Water's surface tension is caused by the attraction of water molecules to each other through hydrogen bonding. This allows paperclips and other objects to float, but soap disrupts this bonding causing objects to sink. Alcohol droplets have less surface tension than water droplets as the molecules are not as strongly bonded.
A meniscus is the curved surface of liquid in a narrow container. Glass shows a larger meniscus than plastic because water molecules are more strongly attracted to the glass surface. Graduated cylinders account for the meniscus in measurements. Liquid level should be read from the bottom of the meniscus.
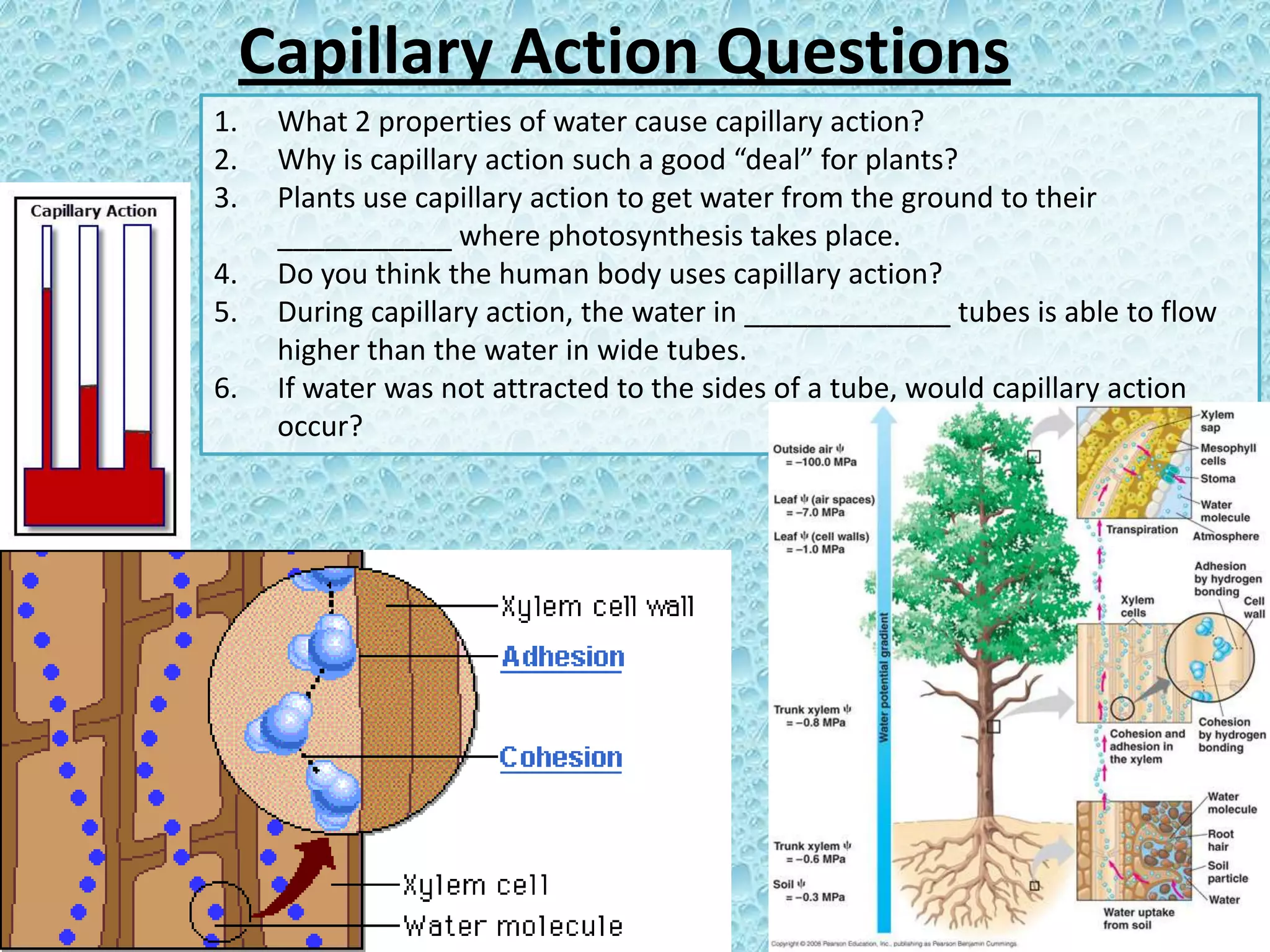
Capillary action is caused by two properties - adhesion of water to surfaces and cohesion of water