Pradip liposomes
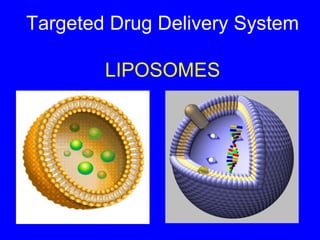
- 1. Targeted Drug Delivery System LIPOSOMES
- 2. Learning Objectives • Introduction • Mechanism of liposome formation • Classification of liposomes • Material used in preparation of liposomes • Methodes of liposome preparation • Characterization of liposomes • Stability • Therapeutic application • References
- 3. What are Liposomes? • They are simply vesicles or ‘bags’ in which an aqueous volume is entirely enclosed by a membrane composed of lipid (fat) molecules, usually phospholipids.
- 4. •Structurally, liposomes are bilayered vesicles in which an aqueous volume is entirely enclosed by a membranous lipid bilayer mainly composed of natural or synthetic phospholipids. • These vesicles can encapsulate water-soluble drugs in their aqueous spaces and lipid soluble drug within the membrane itself. • The unique property of liposomes, namely their versatile, biodegradable, hypoallergenic nature, along with their similarity to biological membranes are the important factors in the continued efforts to develop liposomal drug delivery froms.
- 5. Advantages of liposome : • Provides selective passive targeting to tumor tissues • Increased efficacy and therapeutic index • Increased stability via encapsulation • Reduction in toxicity of the encapsulated agent. • Improved pharmacokinetic effects • Used as carriers for controlled and sustained drug delivery • Can be made into variety of sizes.
- 6. Disadvantages of liposome : • Leakage of encapsulated drug during storage. • Uptake of liposomes by the reticuloendothelial system • Batch to batch variation • Difficult in large scale manufacturing and sterilization • Once administered, liposomes can not be removed • Possibility of dumping, due to faulty administration
- 7. Liposomes Evolution : • 1965 First description of closed lipid bilayer vesicles. • 1967 introduction of the term liposomes to describe closed lipid bilayer vesicles • 1972 liposomes first used as delivery systems of drugs • 1974 first patients to be injected with liposomes • 1979 liposomes first used as delivery systems of nucleic acids to cells • 1980 first monoclonal anti body targeted liposomes termed imunoliposomes
- 8. • 1987 first synthetic cationic liposomes deliver genes to cells • 1987 first sterically stabilized long circulating liposomes system introduced • 1992 first lioposome based non viral vecrtor gene therapy clinical trail on cystic fibrosis patients • 1993 frist liposome based vaccine against hepatitis A is marketed • 1995 first long circulating immunoliposomes • 1995 the liposomes encapsulated from of the anticancer drug doxorubicin and daunorubicin approved for human use • 1997 first liposomes based DNA vaccine
- 9. Mechanism of liposome formation • In order to understand why liposomes are formed when phospholipids are hydrated, it requires a basic understanding of physiochemical features of phospholipids. • Phospholipids are amphipathic molecules (having affinity for both aqueous and polar moieties) as they have a hydrophobic tail is composed of two fatty acids containing 10-24 carbon atoms and 0-6 double bonds in each chain.
- 10. • In aqueous medium the phospholipid molecules are oriented in such a way that the polar portion of the molecule remains in contact with the polar environment and at the same shields the non-polar part. • They align themselves closely in planer bilayer sheets to minimize the interaction between the bulky aqueous phase and long hydrocarbon fatty acyl chains. • This alignment requires input of sufficient amount of energy (in the form of shaking, sonication, homogenization, heating, etc).
- 11. • Interactions are completely eliminated when these sheets fold over themselves to form closed, sealed and continuous bilayer vesicles.
- 12. Classification of liposomes 1. Multilamellar vesicles (MLVs) : consist of several bilayers and having size ranging from 100nm-20m 2. Small unilamellar vesicles (SUVs) : composed of single lipid bilayer with dimeter ranging from 20- 100nm 3. Large unilamellar vesicles (LUVs) : consist of single bilayer with diameter ranging from 0.1-1m 4. Multivesicular vesicles (MVVs) : consist of vesicles with size ranging from 100nm-20m
- 14. Materials used in preparation of liposomes A.Phospholipids : • It is the major component of the biological membrane. • Two types of phospholipids are used natural and synthetic phospholipids. • The most common natural phospholipid is the phospatidylcholine (PC) is the amphipathic molecule and also known as lecithin. • It is originated from animal (hen egg) and vegetable (soyabean).
- 16. B. Steroids : • Cholesterol is generally used steroid in the formulation of liposomes. • It improves the fluidity of the bilayer membrane and reduces the permeability of bilayer membrane in the presence of biological fluids such as blood / plasma. • Cholesterol appers to reduce the interactions with blood proteins.
- 17. Methods of liposomes preparations Passive loading Active loading technique technique Mechanical dispersion Detergent removal Solvent dispersion methods methods methods
- 18. • Mechanical dispersion methods Lipid is solublised in organic solvent, drug to be entrapped is solubilise in aqueous solvent, the lipid phase is hydrated at high speed stirring due to affinity of aqueous phase to polar head it is entrapped in lipid vesicles. e.g. Lipid film hydration, Micro-emulsification ( Microfluidizer ), Sonication, Dried reconstituted vesicle
- 19. • Solvent dispersion methods in this method, lipids are first dissolved in organic solvent, which then brought in to contact with aqueous phase containing material which is to be entrapped in liposome under rapid dilution and rapid evaporation of organic solvent. e.g. Ethanol injection Ether injection De-emulsification • Detergent removal methods
- 20. Physical Characterization Parameter Characterization method Vesicle shape and surface Transmission electron microscopy, morphology Freeze-fracture electron microscopy Mean vesicle size and size Dynamic light scattering, zetasizer, distribution Photon correlation spectroscopy, laser light scattering, gel permeation and gel exclusion Surface charge Free-flow electrophoresis Electrical surface potential Zetapotential measurements & pH sensitive and surface pH probes Percent of free drug/ Minicolumn centrifugation, ion-exchange percent capture chromatography, radiolabelling Drug release Diffusion cell/ dialysis
- 21. Chemical Characterization Parameter Characterization method Phospholipid concentration Barlett assay, stewart assay, HPLC Cholesterol concentration Cholesterol oxidase assay and HPLC Phopholipid peroxidation UV absorbance, Iodometric and GLC Phospholipid hydrolysis, HPLC and TLC Cholesterol auto-oxidation Osmolarity Osmometer
- 22. Biological Characterization Parameter Characterization method Sterility Aerobic or anaerobic cultures Pyrogenicity Limulus Amebocyte Lysate (LAL) test Animal toxicity Monitoring survival rates, histology and pathology
- 23. Stability • Physical stability : Once liposomes are formed, they behave similar to the other colloidal particles suspended in water. Neutral particles tend to aggregate or flocculate and sediment with increase in size on storage. Adding charged lipids such as stearyl amine, diactyl phosphate and phosphatidyl serine can control the aggregation. The addition of charged lipids causes repulsion and prevents major changes in the overall size of liposomes.
- 24. • Chemical stability : Phospholipids, especially those derived from natural sources, are subject to two major degradative reaction : A. Lipid peroxidation : most phospolipid liposomes contain unsaturated acyl chains as part of their molecular structure and susceptible to oxidative degradation. It can be minimized by the use of animal derived lipids like egg PC, which has less saturated lipids, use of light resistant containers, use of antioxidants are useful in minimizing oxidation.
- 25. B. Lipid hydrolysis : hydrolysis in phospolipids results in the formation of free fatty acids and lyso-lecithin. Selecting a good source of lipid, temperature, pH, and minimizing oxidation. • Biological stability : liposomes release entrapped molecules rapidly when incubated with blood or plasma. This instability is attributed to the transfer of bilayer lipids to albumin and high density liposomes.
- 26. Therapeutic applications of liposomes 1. Liposomes as drug / protein delivery vehicles • Controlled and sustain release in situ • Enhanced drug solubilization • Enzyme replacement therapy and lysosomal storage disorders • Altered pharmacokinetics and biodistribution 2. Liposomes in antimicrobial, antifungal and antiviral therapy 3. Liposomes in tumour therapy • Carrier of small cytotoxic molecules • Vehicle for macromolecules as cytokines and genes
- 27. 4. Liposomes in gene delivery • Genes and antisense therapy • Genetic (DNA) vaccination 5. Liposomes in immunology 6. Liposomes as radiopharmaceutical and radio diagnostic carrier 7. Liposomes in cosmetic and dermatology 8. Liposomes in enzyme immobilization and bioractor technology
- 28. Drug Route of Targeted administration Diseases Amphotericin-B Oral delivery Mycotic infection Insulin Oral, Ocular, Diabetic mellitus Pulmonary and Transdermal delivery Ketoprofen Ocular delivery Pain muscle condition Pentoxyfylline Pulmonary delivery Asthma Tobramycin Pulmonary delivery Pseudomonas infection, aeruginosa
- 29. Drug Route of Targeted administration Diseases Salbutamol Pulmonary delivery Asthma Benzocain Transdermal ulcer on mucous surface with pain Ibuprofen Oral delivery Rheumatoid arthritis Adrenaline Ocular delivery Glucoma, Conjectivitis Penicillin G Pulmonary delivery Meningococal, staphylococcal Methotrexate Transdermal Cancer
- 30. Marketed Drug used Target Company product diseases DoxilTM or Doxorubicin Kaposi’s SEQUUS, USA CaelyxTM sarcoma DaunoXomeTM Daunorubicin Kaposi’s NeXstar, USA sarcoma, breast & lung cancer AmphotecTM Amphotericin-B fungal infections, SEQUUS, USA Leishmaniasis VENTUSTM Prostaglandin-E1 Systemic The liposome inflammatory company, USA diseases ALECTM Dry protein free Expanding lung Britannia Pharm, powder of DPPC- diseases in UK PG babies
- 31. Marketed Drug used Target Company product diseases Topex-Br Terbutaline Asthma Ozone, USA sulphate Depocyt Cytarabine Cancer therapy Skye Pharm, USA Novasome® Smallpox vaccine Smallpox Novavax, USA Avian retrovirus Killed avian Chicken pox Vineland lab, vaccine retrovirus USA Epaxal –Berna Inactivated Hepatitis A Swiss serum & Vaccine hepatitis-A vaccine institute, Virions Switzerland
- 32. Conclusion • Liposomes are one of the unique drug delivery system, which can be of potential use in controlling and targeting drug delivery. • Liposomes are administrated orally, parenterally and topically as well as used in cosmetic and hair technologies, sustained release formulations, diagnostic purpose and as good carriers in gene delivery. • Nowadays liposomes are used as versatile carriers for targeted delivery of drug.
- 33. Reference • Targeted and controlled drug delivery, S.P.Vyas and R.K.Khar, CBS Publication 2008, Nanoparticles – page no 331 to 386. Resealed Erythrocytes – page no 387 to 416. Liposomes – page no 173 to 248. • Text book of Industrial Pharmacy, Shobha Rani Hiremath, Orient Longman Private ltd. Nanoparticles – 129 to 142. Liposomes – 97 to 110
- 34. • Medical applications of nanotechnology by Timothy E. Morey, University of Florida. • Liposome: A versatile platform for targeted delivery of drugs by Sanjay S. Patel • Resealed Erythrocytes : A Review by A.V.Gothoskar MIT Campus, Pune. • www.pharmainfo.net • www.google.com
- 35. Any question...
- 36. Thank You
