vasic et al., 2019)
- 5. Cell Types Involved with Alzheimer’s Disease • Microglia • Astrocytes • Oligodendrocytes • NG2-Glia • Neurons (Vasic et al., 2019)
- 6. Microglia • Resident macrophages • Function impaired in AD (Katsumoto et al., 2018) • Activation of inflammatory pathways (Wyss-Coray et al., 2011) • Circulating blood and mesenchyme • Continuously survey the microenvironment • Remove detritus • Synaptic remodeling and plasticity (Vasic et al., 2019)
- 7. Figure 3. Study showing activities of resting microglia (Nimmerjahn et al., 2005).
- 8. • Impaired Aβ homeostasis alters morphology • Production of inflammatory cytokines • TREM2 activation and microgliosis through APOE4 • ABCA7 regulation of lipid homeostasis and phagocytosis • P2X7 receptor overactivation mediated by extracellular ATP Microglia Continued Figure 4. Different forms of Aβ and how they affect microglia (Heppner et al., 2015). (Vasic et al., 2019)
- 9. Figure 5. Colocalization in the Phagocytic Cup (Jehle et al., 2006)
- 10. Astrocytes in Alzheimer’s Disease • Multiple interactions with neurons • Synaptic plasticity • Chemotactic response to Aβ • Bind, take up, and degrade Aβ • Secrete enzymes for extracellular degradation of Aβ Figure 6. Astrocytes remove neurotransmitters and other substances from synaptic terminals (Fields et al., 2013) (Vasic et al., 2019)
- 11. Astrocytes in Alzheimer’s Disease Continued • Aggregation of Aβ alters receptor expression • Increased Ca2+ uptake • Decreased glutamate uptake • Inflammatory cytokines trigger Aβ production (Blasko et al., 2000) Figure 7. Distribution of Aβ in neurons and astrocytes (Nagele et al., 2003) (Vasic et al., 2019)
- 12. Oligodendrocytes in Alzheimer’s Disease • Myelin deteriorates • Axons are destroyed • No reduction in myelin • Aβ is cytotoxic (Vasic et al., 2019)
- 13. NG2-glia • Newly discovered cell type • Differentiation is inhibited by Aβ Figure 8. Developmental stages of NG2-glia (Nishiyama et al., 2009) (Vasic et al., 2019)
- 14. Neurons • Hyperphosphorylated tau protein forming intracellular neurofibrillary tangles • Phosphorylation normally allows interaction with microtubules transporting mitochondria • Energy dysfunction, reactive oxygen and nitrogen species (Vasic et al., 2019)
- 15. Organelles and Related Processes • Mitochondria • Autophagy • Endocytic processes (Vasic et al., 2019)
- 16. Mitochondria • Fission and fusion • Distribution of mitochondria • Neuroprotective effects Figure 9. Loss of functional mitochondria promotes oxidative stress (Moreira et al., 2010) (Vasic et al., 2019)
- 17. Autophagy • Pathway for degradation of cellular components • Mitophagy • Essential housekeeping process • Autophagy gene mutation and AD • Autophagy is neuroprotective and prevents protein accumulation (Menzies et al., 2015) (Vasic et al., 2019)
- 18. Endocytic Processes • Clathrin-mediated endocytosis • Clathrin-independent endocytic pathways • Flotillin-1 and -2 are associated with membrane lipid-rafts • Caveolae-1, -2, -3 in ordered lipid raft domains • APP and Aβ toxicity • Endosomes increased in AD • Cav-1 downregulation (Vasic et al., 2019)
- 19. Stem Cell Therapy • Endogenous regeneration • Engrafted regeneration • ESCs • NSCs • iPSCs • MSCs • Translation (Vasic et al., 2019)
- 20. Endogenous Regeneration • Neurogenesis in adult hippocampus directs spatial navigation (Ekstrom et al., 2003) • AD affects hippocampal neurons • 700 new neurons per day • Induced neurogenesis in animals • Chemicals and growth factors ex. erythropoietin, BDNF • Exercise and CA1 region (Vasic et al., 2019)
- 21. Figure 10. Exercise decreases loss of neurons in transgenic mouse model for Alzheimer’s disease (Hüttenrauch et al., 2016)
- 22. Engrafted Regeneration • Sources of stem cells for treatment vary • Embryonic stem cells • Neural stem cells • Induced pluripotent stem cells • Mesenchymal stem cells • Treatment strategies vary Figure 1. Different stem cell types are used to develop various treatments for Alzheimer’s disease (Vasic et al., 2019) (Vasic et al., 2019)
- 23. Embryonic Stem Cells • Pluripotent • Unlimited self-renewal ability • Derived from blastocyst • Regulation of donated embryos • Pre-differentiated into basal forebrain cholinergic neurons & medial ganglionic eminence-like progenitor cells (Vasic et al., 2019)
- 24. Figure 11. Characterization of neurons derived from stem cells (Moghadam et al., 2009)
- 25. Neural Stem Cells • Multipotent, self-renewing producing neurons, oligodendrocytes, or astrocytes • Improving survival outcome • Grafting improved mouse models • Genes or drugs added • Glia and TLR4 activation associated with neuroinflammation (Vasic et al., 2019)
- 26. Induced Pluripotent Stem Cells • Production of autologous cells for transplantation • Immune rejection • Patient derived skin fibroblasts showed ApoE4 pathogenic • Transplanted glial cells improved cognitive function • Neuronal precursor cells developed into cholinergic neurons • Parkinson’s and stroke models (Vasic et al., 2019)
- 27. Figure 12. Characterization of grafted hiPS neurons in mouse hippocampus (Fujiwara et al., 2013)
- 28. Mesenchymal Stem Cells • Present in bone marrow, umbilical cord, and adipose tissue • Low rate of neuronal differentiation • Intravenous application crosses blood brain barrier • Results like other SC therapies • Improved neuroprotection with downregulation of inflammation • Genetic alterations for supplemental extracellular vesicles • Overexpression of cytokines (Vasic et al., 2019)
- 29. Translational Research • Ongoing clinical trials • Transplanted cells stimulate endogenous mechanisms • MSCs ease of use • Phase I trials of hUBC-MSCs showed no improvement • Transduced autologous fibroblasts improved cognitive decline • Results posted on ClinicalTrials.gov (Vasic et al., 2019)
- 30. Conclusion • Understanding of AD limited • New data for treatments • Translation from rodent model problematic • Multiple factors affect clinical trial • Evaluation of transplanted cells • Diagnostic tools for early detection • Combination of methods for treatments Figure 13. Table summarizing types of stem cells (Vasic et al., 2019) (Vasic et al., 2019)
- 31. Works Cited Blasko, I., Veerhuis, R., Stampfer-Kountchev, M., Saurwein-Teissl, M., Eikelenboom, P., & Grubeck-Loebenstein, B. (2000). Costimulatory effects of interferon-γ and interleukin-1β or tumor necrosis factor α on the synthesis of Aβ1-40 and Aβ1-42 by human astrocytes. Neurobiology of Disease, 7, 682-689. https://doi.org/10.1006/nbdi.2000.0321 Ekstrom, A. D., Kahana, M. J., Caplan, J. B., Fields, T. A., Isham, E. A., Newman, E. L., & Fried, I. (2003). Cellular networks underlying human spatial navigation. Nature, 425, 184-188. https://doi.org/10.1038/nature01964 Fields, R. D., Araque, A., Johansen-Berg, H., Lim, S.-S., Lynch, G., Nave, K.-A., Nedergaard, M., Perez, R., Sejnowski, T., & Wake, H. (2013). Glial biology in learning and cognition. The Neuroscientist, 20, 426-431. https://doi.org/10.1177/1073858413504465 Fukiwara, N., Shimizu, J., Takai, K., Arimitsu, N., Saito, A., Kono, T., Umehara, T., Ueda, Y., Wakisaka, S., Suzuki, T., & Suzuki, N. (2013) Restoration of spatial memory dysfunction of human APP transgenic mice by transplantation of neuronal precursors derived from human iPS cell, Neuroscience Letters, 557, 129-134. https://doi.org/10.1016/j.neulet.2013.10.043 Heppner, F. L., Ransohoff, R. M., & Becher, B. (2015). Immune attack: the role of inflammation in Alzheimer’s disease. Nature Reviews Neuroscience, 16, 358-372. https://doi.org/10.1038/nrn3880 Hüttenrauch, M., Brauß, A., Kurdakova, A., Borgers, H., Klinker, F., Liebetanz, D., Salinas-Riester, G., Wiltfang, J., Klafki, H. W., & Wirths, O. (2016). Physical activity delays hippocampal neurodegeneration and rescues memory deficits in an Alzheimer disease mouse model. Translational Psychiatry, 6, e800. https://doi.org/10.1038/tp.2016.65 Jehle, A. W., Gardai, S. J., Li, S., Linsel-Nitschke, P., Morimoto, K., Janssen, W. J., Vandivier, R. W., Wang, N., Greenberg, S., Dale, B. M., Qin, C., Henson, P. M., & Tall, A. R. (2006). ATP-binding cassette transporter A7 enhances phagocytosis of apoptotic cells and associated ERK signaling in macrophages. Journal of Cell Biology, 174, 547-556. https://doi.org/10.1083/jcb.200601030 Katsumoto, A., Takeuchi, H., Takahashi, K., & Tanaka, F. (2018). Microglia in Alzheimer’s disease: risk factors and inflammation. Front. Neurol., 9, 978. https://doi.org/10.3389/fneur.2018.00978 Mattson, M. P. (2004). Pathways toward and away from Alzheimer’s disease. Nature, 430, 631-639. https://doi.org/10.1038/nature02621 Menzies, F. M., Fleming, A., & Rubinsztein, D. C. (2015). Compromised autophagy and neurodegenerative diseases. Nature Reviews Neuroscience, 16, 345-347. https://doi.org/10.1038/nrn3961 Moghadam, F. H., Alaie, H., Karbalaie, K., Tanhaei, S., Esfahani, M. H. N., & Baharvand, H. (2009). Transplantation of primed or unprimed mouse embryonic stem cell-derived neural precursor cells improves cognitive function in Alzheimerian rats. Differentiation, 78, 59-68. https://doi.org/10.1016/j.diff.2009.06.005 Moreira, P. I., Carvalho, C., Zhu, X., Smith, G., & Perry, G. (2010). Mitochondrial dysfunction is a trigger of Alzheimer’s disease pathophysiology. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1802, 2-10. https://doi.org/10.1016/j.bbadis.2009.10.006 Nagele, R. G., D’Andrea, M. R., Lee, H., Venkataraman, V., & Wang, H.-Y. (2003). Astrocytes accumulate Aβ42 and give rise to astrocytic amyloid plaques in Alzheimer disease in brains. Brain Research, 971, 197-209. https://doi.org/10.1016/S0006- 8993(03)02361-8 Nimmerjahn, A., Kirchoff, F., & Helmchen, F. (2005). Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science, 308, 1314-1318. https://doi.org/10.1126/science.1110647 Nishiyama, A., Komitova, M., Suzuki, R., & Zhu, X. (2009). Polydendrocytes (NG2 cells): multifunctional cells with lineage plasticity. Nature Reviews Neuroscience, 10, 9-22. https://doi.org/10.1038/nrn2495 Vasic, V., Barth, K., & Schmidt, M. H. H. (2019). Neurodegeneration and neuro-regeneration—Alzheimer's disease and stem cell therapy. International Journal of Molecular Sciences, 20(17), 4272. https://doi.org/10.3390/ijms20174272 Wyss-Coray, T., & Rogers, J. (2012). Inflammation in Alzheimer disease—a brief review of the basic science and clinical literature. Cold Spring Harb. Perspect. Med., 2, a006346. doi: 10.1101/cshperspect.a006346
Editor's Notes
- time-lapse recording showing spontaneous engulfment and subsequent evacuation of tissue components by microglial processes microglial processes and protrusions contact neighboring astrocytes (left), neuronal cell bodies (center; unstained dark areas), and the astrocytic sheath around a microvessel (right). Images are overlays of the green microglia and red SR101 stain.
- triggering receptor expressed in myeloid cells 2, AD decrease of their phagocytic activity metallopeptidase 9
- LDL receptor-related protein-1 (LRP1),
- insulin degrading enzyme and metalloproteases
- astrocyte-derived plaques AP subpial portion of the molecular layer ML Activated astrocytes AA neuron-derived plaques NP underlying pyramidal cell layers PCL
- Polydendrocytes oligodendrocyte precursor cells (OPCs) Aβ activates GSK3β Glycogen synthase kinase 3 beta increased phosphorylation of β-catenin β-catenin degradation inhibition of the (Wnt) signaling pathway inhibition of the differentiation of NG2-glia
- more than 45 phosphorylation site control microtubule hyperP, dissociation, intracell neurofib tangle, No axonal transport of M, between cell bodies Energy dysfunction ROS N spec
- awtohpfagee
- Constant Across axons High E demand Process facilitates: Neuroprot Removal of defects Protects against ROS AD: Abnormal morphology Less ATP Antioxidant enzymes impaired Oxidative phosphorylation complexes defective AD neurons, perinuclear, oxidative stress, synaptic dysfunction Reasons: hPtau in neurofib tangles, also complex Abeta interaction, ROS prod by free metal ions, defective autophagy,
- organelle-, protein- lipid-degrading pathway mediated by membranes, vesicles and lysosomes, essential for protein, lipid and organelle homeostasis to ensure cell health turn-over of mitochondria called mitophagy adapt mitochondria to energy demands eliminate dysfunctional mitochondria, lysosomal degradation Macroautophagy Microautophagy Chaperon-mediated autophagy synaptic plasticity anti-inflammatory function in glial cells oligodendrocyte development myelination process cellular energy levels, increased ROS, and impaired neuroplasticity Mouse model
- enriched environment standard housing Doublecortin Heterozy/homozyg
- human neuronal cell adhesion molecule, Neurofilament Medium Chain, Choline Acetyltransferase--ChAT catalyzes the transfer of an acetyl group from the coenzyme acetyl-CoA to choline, yielding acetylcholine (ACh), Vesicular GABA Amino Acid Transporter (VGAT) is responsible for transport of the inhibitory neurotransmitter into synaptic vesicles, anti-human nuclei
- reduced Aβ deposits tau-phosphorylation, increased neurogenesis secreted factors improved learning and memory
- participant enrollment, time for transplantation, gender differences duration of monitoring cell type and source, delivery systems, long-term safety and efficacy, reaction of the implanted cell to the AD environment mechanisms of action in the AD model exogenous neuroreplacement, endogenous neurogenesis, genetic manipulations and pharmacological agents