Stages of Bone healing and madalities to enhance bone healing
- 1. Stages of bone healing and madalities to enhance bone healing By Dr surya Vijay Singh
- 2. Content Structure of bone Bone healing Types of bone healing Factors affecting bone healing Modalitiesto enhance bone healing Complications
- 3. Bone is unique structure with several specific functions Major reservoir of calcium Support the human frame Origin and insertion of muscles Protects vitals soft tissues Hepls in motion
- 4. Bone structure Periosteum : 2 layers – 1) fibrous layer, 2) cambium layer Outer fibrous layer Consist dense irregular connecting tissue. Inner cambium layer/ osteogenic layer Consist primitive stem cells. It is source of new bone cells. Periosteal membrane atteched to blood vessels and nerve fibrous. Compact bone/cortical bone : Outer bone made up of dense bone contain haversian and volksmancanal, osteons, lamellae. Haversian system or osteon is functional unit of mature bone. Spongy bone/cancellous bone: Inner bone, more at ends of bone contain trabeculae, weaker than compact bone. Endosteum : Inner portion of bone marrow cavity is lined with fibrous sheet called as endosteum.
- 6. Bone composition Water-8% Solidmaterial-92% Orgenic phase – 21% inorganic phase – 71%
- 7. Fracture healing Definition : Fracture healing is complex process that requires the recruitment of appropriate cell (fibroblasts, macrophages, chondroblasts, osteobasts, osteoclast) and the subsequent expansion of the appropriate genes (those control matrix production and organisation, growth factors, transcription factors) at the right time and in the right Anatomical location.
- 8. Types of bone healing Two types of bone healing 1) Direct bone healing / primary bone healing / callous free bone healing. 2) Indirect bone healing / secondary bone healing / callous formation bone healing.
- 9. Primary bone healing If the fracture is reduced and held absolute rigidity following internal fixation and fracture Compression, then bone Heals directly with bone. Following conditions for primary bone healing ; 1. Excellent Anatomical reduction 2. Minimal or no mobility 3. Good vascular supply 4. Absolute stability achieved by interfragmentary compression with lag screw, compression plate or both. 2 types of direct / primary bone healing.
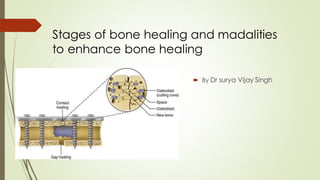
- 10. Types of direct bone healing 1) Gap healing 2) contact healing GAP HEALING : Even with rigid fixation, in some area of fracture, small gaps occur between the bone segment. Blood vessels from periosteum, endosteumor harvesian canals invade the gaps bringing osteogenic cells into gap. Bone is deposited directly over the fracture ends without resorption or Intermediate cartilage formation.
- 11. Gap healing of bone If Gap is < 0.3mm then lamellar bone forms directly. If 0.5-1 mm then woven bone formation and lamellar bone subsequently laid down with in the trabeculae space. At the end of 6 weeks lamellar bundles oriented at right angle to the Longitudinal Axis of remaining bone. Over several months, remodeling then leads to change in this direction.
- 12. Contact healing of bone Contact Healing occurs only when there is less then 0.01 mm gap at ends and Interfragmentarystrain is less than 2%. The fragments are in direct opposition and osteons are able to grow across the fracture site parallel to the long axis. Osteoclasts on one side of the fracture undergo a tunneling resorptive response(cutting cone). This develops the penetration of the capillary loops which establishes the Haversians systems. The end result of contact healing is the regenerationof the bone architecture. Osteon forms at rate of 1-2um/day.
- 13. Cutting cone of contact bone healing Cuttingcones are formed at the ends of the osteons closest to the fracture site. The tips of the cuttingcones consist of osteoclastswhich cross the fracture line, generating longitudinalcavities at a rate of 50–100 μm/day. These cavities are later filled by bone produced by osteoblastsresiding at the rear of the cuttingcone. This results in the simultaneousgeneration of a bony union and the restoration of Haversian systems formed in an axial direction.The re-established Haversian systems allow for penetrationof bloodvessels carrying osteoblasticprecursors. At cutting cone site osteoclastsform a specialized membrane, the ‘ruffled border’, which adheres to woven bone, creating Howship‘s lacunae.
- 14. Parren‘s interfragmentary strain hypothesis Perren‘s Interfragmentary strain hypothesis states that the local mechanical environment affects the tissue response. The interfragmentary strain is defined as the ratio of the relativedisplacement of fracture ends verses the initial fracture gap width. Interfragmentary strain governs the type of tissue that forms between the fracture fragments. It is inverselyproportional to the fracture gap size.
- 15. Parren’s strain hypothesis If Interfragmentary strain < 2 % = a state of absolute stability, and primary (direct) healing occurs. When 2 to 10 % = state of relativestability and secondary (indirect) bone healing occurs. If strain > 10 % = poor stability at fracture site, granulation tissue toleratethis elongation
- 16. Indirect bone healing / secondary bone healing 4 phases of fracture healing : 1) inflammatory phase 2) soft callus formation 3) hard callus formation 4) remodeling phase
- 17. Inflammatory phase of bone healing (week 1) Injury cause a haematomaand local inflammation. The fracture hematomacontains seven-foldgreater amount of membrane-boundtumor necrosis factor alpha (TNF-α) and IL-1, IL-6, IL-11 and IL-8 also elevated. The presence of cellular debris initiates an inflammatory response mediated by both local and infiltrating inflammatory cells, including platelets, polymorphonuclear cells (PMN), macrophages, and lymphocytes from surrounding soft tissue and granulation tissue formed. Neutrophils are the first cells to arrive at the fracture site, and are present at least as early as 3 hours after fracture. Cliniallythere is erythema, heat, swelling and pain (rubor, calor, tumor and door). The fracture ends are mobileand may crept us and causing pain.
- 18. Soft callus phase of bone healing (weeks 2-3) The soft callusstage of fracture healing is the differentiationof progenitor cells into chondrocytesand osteoblasts. Granulation tissueis graduallyreplacedwith fibrous connective tissue (by fibroblasts) and cartilage (by chondroblasts). Blood vessels grows into the callus(neovasularisation).This process is mainly regulated by two molecularpathways,an angiopoietin-dependentpathway, and a vascularendothelialgrowth factor (VEGF)-dependentpathway. Fragments ends become sticky and sufficient stabilityto prevent shortening, but not angulation.
- 19. Hard callus phase of bone healing (weeks 4-12) Hard callus develops when the fracture ends are held together by soft callus. Bone can formedin two ways; by intramembranous ossification (which occurs under the periosteum away from the fracture site) and by endochondral ossification (from a cartilage precursor at the fractured bone ends). This progresses centrally across the fracture gap, gradually reducing movement and strain. Calcium is laid down in the matrix and the callus then becomes visibleon radiograph. Fracture becomes rigidwith this woven bone and is united when there is no movement or crepitus at fracture site. Clinically reduction of pain and increase sense of stability at the site of fracture.
- 20. Location of callus formation 1) Gap callus between the bone ends 2) medullary (endosteal) callus along the medullary cavity. 3) periosteal callus under periosteum. Medullary callus provides some resistance to bending movements.Use of intramedullary nail minimizationmedullary callus but abundance periosteal callus production. Periosteal callus is most effective in providing resistanceto bending and torsional forces, and resisting force is proportional to the fourth power of the radius of the cross section of bone through the callus mass. In plate fixation, there is plenty of medullary callus as well as periosteal callus on opposite (compression) side of the plate.
- 21. Stages of bone healing (biochemistry)
- 22. Remodelling Of bone healing (months to years) During remodeling, the canalicular architecture of bone is reestablished, and the haversian system with its osteocytes is restored. The osteoclasts ream out a tunnel in the dead cortical bone down that a blood vessels follows,bringing in the osteoblasts that lay down the lamellar bone. Creeping substitutionreplaces the woven bone with lamellar bone. (in cancellous bone the cells are closet blood vessels and the process of bone replacement take on the surface of trabeculae, phenomenon referred to as creeping substitution). By Wolff’s law, involves the strengthening of the internal and cortical architecture of bones in response to applied physiological (dynamic) loads. The activity of bone resorption by osteoclasts and bone formation by osteoblasts is linked through the actions of RANK, RANKL, and osteoprotegerin (OPG).
- 23. Stages of bone healing (graph)
- 24. Diamond concept of bone healing Fracture healing is complex physiological process and requires the spatial and timely coordinated action of several different proteins, cell types, and expressions of hundreds of genes working towards restoring its structural integrity without scar formation. The four sided model of bone fracture healing interaction. 1) osteogeniccells (multipotent mesenchymal stem cells – MSCs) 2) growth factors – fracture hematomawhich consists signalling molecules (TNF-a, FGF, PDGF, IGF, VEGF, Interleukin IL1, IL-6,). 3) osteoconductive scaffold – the extracellular matrix provides a natural scaffold for all cellular events and interactions. 4) mechanical environment – mechanical stability is a decisive factor for bone healing.
- 25. Factors influencing # healing
- 26. Modalities to enhance bone healing Natural Bone graft Bone graft substitute Electrical and ultrasound stimulus Shock wave therapy Indications for their use include malunions, nonunions,arthrodesis, and reconstructive procedures.
- 27. Autologous bone graft It contain 3 required components for formation of bone Iliac crest is commonarea for autografting. Posterior iliac crest offers more bone for grafting than anterior. Fibulacan be used for a structural graft, ribs, distal end radius, olecrenon,Greater trochenter and proximal tibia can also be used. Either cancellous or cortical bone can be harvested depending on the procedure. The morbidity associatedwith graft harvesting, such as donor-site pain, iliac crest fracture, incisional hernia in iliac bone graft, nerve or arterial injury, and infection rates of between 8% and 10%. For defects up to 6 cm in length where immediate structural support is desired, nonvascularized cortical bone graft used. >12 cm are good candidates for vascularized grafting procedures. 6-12 cm defect have controversy. •Refers to the scaffolding /structural framework that allows bone growth. osteoconduction •Is the ability to induce /stimulate the production of osteoblasts. BMP is most common osteoinductivemediator. Osteoinduction •Material directly provides cells that will produce bone including primitive mesenchymal stem cells, osteobasts and osteoclasts. Osteogenesis
- 29. RIA(reamer, irrigator, aspirator) This is a novel intramedullary (IM)-reaming devicethat is used to irrigate and aspirate the bone marrow canal. A relatively new technique developedin 2005 to overcome the donor-site morbidity associated with autologous bone- graft. RIA developedto decrease intramedullary pressure and fat embolismduring reaming. These marrow bony fragments are rich in mesenchymal stem cells along with rich in FGF-2, IGF-B1, and TGF-B But not BMP2. Overall, RIA appears to be a safe and effective method for autologous graft harvesting, leading to less persistent postoperative pain. Complications like perforation of ant. Cortex of femur.
- 30. Bone graft substitute Bone graft substitute classifiedinto 5 major categories : 1) allograft based 2) factor based 3) cell based 4) ceramicbased 5) polymer based
- 31. 1) Allograft based bone graft substitute Allograft bone is prepared and sterilizedvia freeze-drying, freezing, or irradiation. Freeze-drying, or lyophilization, viaa removal of water and vacuum packing of the tissue significantly reduces immunogenicity. Such treatment of the graft reduces its mechanical integrity, thereby diminishingits load-bearing properties. Irradiation has a similar effect on mechanical strength, decreasing it in a dose-dependent fashion. Both freeze-drying and irradiation reduce the osteoinductive potential of the allograft by inducing the death of its osteogenic cells. Demineralized bone matrix (DBM) is the decalcified form of allograft that contains the osteoinductive proteins that stimulate bone formation. It is supplied as a putty, injectable gel, paste, powder, strips, and mixtures of these. Demineralized allograft usually is mixed with a carrier such as glycerol, calcium sulfate powder, sodium hyaluronate, and gelatin. Sterilization of DBM by gamma irradiation and ethylene oxide exposure decreases the risk of disease transmission but also may decrease the osteoinductive activity of the product.. contraindicated in patients with severe vascular or neurologic disease, fever, uncontrolled diabetes, severe degenerative bone disease, pregnancy, hypercalcemia, renal compromise, Pott disease, or osteomyelitis or sepsis at the surgical site. Risk of disease transmission like bacterial infection, hepatitis B, C .
- 32. 2)Factor based bone graft substitute Urist first discovered bone morphogenetic protein (BMP) in 1965 when he recognized its ability to induce enchondral bone formation. Most of the BMPs used today are in the bone superfamily transforming growth factor-beta (TGF-β). Presently, only two proteins have been isolated, produced, and approved for use in humans. Because they are produced by the recombinant process, they are designated rhBMP-2 and rhBMP-7. Other BMPs that have been shown to have osteogenic properties are BMP-4, -6, and -9. FDA) has approved rhBMP-2 for use in anterior lumbar fusion with a titanium cage. BMP-2 and BMP-7 are water soluble and require a carrier to remain in the operative area to be effective. Other proteins bone formation include platelet-derived growth factor (PDGF) and vascular endothelial growth factor (VEGF).
- 33. 3) Cell based bone graft substitute Cells may be used to stimulate or seed cells for new tissue.Presently, the most frequently used cell-based graft is autologous bone marrow. In the future, adult and embryonic stem cells, somatic stem cells such as bone marrow stromal cells, dermal stem cells, and cells in fetal cord blood may be grown for use as grafts. Collagen in its denatured form is an osteoinductive material. It is used as a carrier for BMP.
- 34. 4)Ceramic based bone graft substitute Ceramic and collagen bone substitutes can provide osteoconduction without the risk of disease transmission. ceramics include calcium sulfate, calcium phosphate, and bioactive glass. In addition to osteoconduction these products are osteointegrative, having the ability to form intimate bonds with the tissue. Calcium phosphate ceramics come in several varieties, including tricalcium phosphate and synthetic hydroxyapatite. Bioactive glass is silicate-based glass that is biologically active.Presently, it is used with polymethyl methacrylate to improve bonding. Used with other products as a carrier or for protection (such as cages).
- 35. 5)Polymer based bone graft substitute include both natural and synthetic polymers, biodegradable and nonbiodegradable. Some nonbiodegradable natural and synthetic polymers are composites of the polymer and a ceramic and can be used in load- bearing areas. Biodegradable natural and synthetic materials include polyglycolic acid and poly(lacticco-glycolic) acid.
- 36. Electrical and ultrasound stimulus For bone healing Electromagnetic stimulation has been used since the early 1970s in the treatment of delayed unions and nonunions, with reported success rates of 64% to 85%. but it has not been proved to be effective in the treatment of fresh fractures. Electromagnetic stimulate bone healing in following ways: 1) Direct current delivery through implantedelectrodes ( DCES- direct current electric stimulation). 2) Alternating current deliveredthrough an external coil used intermittently ( PEMF- pulsed electromagnetic field). 3) current Delivered between two plates that form a magnetic field used continuously for promoting bone healing ( CCS- capacitively coupled electrical stimulation). It helps to stimulate the secretion of numerous growth factors, including BMP-2 and BMP-4, TGF-β, and IGF-2.
- 37. Ultrasound Therapy for bone healing Low-intensity ultrasound has been shown to increase the incorporation of calcium ions in cultures of cartilage and bone cells and to stimulate the expression of numerous genes involved in the healing process, including IGF and TGF-β. Low-intensity ultrasound also has been suggested to enhance fracture healing in smokers, a group at risk for delayed unions.
- 38. Shock wave therapy for bone healing Sock wave therapy is used in hypertrophic non-union. The waves created microfractures in the hypertrophic tissue, leads to neovasularisation and osteoinduction.
- 39. Reference Campbell’s Operative Orthopaedics 13th edition Rockwood & Green’s fracture in adult 8th edition Muller manual of fracture fixation AO Group McRae’s Orthopaedics trauma