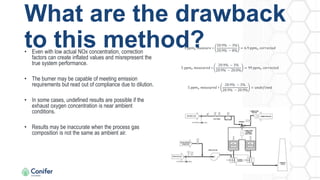
The document provides an overview of nitrogen oxides (NOx) emissions, highlighting their sources, regulation, and harmful effects. It discusses methods for quantifying NOx emissions, including the common practice of expressing them in ppmv corrected to 3% O2 and the preferred method of lb/mmbtu, which accounts for mass flow. Additionally, the document outlines various technologies for controlling NOx emissions in combustion processes.