Transfer of heat
•Download as PPTX, PDF•
3 likes•3,486 views
conduction, convection and radiation
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Heat Transfer Lesson PowerPoint, Convection, Conduction, Radiation, Labs

Heat Transfer Lesson PowerPoint, Convection, Conduction, Radiation, Labs
Viewers also liked
Viewers also liked (15)
Similar to Transfer of heat
Similar to Transfer of heat (20)
coductionconvectionandradiation-120822223812-phpapp02.ppt

coductionconvectionandradiation-120822223812-phpapp02.ppt
Heat Transfer Basic Laws and their applications in simple language .pdf

Heat Transfer Basic Laws and their applications in simple language .pdf
0222 conduction-convection-radiation-1203645646948318-3

0222 conduction-convection-radiation-1203645646948318-3
More from Javed Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
This presentation illustrates the research study which I pursued during my M.S. program at the University of Tennessee-Knoxville. This is a qualitative Meta-Analysis of science teachers professional development in formative assessment.Javed Iqbal Thesis Defense Presentation

Javed Iqbal Thesis Defense PresentationJaved Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
Presentation on Improving Students Presentation Skills

Presentation on Improving Students Presentation SkillsJaved Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
Presentation on vocabulary building

Presentation on vocabulary buildingJaved Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
Students' alternatiave framework

Students' alternatiave frameworkJaved Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
Congnitive domain of blooms' taxonomy

Congnitive domain of blooms' taxonomyJaved Iqbal Student of M.S (Teacher Education) at University of Tennessee USA
More from Javed Iqbal Student of M.S (Teacher Education) at University of Tennessee USA (20)
Presentation on Improving Students Presentation Skills

Presentation on Improving Students Presentation Skills
Psychomotor and affective domain of blooms' taxonomy

Psychomotor and affective domain of blooms' taxonomy
Recently uploaded
VIP Call Girls Napur Anamika Call Now: 8617697112 Napur Escorts Booking Contact Details WhatsApp Chat: +91-8617697112 Napur Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable. Independent Escorts Napur understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together. We provide –(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7

(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7Call Girls in Nagpur High Profile Call Girls
Recently uploaded (20)
FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756

FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756
Cheap Rate Call Girls In Noida Sector 62 Metro 959961乂3876

Cheap Rate Call Girls In Noida Sector 62 Metro 959961乂3876
Russian Call Girls In Rajiv Chowk Gurgaon ❤️8448577510 ⊹Best Escorts Service ...

Russian Call Girls In Rajiv Chowk Gurgaon ❤️8448577510 ⊹Best Escorts Service ...
Value Proposition canvas- Customer needs and pains

Value Proposition canvas- Customer needs and pains
Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...

Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...
Falcon Invoice Discounting: Empowering Your Business Growth

Falcon Invoice Discounting: Empowering Your Business Growth
Marel Q1 2024 Investor Presentation from May 8, 2024

Marel Q1 2024 Investor Presentation from May 8, 2024
The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait![The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait
Call Girls Jp Nagar Just Call 👗 7737669865 👗 Top Class Call Girl Service Bang...

Call Girls Jp Nagar Just Call 👗 7737669865 👗 Top Class Call Girl Service Bang...
SEO Case Study: How I Increased SEO Traffic & Ranking by 50-60% in 6 Months

SEO Case Study: How I Increased SEO Traffic & Ranking by 50-60% in 6 Months
Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...

Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...
Call Girls In Noida 959961⊹3876 Independent Escort Service Noida

Call Girls In Noida 959961⊹3876 Independent Escort Service Noida
Call Girls In Majnu Ka Tilla 959961~3876 Shot 2000 Night 8000

Call Girls In Majnu Ka Tilla 959961~3876 Shot 2000 Night 8000
Falcon's Invoice Discounting: Your Path to Prosperity

Falcon's Invoice Discounting: Your Path to Prosperity
(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7

(Anamika) VIP Call Girls Napur Call Now 8617697112 Napur Escorts 24x7
Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...

Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...
Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...

Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...
Transfer of heat
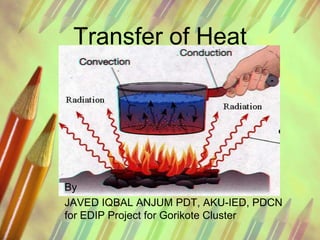
- 1. Transfer of Heat By JAVED IQBAL ANJUM PDT, AKU-IED, PDCN for EDIP Project for Gorikote Cluster
- 2. Flow of Presentation • Session Learning Outcomes • Transfer of Heat • Condition • Convection • Radiation • Expansion matter
- 3. Session Learning Outcomes • By the end of this session C.Ps could be able to; • Develop their understanding regarding conduction, convection and radiation. • Demonstrate some experiments regarding conduction, convections, expansion in liquids and solids.
- 4. Transfer of Heat • Heat can be transferred from place to place by conduction, convection and radiation. Dark matt surfaces are better at absorbing heat energy than light shiny surfaces. http://www.bbc.co.uk/schools/gcsebitesize/science/aqa_pre_2011/energy/heatrev1.s html • In the simplest of terms, the discipline of heat transfer is concerned with only two things: temperature, and the flow of heat. Temperature represents the amount of thermal energy available, whereas heat flow represents the movement of thermal energy from place to place. • http://www.efunda.com/formulae/heat_transfer/home/overview.cfm
- 5. Cont…
- 6. Conduction • Conduction is heat transfer by means of molecular agitation within a material without any motion of the material as a whole. If one end of a metal rod is at a higher temperature, then energy will be transferred down the rod toward the colder end because the higher speed particles will collide with the slower ones with a net transfer of energy to the slower ones.
- 7. Convection • Convection is heat transfer by mass motion of a fluid such as air or water when the heated fluid is caused to move away from the source of heat, carrying energy with it. Convection above a hot surface occurs because hot air expands, becomes less dense, and rises. Hot water is likewise less dense than cold water and rises, causing convection currents which transport energy • Convection can also lead to circulation in a liquid, as in the heating of a pot of water over a flame. Heated water expands and becomes more buoyant. Cooler, more dense water near the surface descends and patterns of circulation can be formed, though they will not be as regular as suggested in the drawing http://hyperphysics.phy- astr.gsu.edu/hbase/thermo/heatra.html • .
- 8. Radiation • Definition: Radiation is the emission and propagation of energy in the form of waves, rays or particles. • Examples: • A burning candle emits radiation in the form of heat and light. Electrons dropping from one energy state to a lower state emit radiation in the form of a photon. • http://chemistry.about.com/od/chemistryglossary/g/Radiation-Definition.htm
- 9. Expansion in matters • Solids, Liquids and gases expand when they are heated. This is because the particles in liquids and gases move faster when they are heated than they do when they are cold. As a result, the particles take up more volume. This is because the gap between particles widens, while the particles themselves stay the same size. • The liquid or gas in hot areas is less dense than the liquid or gas in cold areas, so it rises into the cold areas. The denser cold liquid or gas falls into the warm areas. In this way, convection currents that transfer heat from place to place are set up.
- 10. Food for thought • Science is a first-rate piece of furniture for a man's upper chamber, if he has common sense on the ground floor. • Oliver Wendell Holmes (1809-1894) The Poet at the Breakfast-Table, 1872 Chapter 5
- 11. Questions Please
