UTI power point about urinary tract infection .pptx
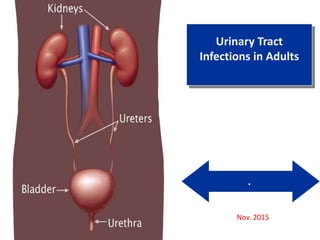
- 1. . Urinary Tract Infections in Adults Nov. 2015
- 2. Outline • Epidemiology • Pathogenesis • Diagnosis • Management
- 3. Epidemiology • UTI- second most common infection • Except among infants and the elderly, F>>> M • bellow 1 yrs of age, M > F(congenital UT anomalies) • 1- 50 years of age- predominantly diseases of females • > 50 years of age, obstruction from BPH - F=M • The prevalence of ABU is ~5% among women between ages 20 and 40 • ABU as high as 40–50% among elderly women and men
- 4. Epidemiology… • 50–80% of women acquire at least one UTI during their lifetime • About 20–30% of women who have had one episode of UTI will have recurrent episodes
- 5. Classification By symptoms • Symptomatic • Asymptomatic By Recurrences • Sporadic ≤ 1 UTI/6 mos and ≤ 2 UTIs/year • Recurrent ≥ 2 UTIs/6 mos or ≥ 3 UTIs /year Relapse Re- infection Complexity • Complicated • Uncomplicated Anatomic categories: • Lower UTI(urethritis prostaitis and cystitis) • Upper UTI (pyelonephritis, perinephric abscesses)
- 6. Mechanisms that maintain sterility of urine a.Adequate urine volume b.Free-flow from kidneys through urinary meatus c. Complete bladder emptying d.Normal acidity of urine e.Peristaltic activity of ureters and competent UV junction f. Increased intravesicular pressure preventing reflux g.In males, antibacterial effect of zinc in prostatic fluid Pathogenesis
- 7. Pathogenesis… Bacteria can get access to the urinary tract through: 1) Ascending from the urethra (main route) 2) Bloodstream ( <2%) Virulent organisms(Salmonella and S. aureus Candiduria (hematogenous route is common)
- 8. Pathogenesis… • Whether introduced bacteria into the bladder lead to sustained and symptomatic infection depends on the interplay between 1) Host 2) Pathogen and 3) Environmental factors Anything that increases the likelihood of bacteria entering the bladder and staying there increases the risk of UTI(catheter, stone , residual urine volume)
- 9. .
- 10. .
- 11. Clinical features General manifestations of cystitis a. Dysuria b. Frequency and urgency c. Nocturia d. Urine has foul odor, cloudy (pyuria), bloody (hematuria) e. Suprapubic pain and tenderness Older clients may present with different manifestations a. Nocturia, incontinence b. Confusion c. Behavioral changes d. Lethargy e. Anorexia f. Fever or hypothermia
- 12. Work up • Urine culture( indication and technique) • Urine microscope- bacteriuria(gram stain), pyuria, hematuria • Biochemical test- 1) Nitrite 2) Leukocyte esterase
- 13. Work up Urologic Evaluation: Indications women with relapsing infection a history of childhood infections stones or painless hematuria recurrent pyelonephritis most male patients with UTI signs and symptoms suggestive of an obstruction
- 14. Clinical syndromes ABU Uncomplicated cystitis Pyelonephritis Prostatitis Complicated UTI Recurrent UTI CA-UTI This will shape the diagnostic and therapeutic approach
- 15. Asymptomatic bacteriuria • Dx= both microbiologic and clinical criteria • Microbiologic criterion ≥ 105 bacterial cfu/mL (in CA- AUB≥ 102 cfu/mL) • Clinical criterion - no signs or symptoms referable to UTI • Incidental finding during a screening urine culture for other reason • Screening and treatment indicated during pregnancy • Others where Rx needed- urologic surgery, neutropenic patients and renal transplant recipients
- 16. Uncomplicated cystitis in women • Uncomplicated UTI refers to acute cystitis or pyelonephritis in nonpregnant outpatient women without anatomic abnormalities or instrumentation of the urinary tract • Risk factors: Recent use of spermicide, frequent sexual intercourse, & a history of UTI • In postmenopausal women -sexual activity, DM, and incontinence
- 17. Complicated UTI Complicated UTI is defined as UTI that increases the risk for serious complications or treatment failure
- 18. . Functional or anatomic abnormality of UT Urinary tract obstruction Prostatic hypertrophy Urethral stricture Presence of an indwelling urethral catheter, stent, nephrostomy tube or urinary diversion
- 19. UTI in DM • Diabetic Women— 2-3X higher rate of ABU and UTI • Duration of diabetes and the use of insulin associated with a higher risk of UTI • Associated with several syndromes of complicated UTI Renal and perirenal abscess Emphysematous pyelonephritis and cystitis Papillary necrosis
- 20. Emphysematous Pyelonephritis • Fulminant, necrotizing, lifethreatening variant of acute pyelonephritis • Caused by gas-forming organisms, including E. coli, K. pneumoniae, P. aeruginosa, and Proteus mirabilis • Up to 90% of cases occur in diabetic patients • Gas is usually detected by a plain abdominal radiograph or ultrasound • CT is the diagnostic modality of choice
- 21. Emphysematous… Rx • Less severely ill- Parenteral broad-spectrum antibiotics and percutaneous catheter drainage with relief of obstruction • More severely ill - Nephrectomy • Medical treatment - mortality rate of 60% to 80% • Surgical Rx- mortality lowered to 20% or less
- 22. Catheter-Associated UTI • In Patients with indwelling urethral, indwelling suprapubic, or intermittent catheterization • Defined by the presence of symptoms or signs of UTI with no other identified source of infection along with 103 cfu/mL of 1 bacterial species in a single catheter urine specimen OR • In a midstream voided urine specimen from a patient whose urethral, suprapubic, or condom catheter has been removed within the previous 48 h • The most frequent health care–associated infection worldwide(40% of all HAIs)
- 23. CA-UTI • Biofilm—a living layer of uropathogens—on the urinary catheter is central to the pathogenesis Urine for culture • Removing the indwelling catheter and from midstream urine • Replaced catheter(if ongoing catheter needed)- biofilm
- 24. CA-UTI • While the catheter is in - from the port in the drainage system • Separating the catheter from the drainage system • From the drainage bag- cannot be used to guide Rx • Condom catheters - a clean catch midstream specimen or from a freshly applied condom catheter after cleaning the glans
- 25. Recurrent UTI • ≥ 2/6 month or ≥3/year • Relapse or re-infection For clinical practice reinfection is considered 1) If the recurrence is caused by a different strain 2) Recurrence occurring >2 weeks after Rx even with the same strain as the original. 3) Urine culture (off antibiotics) is documented b/n the two UTIs • Relapse: if the infecting strain is the same and the recurrence occurs within 2 weeks of Rx completion for the original infection.
- 26. Recurrent UTI … • Risk factors — Several host genetic, biologic, and behavioral factors • Frequent sexual intercourse and spermicide use • In postmenopausal women - cystoceles, urinary incontinence, and residual urine
- 27. Recurrent UTI …risks No clear benefit from evidences: pre- and postcoital voiding patterns frequency of urination delayed voiding habits wiping patterns Douching use of hot tubs frequent use of pantyhose or tights body mass index
- 28. UTI in males • Much less common in men than in women • Due to longer urethral length, drier periurethral environment , and antibacterial substances in prostatic fluid • A diagnostic urine culture in men has colony count ≥10(4) CFU/mL. • Risks- obstruction(BPH), Lack of circumcision • Prostatitis , cystitis
- 29. Treatments Uncomplicated Cystitis in Women Preferred regimens: • Nitrofurantoin (100 mg orally twice daily for 5 days) • TMP-SMX [160/800 mg] twice daily for 3 days) • Fosfomycin (3 grams single dose) • Pivmecillinam (400 mg orally twice daily for 3-7days Fluoroquinolones are reasonable alternative agents →Collateral damage
- 30. Pyelonephritis 1) Outpatient of uncomplicated pyelonephritis Po ciprofloxacin or levofloxacin for 7 days 2) Fluoroquinolone resistance - ceftriaxone or an aminoglycoside (consolidated 24 hour dose) or a carbapenem Treatments…
- 31. UTI in Pregnant Women • Nitrofurantoin • Ampicillin • Cephalosporins Treatments…
- 32. • Uncomplicated cystitis in men- trimethoprim- sulfamethoxazole or fluoroquinolones - a 7- to 14-day course • Nitrofurantoin and beta-lactams- do not achieve reliable prostatic tissue concentrations • Chronic bacterial prostatitis → 4- to 6-week course of antibiotics • Recurrent chronic prostatitis → a 12-week course of treatment. Treatments…
- 33. CA-UTI 7- to 14-day course of antibiotics Remove/change catheter- to remove biofilm- associated organisms Prevention of CAUTI • Avoid unnecessary catheterization • Remove catheters once they are no longer necessary • Intermittent catheterization -prevent both infectious and anatomic complications. Treatments…
- 34. Prevention… Insufficient Data • Alternatives(suprapubic and condom catheters) • Frequent catheter change(every 2-4 weeks) No benefit • Systemic antibiotics • Bladder-acidifying agents • Antimicrobial bladder washes • Topical disinfectants • Antimicrobial drainage-bag solutions • Antimicrobial catheters impregnated Treatments…
- 35. Prevention of Recurrent UTI in Women • Antibiotics- Three prophylactic strategies 1) Continuous(↓recurrences by up to 95 % ) 2) Postcoital (single dose post coital) 3) Patient-initiated therapy Prescribed for 6 months and then discontinued Before prophylaxis , confirm eradication of a previous UTI by negative urine culture 1-2wks after Rx
- 36. .
- 37. Other preventive mechanisms • Cranberry juice • Topical estrogen for postmenopausal women — normalizes the vaginal flora
- 38. Thank you
Editor's Notes
- Bacteria can also gain access to the urinary tract through the bloodstream. However, hematogenous spread accounts for <2% of documented UTIs and usually results from bacteremia caused by relatively virulent organisms, such as Salmonella and S. aureus. Indeed, the isolation of either of these pathogens from a patient without a catheter or other instrumentation warrants a search for a bloodstream source. Hematogenous infections may produce focal abscesses or areas of pyelonephritis within a kidney and result in positive urine cultures. The pathogenesis of candiduria is distinct in that the hematogenous route is common. The presence of Candida in the urine of a noninstrumented immunocompetent patient implies either genital contamination or potentially widespread visceral dissemination.
- However, introduction of bacteria into the bladder does not inevitably lead to sustained and symptomatic infection. The interplay of host, pathogen, and environmental factors determines whether tissue invasion and symptomatic infection will ensue . For example, bacteria often enter the bladder after sexual intercourse, but normal voiding and innate host defense mechanisms in the bladder eliminate these organisms.
- Environmental Factors Vaginal Ecology In women, vaginal ecology is an important environmental factor affecting the risk of UTI. Colonization of the vaginal introitus and perirurethral area with organisms from the intestinal flora (usually E. coli) is the critical initial step in the pathogenesis of UTI. Sexual intercourse is associated with an increased risk of vaginal colonization with E. coli and thereby increases the risk of UTI. Nonoxynol-9 in spermicide is toxic to the normal vaginal microflora and thus is likewise associated with an increased risk of E. coli vaginal colonization and bacteriuria. In postmenopausal women, the previously predominant vaginal lactobacilli are replaced with gram-negative colonization. Anatomic and Functional Abnormalities Any condition that permits urinary stasis or obstruction predisposes the individual to UTI. Foreign bodies such as stones or urinary catheters provide an inert surface for bacterial colonization and formation of a persistent biofilm. Thus, vesicoureteral reflux, ureteral obstruction secondary to prostatic hypertrophy, neurogenic bladder, and urinary diversion surgery create an environment favorable to UTI. In persons with such conditions, E. coli strains lacking typical urinary virulence factors are often the cause of infection. Inhibition of ureteral peristalsis and decreased ureteral tone leading to vesicoureteral reflux are important in the pathogenesis of pyelonephritis in pregnant women. Anatomic factors—specifically, the distance of the urethra from the anus—are considered to be the primary reason why UTI is predominantly an illness of young women rather than of young men Host Factors The genetic background of the host influences the individual's susceptibility to recurrent UTI, at least among women. A familial disposition to UTI and to pyelonephritis is well documented. Women with recurrent UTI are more likely to have had their first UTI before age 15 years and to have a maternal history of UTI. A component of the underlying pathogenesis of this familial predisposition to recurrent UTI may be persistent vaginal colonization with E. coli, even during asymptomatic periods. Vaginal and periurethral mucosal cells from women with recurrent UTI bind threefold more uropathogenic bacteria than do mucosal cells from women without recurrent infection. Epithelial cells from susceptible women may possess specific types or greater numbers of receptors to which E. coli can bind, thereby facilitating colonization and invasion. Mutations in host response genes (e.g., those coding for Toll-like receptors and the interleukin 8 receptor) have also been linked to recurrent UTI and pyelonephritis. Polymorphisms in the interleukin 8–specific receptor gene CXCR1 are associated with increased susceptibility to pyelonephritis. Lower-level expression of CXCR1 on the surface of neutrophils impairs neutrophil-dependent host defense against bacterial invasion of the renal parenchyma. Microbial Factors An anatomically normal urinary tract presents a stronger barrier to infection than a compromised urinary tract. Thus, strains of E. coli that cause invasive symptomatic infection of the urinary tract in otherwise normal hosts often possess and express genetic virulence factors, including surface adhesins that mediate binding to specific receptors on the surface of uroepithelial cells. The best-studied adhesins are the P fimbriae, hairlike protein structures that interact with a specific receptor on renal epithelial cells. (The letter P denotes the ability of these fimbriae to bind to blood group antigen P, which contains a d-galactose-d-galactose residue
- Similarly, a combination of dysuria and urinary frequency in the absence of vaginal discharge increases the probability of UTI to 96%. Further laboratory evaluation with dipstick testing or urine culture is not necessary in such patients before the initiation of definitive therapy. When the patient's history is applied as a diagnostic tool, it is important to recall that the studies included in the meta-analysis cited above did not enroll children, adolescents, pregnant women, men, or patients with complicated UTI
- Urine culture: Indications:- Patients with symptoms or signs of UTIs Follow-up of recently treated UTI Screening for asymptomatic bacteriuria during pregnancy Patients with obstructive uropathy and stasis, before instrumentation Methods: Midstream urine voided into a sterile container after careful washing (water or saline) of external genitalia (any soap must be rinsed away). Urine obtained by single catheterization or suprapubic needle aspiration of the bladder. Sterile needle aspiration of urine from the tube of a closed catheter drainage system (do not disconnect tubing to get specimen). Interpretation of urine cultures: Asymptomatic patients; two consecutive urine specimens of ≥105 bacteria of a single species per milliliter should be demonstrable in both specimens presence of bacteriuria of any degree in suprapubic aspirates ≥102 bacteria per milliliter of urine obtained by catheterization Diuresis may transiently reduce an otherwise high colony count. The first 10ml urine…urethra Mid stream urine…bladder After prostatic massage…prostate B. Microscopic examination of urine. Immediately available and low cost Lack of sensitivity, specificity, or both Microscopic bacteriuria (by Gram-stain) is found in >90% of specimens from patients whose infections are associated with colony counts of at least 105/mL The detection of bacteria by urinary microscopy thus constitutes firm evidence of infection. The absence of microscopically detectable bacteria does not exclude the diagnosis. Pyuria is a highly sensitive indicator of UTI in symptomatic patients but not specific Pyuria in the absence of bacteriuria (sterile pyuria) may indicate infection with unusual agents(Eg. TB) or noninfectious urologic conditions such as calculi. In the catheterized patient, pyuria is not diagnostic of CA-bacteriuria or CA-UTI. C. Biochemical tests for bacteriuria Absence of glucose and presence of nitrite Studies with nitrite-indicator strips show that 85% of women and children with culture-confirmed significant bacteriuria show positive results if three consecutive morning urine specimens are tested. The sensitivity of the glucose-use test is about 90% to 95% in patients without DM. A negative nitrite test in; diuretic treatment, Infection with organisms that do not produce nitrate reductase(Staphylococcal species, Enterococcus species, and Pseudomonas aeruginosa) D. Urologic Evaluation: Indications women with relapsing infection a history of childhood infections stones or painless hematuria recurrent pyelonephritis most male patients with UTI signs and symptoms suggestive of an obstruction Understanding the parameters of the dipstick test is important in interpreting its results. Only members of the family Enterobacteriaceae convert nitrate to nitrite, and enough nitrite must accumulate in the urine to reach the threshold of detection. If a woman with acute cystitis is forcing fluids and voiding frequently, the dipstick test for nitrite is less likely to be positive, even when E. coli is present. The leukocyte esterase test detects this enzyme in the host's polymorphonuclear leukocytes in the urine, whether the cells are intact or lysed. Many reviews have attempted to describe the diagnostic accuracy of dipstick testing. The bottom line for clinicians is that a urine dipstick test can confirm the diagnosis of uncomplicated cystitis in a patient with a reasonably high pretest probability of this disease. Either nitrite or leukocyte esterase positivity can be interpreted as a positive result. Blood in the urine may also suggest a diagnosis of UTI. A dipstick test negative for both nitrite and leukocyte esterase in the same type of patient should prompt consideration of other explanations for the patient's symptoms and collection of urine for culture. A negative dipstick test is not sufficiently sensitive to rule out bacteriuria in pregnant women, in whom it is important to detect all episodes of bacteriuria. Performance characteristics of the dipstick test differ in men (highly specific) and in noncatheterized nursing home residents (highly sensitive). Urine microscopy reveals pyuria in nearly all cases of cystitis and hematuria in ~30% of cases The detection of bacteria in a urine culture is the diagnostic "gold standard" for UTI; unfortunately, however, culture results do not become available until 24 h after the patient's presentation. Identifying specific organism(s) can require an additional 24 h. In men, the minimal level indicating infection appears to be 103/mL. Urine specimens frequently become contaminated with the normal microbial flora of the distal urethra, vagina, or skin. These contaminants can grow to high numbers if the collected urine is allowed to stand at room temperature. In most instances, a culture that yields mixed bacterial species is contaminated except in settings of long-term catheterization, chronic urinary retention, or the presence of a fistula between the urinary tract and the gastrointestinal or genital tract.
- Urologic evaluation is probably not necessary in young healthy men with no obvious complicating factors who have a single episode of cystitis that responds promptly to antimicrobial treatment.
- The differential diagnosis to be considered when women present with dysuria includes cervicitis (C. trachomatis, Neisseria gonorrhoeae), vaginitis (Candida albicans, Trichomonas vaginalis), herpetic urethritis, interstitial cystitis, and noninfectious vaginal or vulvar irritation
- Defined as two separate consecutive clean-voided urine specimens both with ≥ 105 cfu/ml of the same uropathogen in the absence of symptoms referable to the UT In pregnant women, ABU has clinical consequences, and both screening for and treatment of this condition are indicated. Specifically, ABU during pregnancy is associated with preterm birth and perinatal mortality for the fetus and with pyelonephritis for the mother. A Cochrane meta-analysis found that treatment of ABU in pregnant women decreased the risk of pyelonephritis by 75%. In all other populations, screening for and treatment of ABU are discouraged. The majority of cases of catheter-associated bacteriuria are asymptomatic and do not warrant antimicrobial therapy Evaluating for asymptomatic bacteriuria in patients with indwelling catheters is warranted only in the setting of pregnancy or prior to urologic procedures for which mucosal bleeding is anticipated
- Factors independently associated with pyelonephritis in young healthy women include frequent sexual intercourse, a new sexual partner, a UTI in the previous 12 months, a maternal history of UTI, diabetes, and incontinence.
- . Poor bladder function, obstruction in urinary flow, and incomplete voiding are additional factors commonly found in patients with diabetes that increase the risk of UTI. Impaired cytokine secretion may contribute to ABU in diabetic women.
- Xanthogranulomatous Pyelonephritis Xanthogranulomatous pyelonephritis is a poorly understood, uncommon, but severe chronic renal infection associated with obstruction of the urinary tract. The renal parenchyma is replaced with a diffuse or segmental cellular infiltrate of foam cells, which are lipid-laden macrophages. The process may also extend beyond the renal capsule to the retroperitoneum. Its pathogenesis appears to be multifactorial, with infection complicating obstruction and leading to ischemia, tissue destruction, and accumulation of lipid deposits. Patients with xanthogranulomatous pyelonephritis are characteristically middle-aged women and have chronic symptoms such as flank pain, fever, chills, and malaise. Flank tenderness, a palpable mass, and irritative voiding symptoms are common. The urine culture is usually positive with E. coli, other gram-negative bacilli, or S. aureus. CT generally shows an enlarged nonfunctioning kidney, often the presence of calculi and low-density masses (xanthomatous tissue), and, in some cases, involvement of adjacent structures (Fig. 51.16). It may be difficult to distinguish from neoplastic disease. Broad-spectrum antimicrobials are indicated, but total or partial nephrectomy is usually necessary for cure. Ephysematous-Obstruction may be present. Symptoms are suggestive of pyelonephritis, and there may be a flank mass. Ketoacidosis is common. Pyuria and a positive urine culture are usually present Papillary necrosis may also be evident in some cases of pyelonephritis complicated by obstruction, sickle cell disease, analgesic nephropathy, or combinations of these conditions. In the rare cases of bilateral papillary necrosis, a rapid rise in the serum creatinine level may be the first indication of the condition
- The typical signs and symptoms of UTI, including pain, urgency, dysuria, fever, peripheral leukocytosis, and pyuria, have less predictive value for the diagnosis of infection in catheterized patients. Furthermore, the presence of bacteria in the urine of a patient who is febrile and catheterized does not necessarily predict CAUTI, and other explanations for the fever should be considered. Symptomatic catheter-related bacteriuria (usually referred to as UTI since a clinically significant infection is inferred) is defined as the presence of fever >38ºC, suprapubic tenderness, costovertebral angle tenderness, or otherwise unexplained systemic symptoms such as altered mental status, hypotension, or evidence of a systemic inflammatory response syndrome, together with one of the following laboratory profiles [ 4 ]: Urine culture with >10(5) cfu/mL irrespective of urinalysis results Urine culture with >10(3) cfu/mL with evidence of pyuria (dipstick positive for leukocyte esterase and/or nitrite, microscopic pyuria or presence of microbes seen on Gram stain of unspun urine). defined by bacteriuria and symptoms in a catheterized patient The accepted threshold for bacteriuria varies from 103 cfu/mL to 105 cfu/mL. The formation of biofilm—a living layer of uropathogens—on the urinary catheter is central to the pathogenesis of CAUTI and affects both therapeutic and preventive strategies. Organisms in a biofilm are relatively resistant to killing by antibiotics, and eradication of a catheter-associated biofilm is difficult without removal of the device itself. Furthermore, because catheters provide a conduit for bacteria to enter the bladder, bacteriuria is inevitable with long-term catheter use.
- Ideally urine samples for culture should be obtained by removing the indwelling catheter and obtaining a midstream specimen. If ongoing catheterization is needed, ideally the catheter should be replaced prior to collecting a urine sample for culture, to avoid culturing bacteria present in the biofilm of the catheter but not in the bladder. Many systems have a "needleless" site that can be cleansed prior to specimen collection. If a sample is being collected without catheter removal, urine should be obtained from the port in the drainage system ( figure 1 ) [ 26 ]. For circumstances in which the above approaches are not possible, the culture should be obtained by separating the catheter from the drainage system. Although this approach is associated with some risk of introducing microbes into the closed system, culture results from urine collected from the drainage bag cannot be used to guide treatment. In the setting of condom catheters it can be difficult to distinguish true infection from skin and mucosal contamination [ 14,27 ]. In these cases, a clean catch midstream specimen should be obtained, or urine should be collected from a freshly applied condom catheter after cleaning the glans
- Biologic or genetic factors — Women with recurrent UTI have been shown to have an increased susceptibility to vaginal colonization with uropathogens, even during asymptomatic periods, compared with women without a history of recurrences [ 9-13 ]. This difference appears to partially result from a greater propensity for uropathogenic coliforms to adhere to the uroepithelial cells of women with a history of recurrent UTIs as opposed to those without recurrent infection [ 14-18 ]. Genetic determinants appear to account for this underlying predisposition in some women. The nonsecretor [ 19-21 ] and the P1 [ 22,23 ] phenotypes are overrepresented among girls and women with recurrent UTI and recurrent pyelonephritis, respectively. In addition, uroepithelial cells from women who are nonsecretors of ABH blood group antigens show enhanced adherence of uropathogenic E. coli compared with cells from secretors The interleukin (IL)-8 receptor, IL-8R or CXCR1, is another factor with genetic variability that may influence the development of UTI. IL-8 is an inflammatory cytokine that promotes neutrophil migration across the infected uroepithelium Behavioral risk factors — Sexual intercourse, diaphragm-spermicide use, and a history of recurrent UTI are strong and independent risk factors for UTI Other risk factors identified were: Spermicide use during the past year Having a new sex partner during the past year Having a first UTI at or before 15 years of age Having a mother with a history of UTIs Virulence determinants of uropathogens — Certain virulence determinants of uropathogens have been demonstrated to provide a selective advantage for the ability to colonize and cause infection [ 37,38 ]. Colonization with P-fimbriated strains of E. coli, for example, is a strong risk factor for acute uncomplicated pyelonephritis. However, it is not clear whether bacterial virulence determinants of strains causing recurrent UTIs differ from those causing sporadic UTIs. It is also not known whether women with recurrent UTI have a propensity for colonization with urovirulent strains compared with women who do not have recurrent UTI.
- Prostatitis includes both infectious and noninfectious abnormalities of the prostate gland. Infections can be acute or chronic, are almost always bacterial in nature, and are far less common than the noninfectious entity of chronic pelvic pain syndrome (formerly known as chronic prostatitis). Acute bacterial prostatitis presents as dysuria, frequency, and pain in the prostatic, pelvic, or perineal area. Fever and chills are usually present, and symptoms of bladder outlet obstruction are common. Chronic bacterial prostatitis presents more insidiously as recurrent episodes of cystitis, sometimes with associated pelvic and perineal pain. Men who present with recurrent cystitis should be evaluated for a prostatic focus.
- Collateral damage refers to the adverse ecologic effects of antimicrobial therapy, including killing of the normal flora and selection of drug-resistant organisms. Outbreaks of Clostridium difficile infection offer an example of collateral damage in the hospital environment. The implication of collateral damage in this context is that a drug that is highly efficacious for the treatment of UTI is not necessarily the optimal first-line agent if it also has pronounced secondary effects on the normal flora or is likely to change resistance patterns. Drugs used for UTI that have a minimal effect on fecal flora include pivmecillinam, fosfomycin, and nitrofurantoin. In contrast, trimethoprim, TMP-SMX, quinolones, and ampicillin affect the fecal flora more significantly; these drugs are notably the agents for which rising resistance levels have been documented TMP-SMX should be avoided if the prevalence of resistance is known to exceed 20 percent or if the patient has taken TMP-SMX in the preceding three months Well-studied first-line agents include TMP-SMX and nitrofurantoin. Second-line agents include fluoroquinolone and -lactam compounds. Single-dose fosfomycin treatment for acute cystitis is widely used in Europe but has produced mixed results in randomized trials. Pivmecillinam is not currently available in the United States or Canada but is a popular agent in some European countries. The pros and cons of other therapies are discussed briefly below Traditionally, TMP-SMX has been recommended as first-line treatment for acute cystitis, and it remains appropriate to consider the use of this drug in regions with resistance rates not exceeding 20% Resistance to nitrofurantoin remains low despite >60 years of use. Since this drug affects bacterial metabolism in multiple pathways, several mutational steps are required for the development of resistance. Nitrofurantoin remains highly active against E. coli and most non–E. coli isolates. Proteus, Pseudomonas, Serratia, Enterobacter, and yeasts are all intrinsically resistant to this drug Nitrofurantoin does not reach significant levels in tissue and cannot be used to treat pyelonephritis. Most fluoroquinolones are highly effective for short-course therapy for cystitis; the exception is moxifloxacin, which does not achieve adequate urinary levels. The fluoroquinolones commonly used for UTI include ofloxacin, ciprofloxacin, and levofloxacin. The main concern about fluoroquinolone use for acute cystitis is the propagation of fluoroquinolone resistance, not only among uropathogens but also among other organisms causing more serious and difficult-to-treat infections at other sites. Fluoroquinolone use is also a factor driving the emergence of C. difficile outbreaks in hospital settings. Most experts now call for restricting fluoroquinolones to specific instances of uncomplicated cystitis in which other antimicrobial agents are not suitable. Quinolone use in the elderly has been associated with an increased risk of Achilles tendon rupture. Except for pivmecillinam, -lactam agents generally have not performed as well as TMP-SMX or fluoroquinolones in acute cystitis. Rates of pathogen eradication are lower and relapse rates are higher with -lactam drugs. The generally accepted explanation is that -lactams fail to eradicate uropathogens from the vaginal reservoir. A proposed role for intracellular biofilm communities is intriguing. Many strains of E. coli that are resistant to TMP-SMX are also resistant to amoxicillin and cephalexin; thus, these drugs should be used only for patients infected with susceptible strains. Urinary analgesics are appropriate in certain situations to speed resolution of bladder discomfort. The urinary tract analgesic phenazopyridine is widely used but can cause significant nausea. Combination analgesics containing urinary antiseptics (methenamine, methylene blue), a urine-acidifying agent (sodium phosphate), and an antispasmodic agent (hyoscyamine) are also available.
- Since patients with pyelonephritis have tissue-invasive disease, the treatment regimen chosen should have a very high likelihood of eradicating the causative organism and should reach therapeutic blood levels quickly. High rates of TMP-SMX-resistant E. coli in patients with pyelonephritis have made fluoroquinolones the first-line therapy for acute uncomplicated pyelonephritis. Whether the fluoroquinolones are given orally or parenterally depends on the patient's tolerance for oral intake. A randomized clinical trial demonstrated that a 7-day course of therapy with oral ciprofloxacin (500 mg twice daily, with or without an initial IV 400-mg dose) was highly effective for the initial management of pyelonephritis in the outpatient setting. Oral TMP-SMX (one double-strength tablet twice daily for 14 days) is also effective for treatment of acute uncomplicated pyelonephritis if the uropathogen is known to be susceptible. If the pathogen's susceptibility is not known and TMP-SMX is used, an initial IV 1-g dose of ceftriaxone is recommended. Oral -lactam agents are less effective than the fluoroquinolones and should be used with caution and close follow-up. Options for parenteral therapy for uncomplicated pyelonephritis include fluoroquinolones, an aminoglycoside with or without ampicillin, an extended-spectrum cephalosporin with or without an aminoglycoside, or a carbapenem. Combinations of a -lactam and a -lactamase inhibitor (e.g., ampicillin-sulbactam, ticarcillin-clavulanate, and piperacillin-tazobactam) or imipenem-cilastatin can be used in patients with more complicated histories, previous episodes of pyelonephritis, or recent urinary tract manipulations; in general, the treatment of such patients should be guided by urine culture results. Once the patient has responded clinically, oral therapy should be substituted for parenteral therapy.
- intermittent catheterization may be preferable to long-term indwelling urethral catheterization in certain populations (e.g., spinal cord–injured persons) to prevent both infectious and anatomic complications.
- Routine Catheter Change Data are insufficient to make a recommendation as to whether routine catheter change (eg, every 2–4 weeks) in patients with functional long-term indwelling urethral or suprapubic catheters reduces the risk of CA-ASB or CA-UTI, even in patients who experience repeated early catheter blockage from encrustation.
- The threshold of two or more symptomatic episodes per year is not absolute; decisions about interventions should take the patient's preferences into account. Three prophylactic strategies are available: continuous, postcoital, or patient-initiated therapy. Continuous prophylaxis and postcoital prophylaxis usually entail low doses of TMP-SMX, a fluoroquinolone, or nitrofurantoin. These regimens are all highly effective during the period of active antibiotic intake. Typically, a prophylactic regimen is prescribed for 6 months and then discontinued, at which point the rate of recurrent UTI often returns to baseline. If bothersome infections recur, the prophylactic program can be reinstituted for a longer period. Patient-initiated therapy involves supplying the patient with materials for urine culture and self-medication with a course of antibiotics at the first symptoms of infection. The urine culture is refrigerated and delivered to the physician's office for confirmation of the diagnosis. When an established and reliable patient-provider relationship exists, the urine culture can be omitted as long as the symptomatic episodes respond completely to short-course therapy and are not followed by relapse. Postcoital — Postcoital prophylaxis (a single postcoital dose) may be a more efficient and acceptable method of prevention than continuous prophylaxis in women whose UTIs appear to be temporally related to sexual intercourse. Depending upon the frequency of intercourse, postcoital prophylaxis usually results in receipt of smaller amounts of antimicrobials than continuous prophylaxis Most authorities advocate a six-month trial of antibiotics administered nightly, followed by observation for further infection. The choice of six months is based upon observations that UTIs seem to cluster in some women .However, it appears that most women revert back to the previous pattern of recurrent infections once prophylaxis is stopped (relative risk for at least one microbiologic recurrence 0.82, 95% CI 0.44 -1.53 in the above meta-analysis) Modification of other risk factors, such as sexual activity or diaphragm-spermicide use, would probably be beneficial in this regard. Some authorities advocate prophylaxis for two or more years in women who continue to have symptomatic infections . Use of trimethoprim-sulfamethoxazole or other agents for as long as five years has been reported to be effective and well-tolerated
- Cranberry juice — We do not routinely suggest cranberry juice to reduce the incidence of recurrent UTI. Although there are plausible biological mechanisms for such an effect, clinical studies to date have not definitively demonstrated efficacy in prevention of recurrent uncomplicated cystitis [ 40-49 ]. However, for women with recurrent UTI who are interested in trying cranberry juice and can tolerate it, there is likely little harmful effect other than an increase in calorie and glucose intake. There may also be an increased likelihood of gastrointestinal side effects such as heartburn with cranberry juice, as suggested by some studies [ 47 ]. Laboratory studies have demonstrated that cranberry juice inhibits adherence of uropathogens to uroepithelial cells [ 50,51 ]. The mediators of this anti-adherence effect may be fructose, which could interfere with adhesion of type 1 fimbriated E. coli to uroepithelium [ 52 ], and proanthocyanidins, which can inhibit adherence of P-fimbriated E. coli [ 53 ]. The reduction in urinary P-fimbriated E. coli strains in the cranberry group of one study of 176 women, while not statistically significant, supports the biological plausibility of cranberry activity