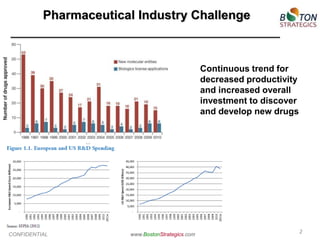
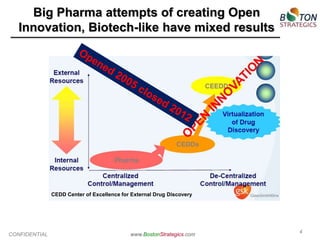
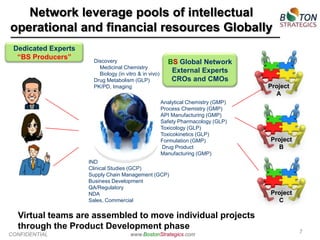
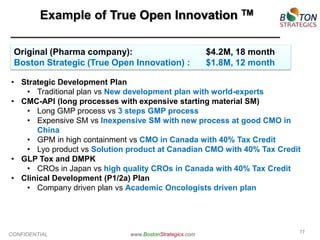
The document discusses the challenges faced by the pharmaceutical industry, particularly big pharma, in drug development due to structural and cultural issues. It introduces the concept of 'true open innovation,' which leverages a global network of resources and expertise to improve efficiency and reduce costs in drug development. Boston Strategics Corporation aims to establish this network, facilitating faster and more affordable drug discovery processes through collaboration with various stakeholders, including academic institutions and biotech firms.