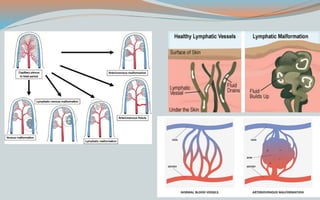
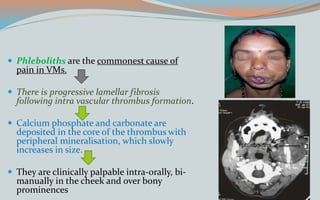
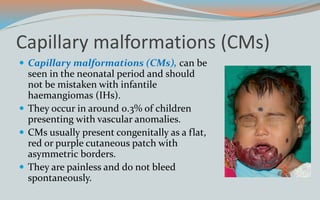
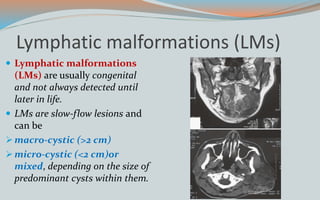
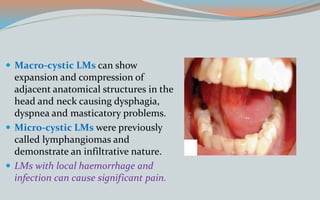
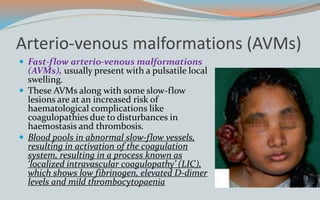
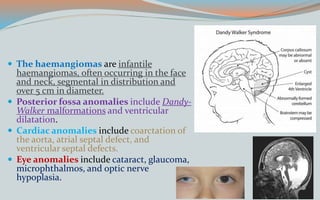
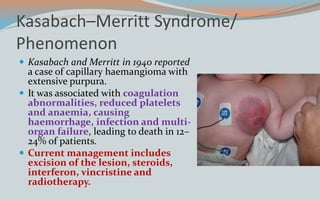
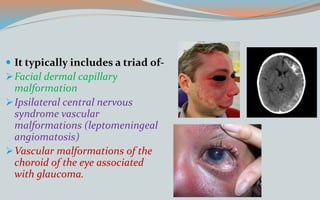
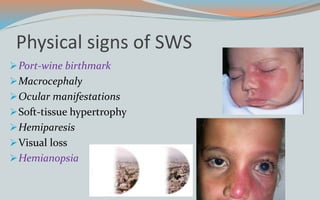
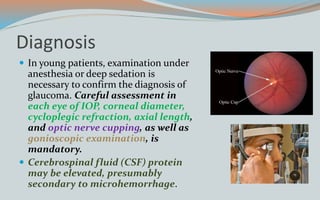
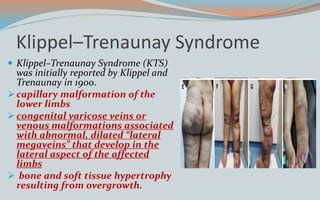
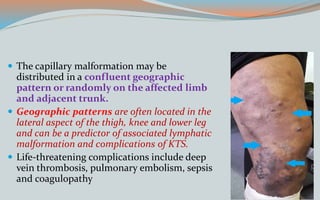
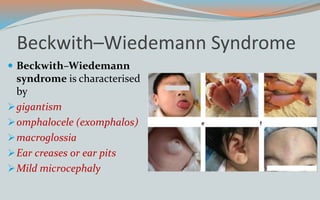
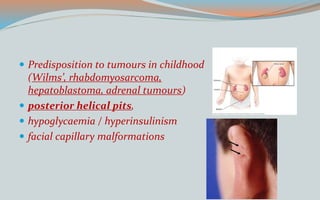
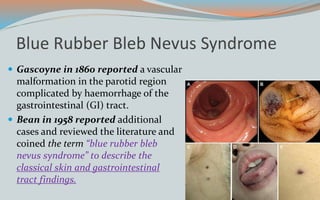
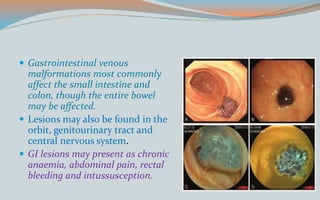
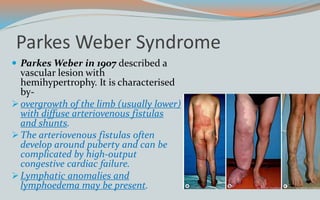
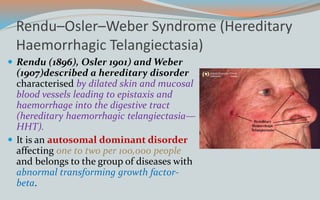
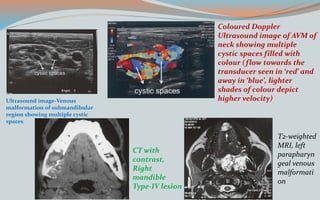
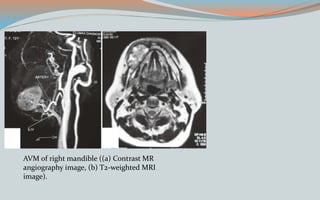
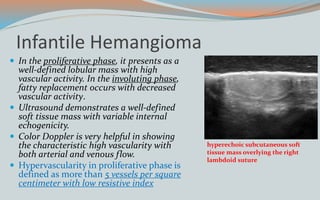
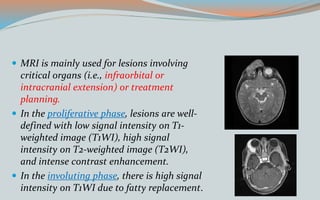
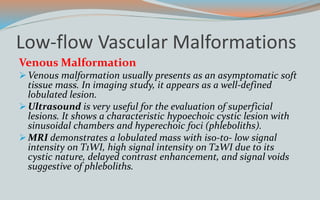
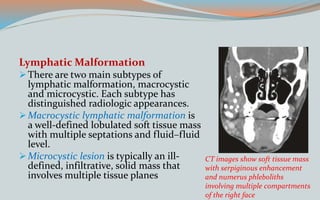
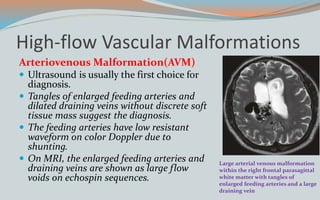
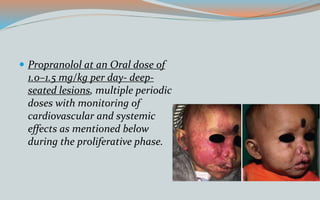
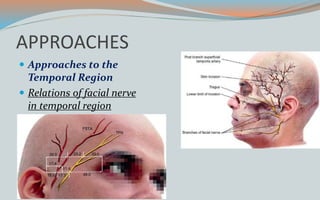
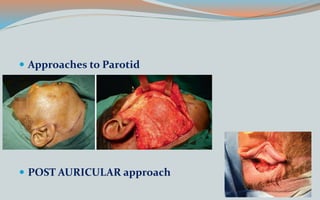
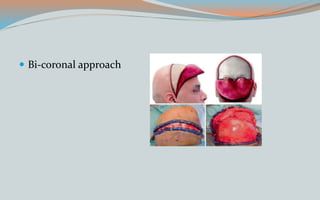
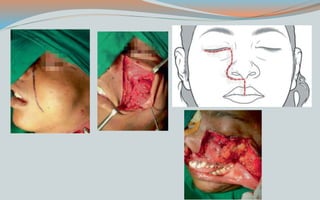
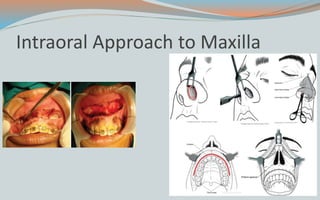
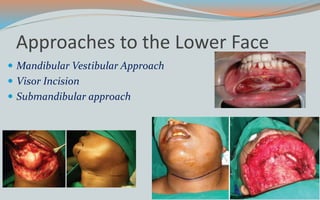
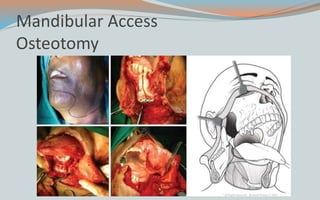
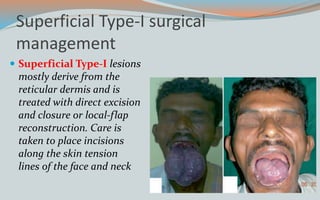
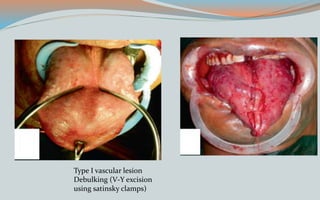
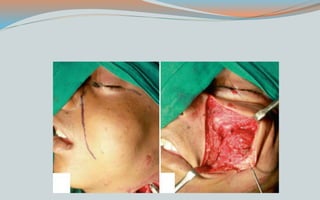
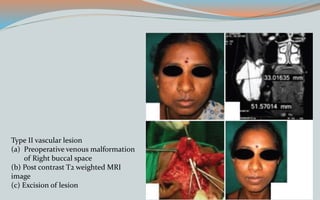
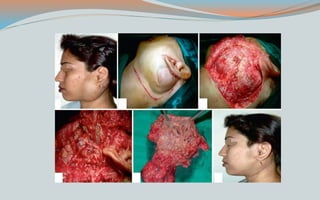
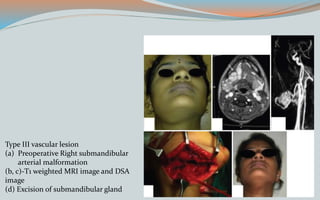
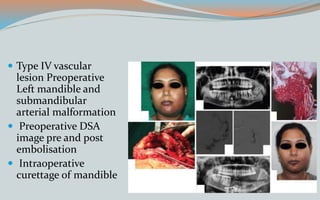
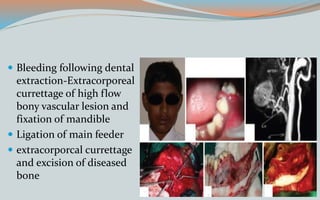
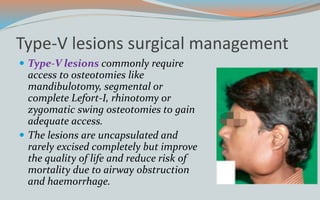
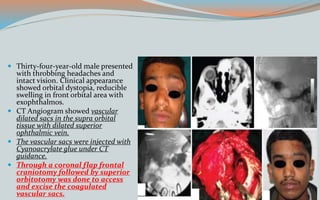
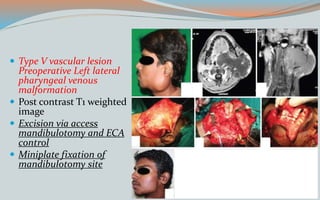
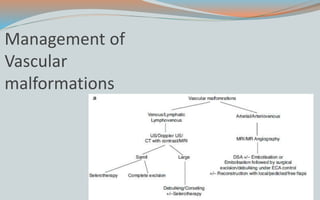
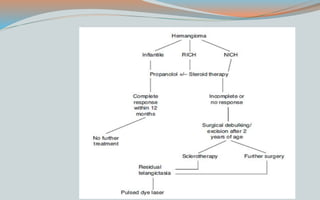
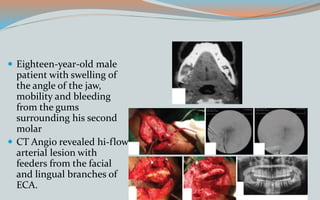
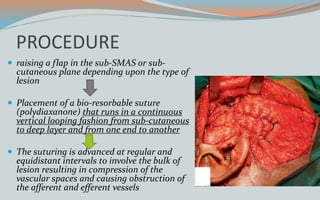
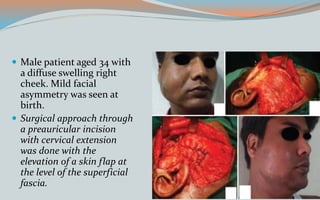
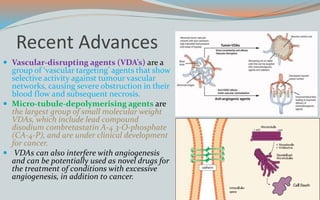
This document discusses various classifications and features of vascular anomalies. It begins with a brief history of vascular anomaly classification from Virchow's early attempts in 1863 to the current ISSVA classification system. It then covers the clinical features and classifications of specific vascular anomalies like hemangiomas, venous malformations, lymphatic malformations, and arteriovenous malformations. The document also discusses several syndromes associated with different types of vascular anomalies.