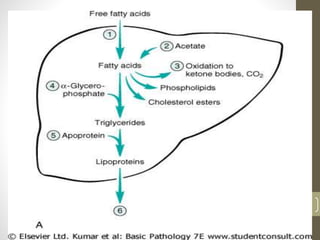
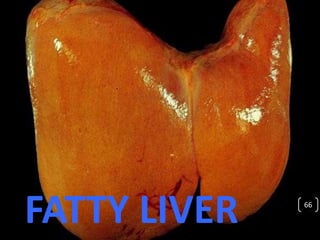
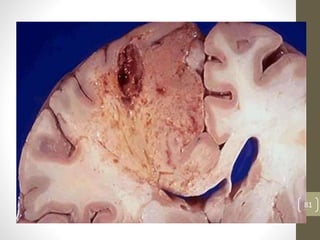
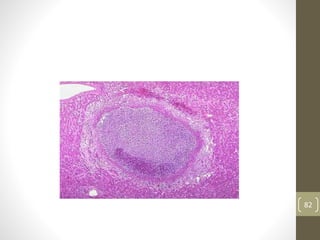
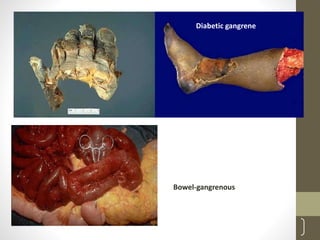
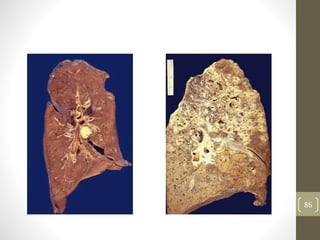
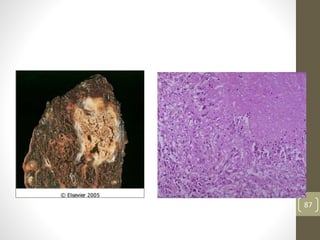
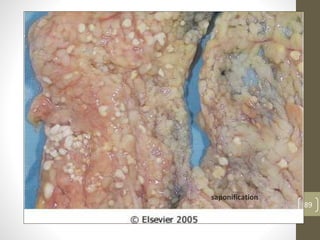
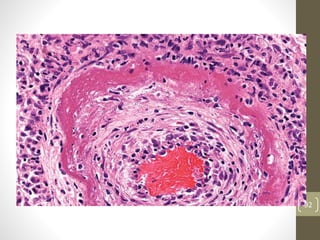
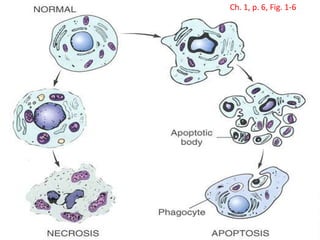
Cells are constantly adjusting to their environment and stresses to maintain homeostasis. Adaptive responses include atrophy, hypertrophy, hyperplasia and metaplasia. If stresses exceed adaptive capabilities, reversible or irreversible cell injury occurs. Causes of injury include oxygen deprivation, chemicals, infections, immune reactions, genetics, nutrition and physical agents. Injuries include ischemic, free radical and toxic injuries. Cells adapt through changes in size, number or cell type to withstand stresses or return to viability.