PANC_1_2023_03162023.pdf
•
0 likes•10 views
The document discusses recommendations from the NCCN Guidelines for Pancreatic Adenocarcinoma panel regarding the inclusion of several drugs as subsequent treatment options for pancreatic cancer. The panel voted to include adagrasib, dostarlimab-gxly, nivolumab plus ipilimumab, and sotorasib as treatment options for patients with certain mutations and performance statuses based on reviews of clinical trial data.
Report
Share
Report
Share
Download to read offline
Recommended
Chair, Amit Singal, MD, MS, Anthony El-Khoueiry, MD, Ahmed Omar Kaseb, MD, CMQ, and Anjana Pillai, MD, prepared useful Practice Aids pertaining to hepatocellular carcinoma for this CME/MOC activity titled “It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collaboration & Modern Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3AbNtC5. CME/MOC credit will be available until December 4, 2023.
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...PVI, PeerView Institute for Medical Education
Recommended
Chair, Amit Singal, MD, MS, Anthony El-Khoueiry, MD, Ahmed Omar Kaseb, MD, CMQ, and Anjana Pillai, MD, prepared useful Practice Aids pertaining to hepatocellular carcinoma for this CME/MOC activity titled “It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collaboration & Modern Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3AbNtC5. CME/MOC credit will be available until December 4, 2023.
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...PVI, PeerView Institute for Medical Education
Chair & Presenter, Robert Z. Orlowski, MD, PhD, Noa Biran, MD, and Ajay K. Nooka, MD, MPH, FACP, prepared useful Practice Aids pertaining to multiple myeloma for this CME/MOC/AAPA activity titled “The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3MPq140. CME/MOC/AAPA credit will be available until July 3, 2024.The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...PVI, PeerView Institute for Medical Education
Jorge E. Cortes, MD, and Stephen A. Strickland, MD, MSCI, prepared useful practice aids pertaining to acute myeloid leukemia for this CME activity titled "Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2u4yk5p. CME credit will be available until April 3, 2019.Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...

Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...PVI, PeerView Institute for Medical Education
Chair William K. Oh, MD, Neeraj Agarwal, MD, Matthew R. Smith, MD, PhD, and Evan Y. Yu, MD, prepared useful Practice Aids pertaining to prostate cancer for this CME activity titled "Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/3bJ932h. CME credit will be available until March 17, 2021.Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...

Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...PVI, PeerView Institute for Medical Education
Kimberly Halla, MSN, FNP-C, Paula J. Anastasia, RN, MN, AOCN, and Nelli Zafman, MSN, CRNP, AOCNP prepared useful Practice Aids pertaining to PARP inhibitor therapy for this CNE activity titled, "Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Oncology Nurses on the Advances and Challenges." For the full presentation, monograph, complete CNE information, and to apply for credit, please visit us at http://bit.ly/2EkO5Ij. CNE credit will be available until May 22, 2020.Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...

Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...PVI, PeerView Institute for Medical Education
Robert Coleman, MD, Maha H.A. Hussain, MD, FACP, FASCO, and Mark E. Robson, MD, prepared useful Practice Aids pertaining to PARP inhibitors for this CME/CNE activity titled, "Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2MWKmUN. CME/CNE credit will be available until November 7, 2020.Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...

Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...PVI, PeerView Institute for Medical Education
More Related Content
Similar to PANC_1_2023_03162023.pdf
Chair & Presenter, Robert Z. Orlowski, MD, PhD, Noa Biran, MD, and Ajay K. Nooka, MD, MPH, FACP, prepared useful Practice Aids pertaining to multiple myeloma for this CME/MOC/AAPA activity titled “The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3MPq140. CME/MOC/AAPA credit will be available until July 3, 2024.The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...PVI, PeerView Institute for Medical Education
Jorge E. Cortes, MD, and Stephen A. Strickland, MD, MSCI, prepared useful practice aids pertaining to acute myeloid leukemia for this CME activity titled "Novel Induction Options in AML: Assessing the Implications for HCT-Eligible Patients." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2u4yk5p. CME credit will be available until April 3, 2019.Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...

Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...PVI, PeerView Institute for Medical Education
Chair William K. Oh, MD, Neeraj Agarwal, MD, Matthew R. Smith, MD, PhD, and Evan Y. Yu, MD, prepared useful Practice Aids pertaining to prostate cancer for this CME activity titled "Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal Insights and Guidance From the Patient CaseBook." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/3bJ932h. CME credit will be available until March 17, 2021.Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...

Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...PVI, PeerView Institute for Medical Education
Kimberly Halla, MSN, FNP-C, Paula J. Anastasia, RN, MN, AOCN, and Nelli Zafman, MSN, CRNP, AOCNP prepared useful Practice Aids pertaining to PARP inhibitor therapy for this CNE activity titled, "Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Oncology Nurses on the Advances and Challenges." For the full presentation, monograph, complete CNE information, and to apply for credit, please visit us at http://bit.ly/2EkO5Ij. CNE credit will be available until May 22, 2020.Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...

Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...PVI, PeerView Institute for Medical Education
Robert Coleman, MD, Maha H.A. Hussain, MD, FACP, FASCO, and Mark E. Robson, MD, prepared useful Practice Aids pertaining to PARP inhibitors for this CME/CNE activity titled, "Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP Inhibitors." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2MWKmUN. CME/CNE credit will be available until November 7, 2020.Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...

Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...PVI, PeerView Institute for Medical Education
Similar to PANC_1_2023_03162023.pdf (20)
The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...
Changing Landscape of Cholangiocarcinoma_Crimson Publishers

Changing Landscape of Cholangiocarcinoma_Crimson Publishers
Improving Access to Innovative Cancer Therapies in Canada

Improving Access to Innovative Cancer Therapies in Canada
New Thinking, New Strategies in Advanced Urothelial Carcinoma

New Thinking, New Strategies in Advanced Urothelial Carcinoma
Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...

Novel Induction Options in AML: Assessing the Implications for HCT-Eligible P...
Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...

Mapping the Pathways to Better Patient Outcomes in Prostate Cancer: Personal ...
Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...

Realizing the Promise of PARP Inhibitors in Solid Tumor Therapy: Guiding Onco...
Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...

Advances in Precision Care in Solid Tumor Oncology: Defining the Role of PARP...
Recently uploaded
Genuine Call Girls Hyderabad 9630942363 Book High Profile Call Girl in Hyderabad Genuine Escort ServiceGenuine Call Girls Hyderabad 9630942363 Book High Profile Call Girl in Hydera...

Genuine Call Girls Hyderabad 9630942363 Book High Profile Call Girl in Hydera...GENUINE ESCORT AGENCY
Recently uploaded (20)
💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...

💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...
7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta

7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta
Race Course Road } Book Call Girls in Bangalore | Whatsapp No 6378878445 VIP ...

Race Course Road } Book Call Girls in Bangalore | Whatsapp No 6378878445 VIP ...
Most Beautiful Call Girl in Chennai 7427069034 Contact on WhatsApp

Most Beautiful Call Girl in Chennai 7427069034 Contact on WhatsApp
❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ ...
❤️Amritsar Escorts Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amri...

❤️Amritsar Escorts Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amri...
Call Girl In Indore 📞9235973566📞 Just📲 Call Inaaya Indore Call Girls Service ...

Call Girl In Indore 📞9235973566📞 Just📲 Call Inaaya Indore Call Girls Service ...
Bhawanipatna Call Girls 📞9332606886 Call Girls in Bhawanipatna Escorts servic...

Bhawanipatna Call Girls 📞9332606886 Call Girls in Bhawanipatna Escorts servic...
Jaipur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Jaipur No💰...

Jaipur Call Girl Service 📞9xx000xx09📞Just Call Divya📲 Call Girl In Jaipur No💰...
Kolkata Call Girls Shobhabazar 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Gir...

Kolkata Call Girls Shobhabazar 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Gir...
Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery

Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery
VIP Hyderabad Call Girls KPHB 7877925207 ₹5000 To 25K With AC Room 💚😋

VIP Hyderabad Call Girls KPHB 7877925207 ₹5000 To 25K With AC Room 💚😋
Whitefield { Call Girl in Bangalore ₹7.5k Pick Up & Drop With Cash Payment 63...

Whitefield { Call Girl in Bangalore ₹7.5k Pick Up & Drop With Cash Payment 63...
Genuine Call Girls Hyderabad 9630942363 Book High Profile Call Girl in Hydera...

Genuine Call Girls Hyderabad 9630942363 Book High Profile Call Girl in Hydera...
Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...

Gastric Cancer: Сlinical Implementation of Artificial Intelligence, Synergeti...
Call Girls Shahdol Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Shahdol Just Call 8250077686 Top Class Call Girl Service Available
PANC_1_2023_03162023.pdf
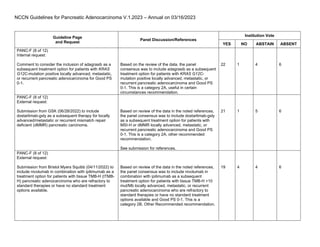
- 1. NCCN Guidelines for Pancreatic Adenocarcinoma V.1.2023 – Annual on 03/16/2023 Guideline Page and Request Panel Discussion/References Institution Vote YES NO ABSTAIN ABSENT PANC-F (8 of 12) Internal request: Comment to consider the inclusion of adagrasib as a subsequent treatment option for patients with KRAS G12C-mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma for Good PS 0-1. Based on the review of the data, the panel consensus was to include adagrasib as a subsequent treatment option for patients with KRAS G12C- mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and Good PS 0-1. This is a category 2A, useful in certain circumstances recommendation. 22 1 4 6 PANC-F (8 of 12) External request: Submission from GSK (06/28/2022) to include dostarlimab-gxly as a subsequent therapy for locally advanced/metastatic or recurrent mismatch repair deficient (dMMR) pancreatic carcinoma. Based on review of the data in the noted references, the panel consensus was to include dostarlimab-gxly as a subsequent treatment option for patients with MSI-H or dMMR locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and Good PS 0-1. This is a category 2A, other recommended recommendation. See submission for references. 21 1 5 6 PANC-F (8 of 12) External request: Submission from Bristol Myers Squibb (04/11/2022) to include nivolumab in combination with ipilimumab as a treatment option for patients with tissue TMB-H (tTMB- H) pancreatic adenocarcinoma who are refractory to standard therapies or have no standard treatment options available. Based on review of the data in the noted references, the panel consensus was to include nivolumab in combination with ipilimumab as a subsequent treatment option for patients with tissue TMB-H >10 mut/Mb locally advanced, metastatic, or recurrent pancreatic adenocarcinoma who are refractory to standard therapies or have no standard treatment options available and Good PS 0-1. This is a category 2B, Other Recommended recommendation. 19 4 4 6
- 2. NCCN Guidelines for Pancreatic Adenocarcinoma V.1.2023 – Annual on 03/16/2023 PANC-F (8 of 12) External request: Submission from Amgen, Inc. (02/17/2022) to include sotorasib monotherapy for treatment of patients with metastatic KRAS G12C-mutated pancreatic cancer who have received prior therapy. Based on the review of the data in the noted references, the panel consensus was to include sotorasib as a subsequent treatment option for patients with KRAS G12C-mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and Good PS 0-1. This is a category 2A, useful in certain circumstances recommendation. See submission for references. 23 0 4 6 PANC-F (9 of 12) Internal request: Comment to consider the inclusion of adagrasib as a subsequent treatment option for patients with KRAS G12C-mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma for Intermediate PS 2 and Poor PS 3-4. Based on the review of the data, the panel consensus was to include adagrasib as a subsequent treatment option for patients with KRAS G12C- mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and: 1. Intermediate PS 2. This is a category 2A, useful in certain circumstances recommendation. 2. Poor PS 3-4. This is a category 2B, useful in certain circumstances recommendation. 20 12 2 9 5 6 6 6 PANC-F (9 of 12) External request: Submission from GSK (06/28/2022) to include dostarlimab-gxly as a subsequent therapy for locally advanced/metastatic or recurrent mismatch repair deficient (dMMR) pancreatic carcinoma. Based on review of the data in the noted references, the panel consensus was to include dostarlimab-gxly as a subsequent treatment option for patients with MSI-H or dMMR locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and: 1. Intermediate PS 2. This is a category 2A, useful in certain circumstances recommendation. 2. Poor PS 3-4. This is a category 2B, preferred regimen recommendation. See submission for references. 20 12 2 9 5 6 6 6
- 3. NCCN Guidelines for Pancreatic Adenocarcinoma V.1.2023 – Annual on 03/16/2023 PANC-F (9 of 12) External request: Submission from Bristol Myers Squibb (04/11/2022) to include nivolumab in combination with ipilimumab as a treatment option for patients with tissue TMB-H (tTMB- H) pancreatic adenocarcinoma who are refractory to standard therapies or have no standard treatment options available. Based on review of the data in the noted references, the panel consensus was to include nivolumab in combination with ipilimumab as a subsequent treatment option for patients with tissue TMB-H >10 mut/Mb locally advanced, metastatic, or recurrent pancreatic adenocarcinoma who are refractory to standard therapies or have no standard treatment options available and Intermediate PS 2. This is a category 2B, useful in certain circumstances recommendation. 18 5 4 6 PANC-F (9 of 12) External request: Submission from Amgen, Inc. (02/17/2022) to include sotorasib monotherapy for treatment of patients with metastatic KRAS G12C-mutated pancreatic cancer who have received prior therapy. Based on the review of the data in the noted references, the panel consensus was to include sotorasib as a subsequent treatment option for patients with KRAS G12C-mutation positive locally advanced, metastatic, or recurrent pancreatic adenocarcinoma and: 1. Intermediate PS 2. This is a category 2A, useful in certain circumstances recommendation. 2. Poor PS 3-4. This is a category 2B, useful in certain circumstances recommendation. 22 15 0 7 5 5 6 6
