Use of dsc in pharmaceuticals drug characterisation
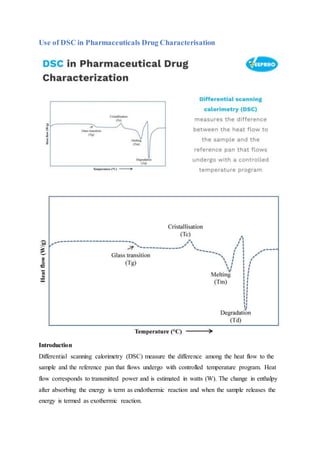
- 1. Use of DSC in Pharmaceuticals Drug Characterisation Introduction Differential scanning calorimetry (DSC) measure the difference among the heat flow to the sample and the reference pan that flows undergo with controlled temperature program. Heat flow corresponds to transmitted power and is estimated in watts (W). The change in enthalpy after absorbing the energy is term as endothermic reaction and when the sample releases the energy is termed as exothermic reaction.
- 2. Different thermal events measured by DSC such as crystallisation, onset of oxidation, melting, cure reaction and heats of transitions i.e. enthalpy. Pharmaceutical Applications: DSC is widely used to measure reaction kinetics, glass transition temp, specific heat capacity, compatibility, stability of samples, effect of aging, impact of additives on crystallization and characterization of drug substance. DSC is useful for the measurement of glass transition temperature (Tg). Glass transition is the stage at which the substance changes it states from a glass like form to a more elastic form. DSC is the useful technique to identify the polymorphic form conversions mainly due to its capability to study the sample under different heating and cooling conditions required to impact the polymorph formation and following are the events: A. Endothermic Events: When sample absorbs some amount of energy during phase conversion then reaction is termed as endothermic event. Endothermic reaction requires additional energy to sustain the zero-temp difference between sample & reference. Following are the different endothermic events: Glass Transition, Melting, Evaporation/Volatilization, Enthalpic Recovery, Polymorphic Transitions, Decompositions B. Exothermic Events: When sample evolves some amount of energy during phase conversion, then reaction is termed as exothermic event. Exothermic reaction requires less energy to sustain zero-temp difference between sample & reference. Following are the different exothermic events: Crystallization, Cure Reactions, Polymorphic Transitions, Oxidation, Decomposition, Freezing
- 3. C. Calorimetric Purity by DSC can be used to measure the absolute purity of crystalline drug substance sample with high sensitivity for detecting the low level of impurity. This is due to the melting point depression caused by the impurity, which lowers and enlarges the temperature range of melting point. A sharp symmetrical curve shows the relative purity. Conclusion: DSC is used mainly to study the thermal stability of sample. DSC is very useful in the characterisation of drug substance and drug product. DSC is speedy, simple and consistent technique which allows fast estimation of polymorphic forms, pharmaceutical drug substance/excipient compatibilities, endotherms and exotherms in the corresponding enthalpies of reaction. References: 1. Physical Characterisation of Pharmaceutical Solids, Edited by Harry G. Brittain, 2. Various Aspects of The Estimation of Impurities in Drugs, Sdndor Gorog, 3. Craig, D.Q.M., Reading, M. Thermal Analysis of Pharmaceuticals. Boca Raton, FL: CRC Press, 4. Sestak, J. Thermophysical properties of solids. In: Svehla, G. ed. Comprehensive Analytical Chemistry: Volume XII, Thermal Analysis Part D. New York, 5. Ford, J.L; Timmins, P. Pharmaceuticals Thermal Analysis – Techniques and Applications, 6. Ahuja S and Scypinski S, Eds Handbook of Modern Pharmaceutical Analysis,
