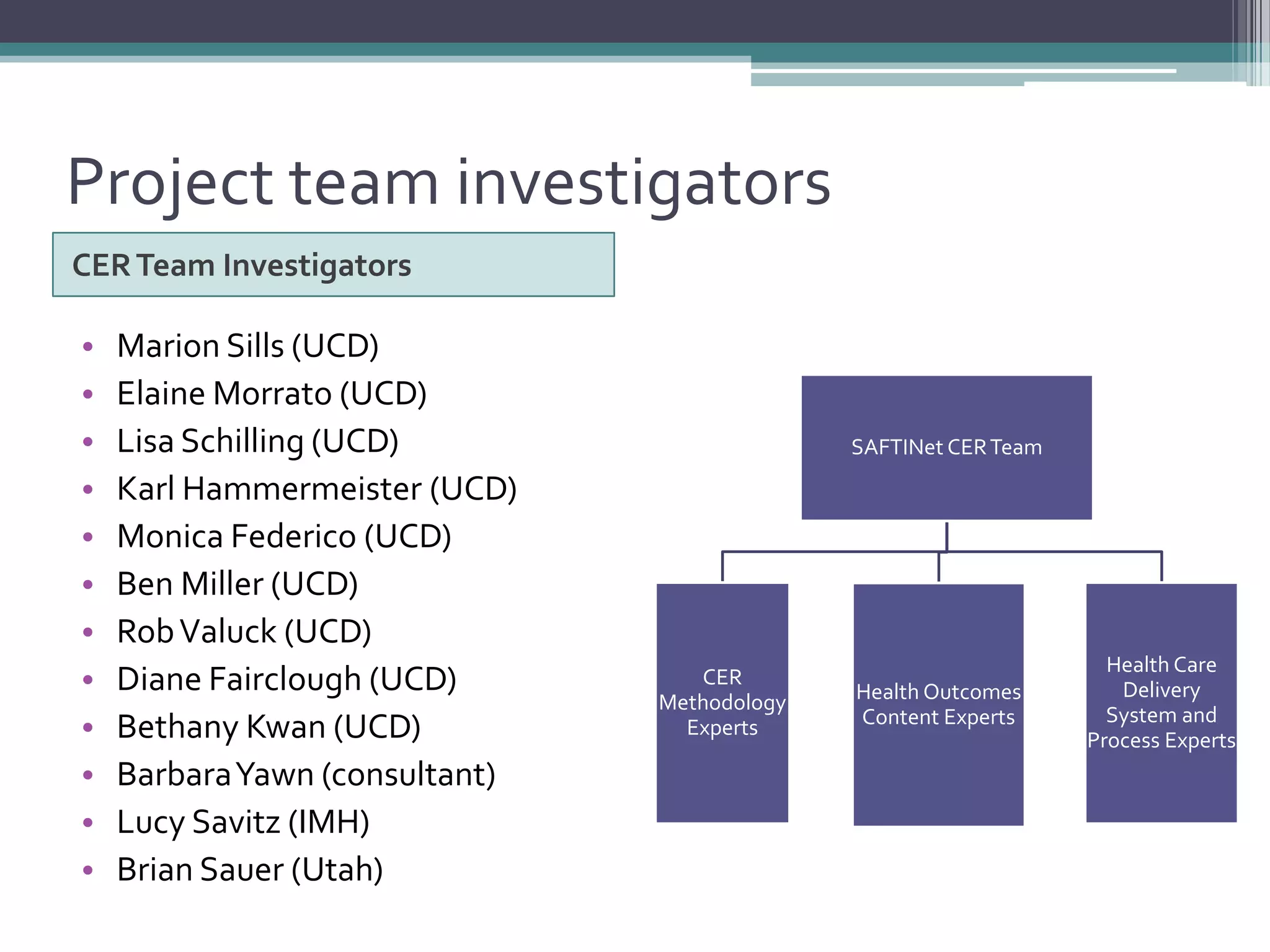
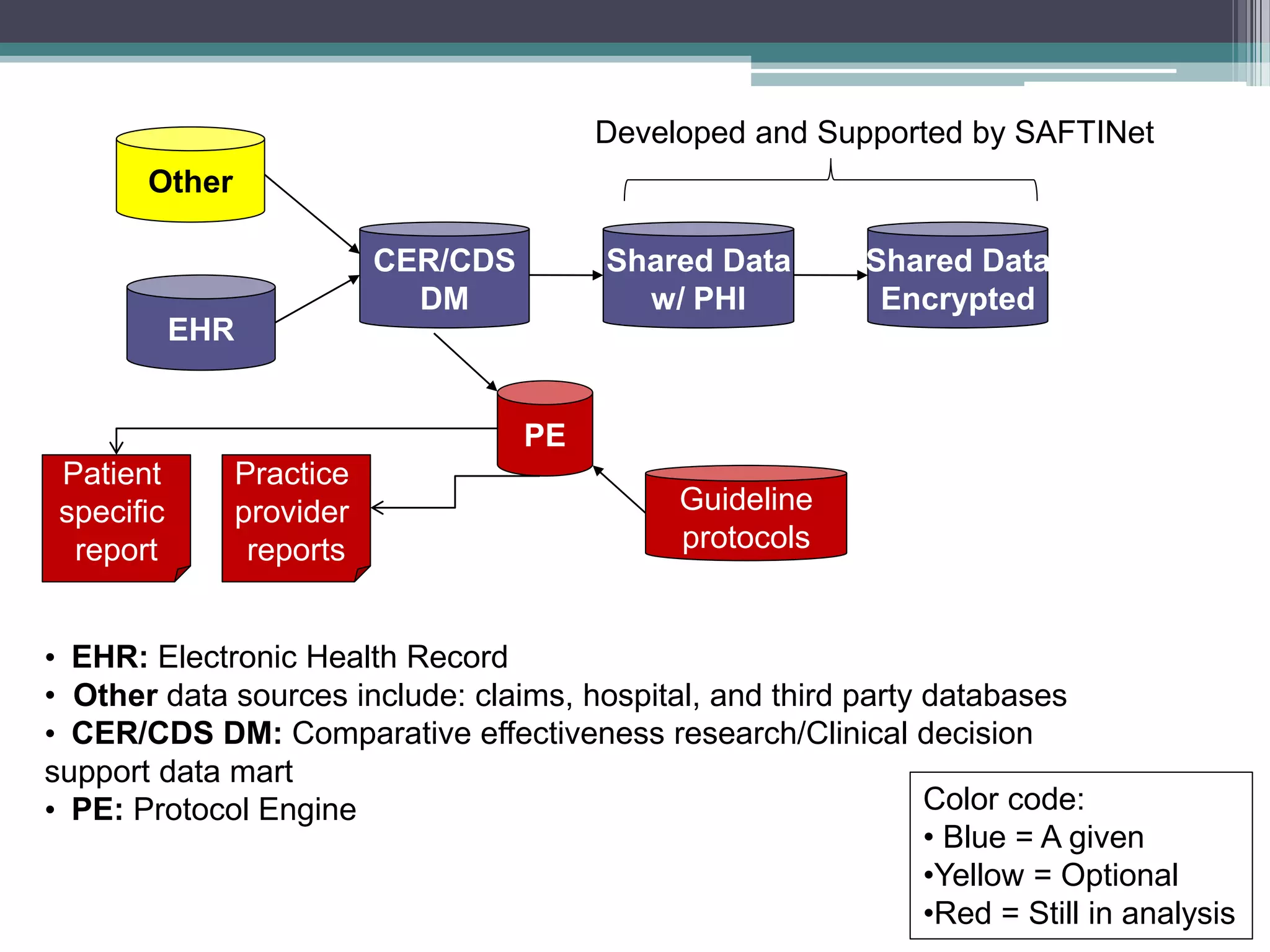
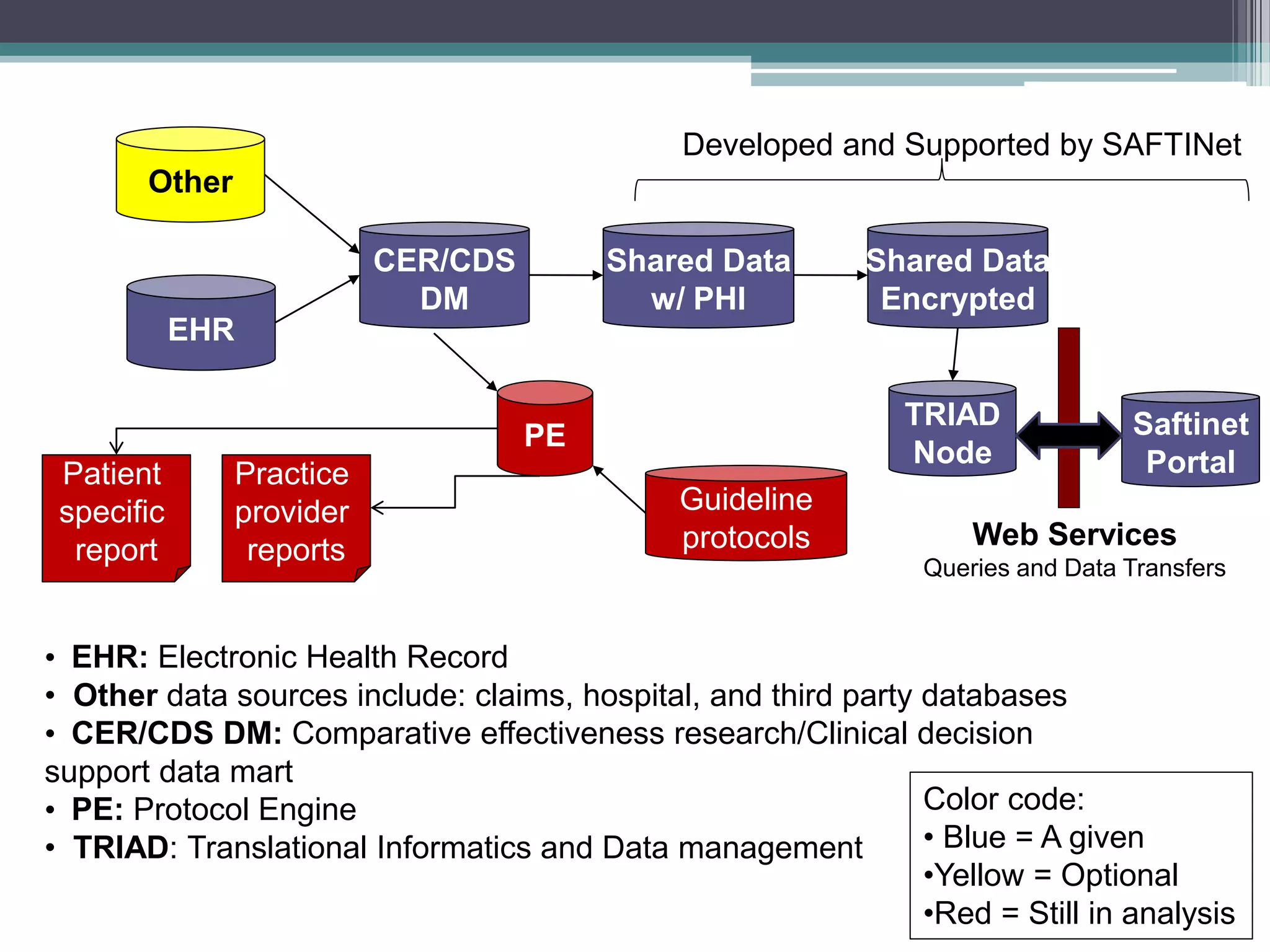
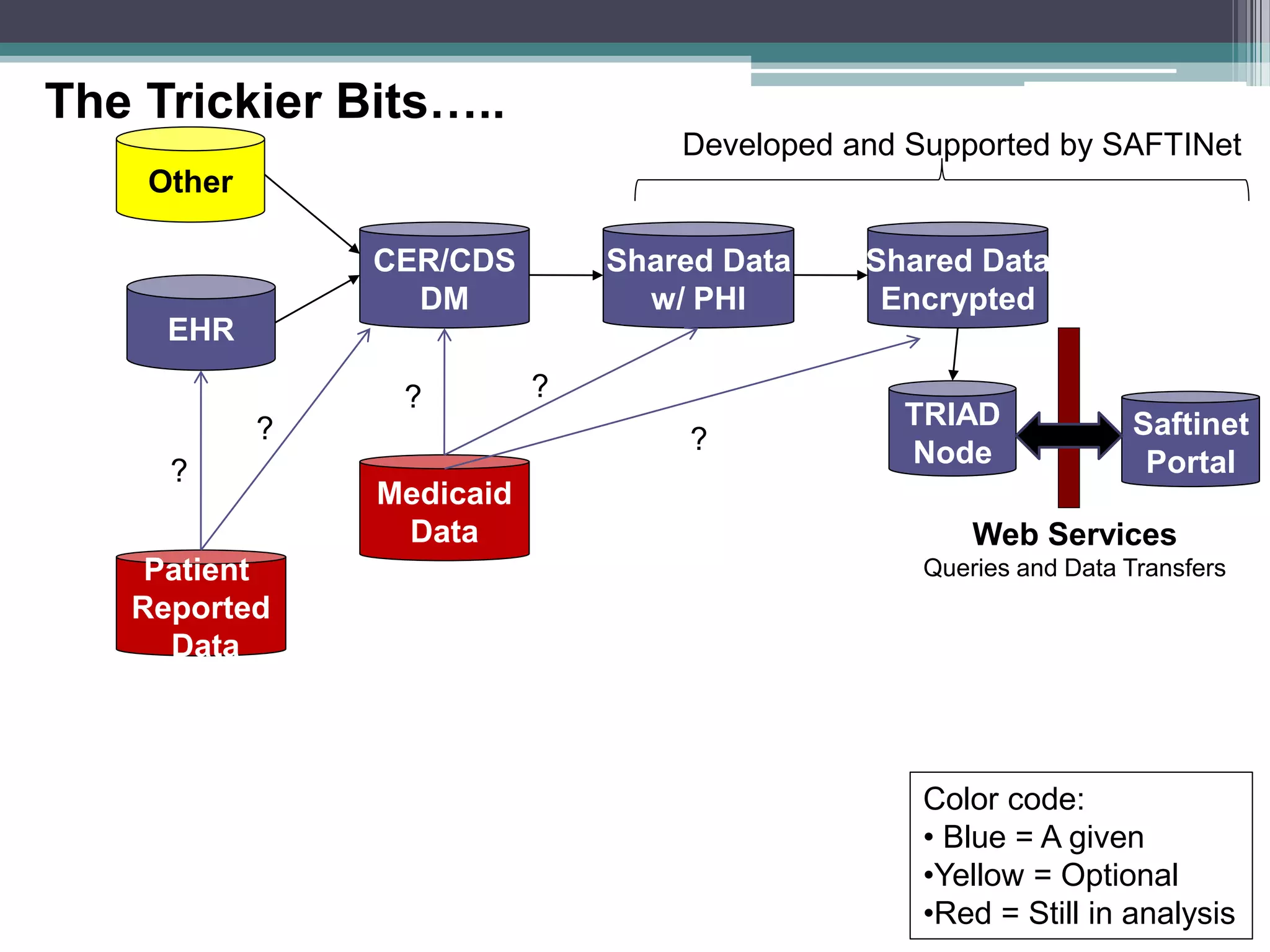
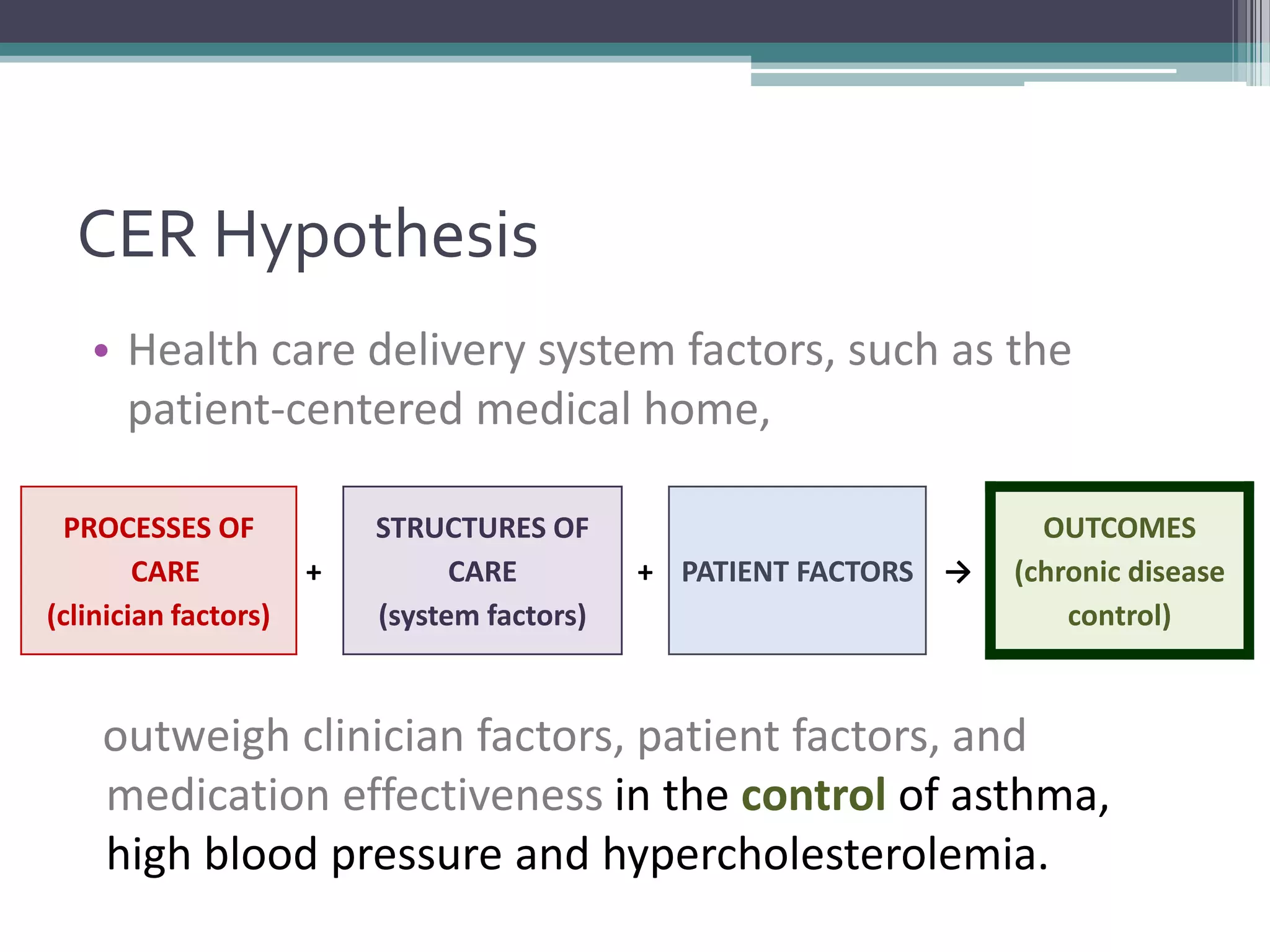
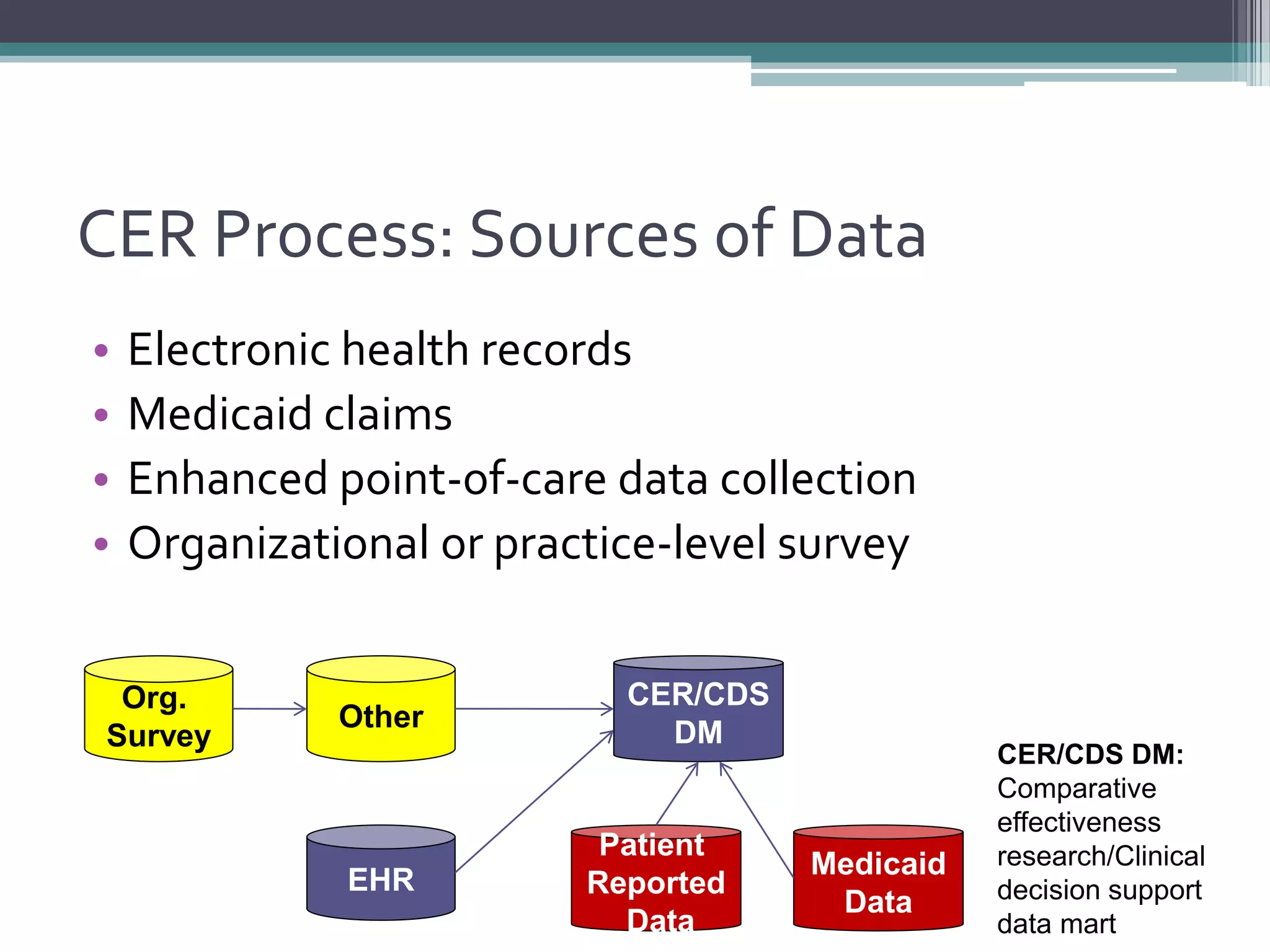
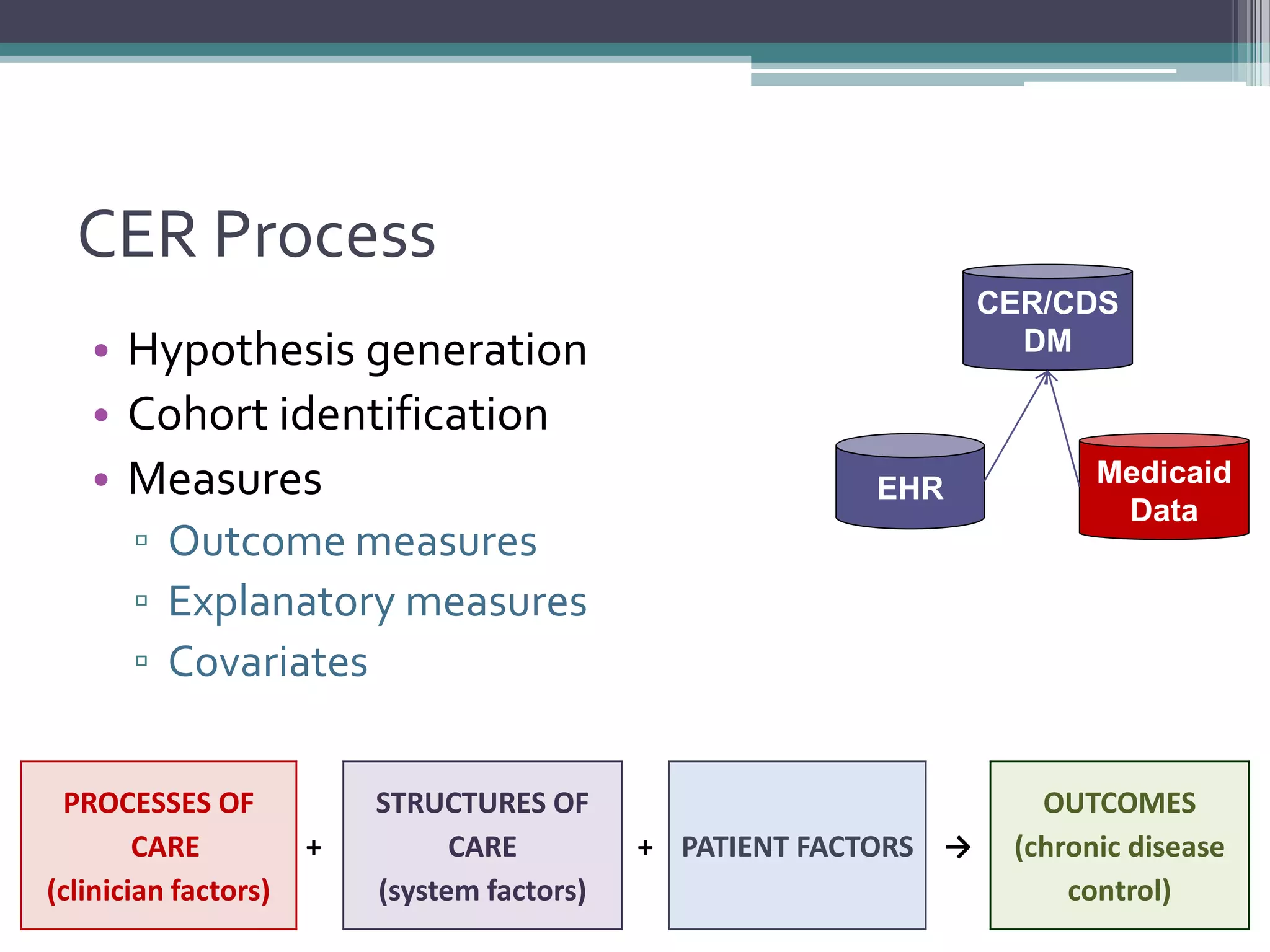
This document summarizes a kick-off meeting for the SAFTINet project. The meeting welcomed collaborators and outlined goals of establishing a distributed research network to conduct comparative effectiveness research using electronic health data from multiple healthcare organizations. The agenda included introductions of participating organizations, presentations on comparative effectiveness research and the technical capabilities needed, and discussions around engaging partners and getting started with the work.