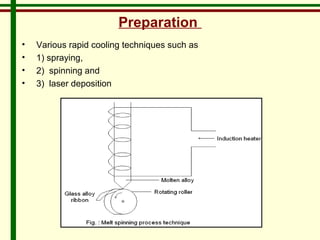
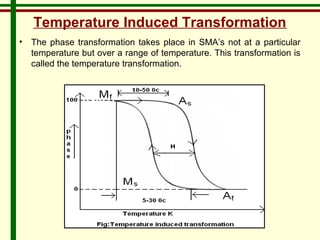
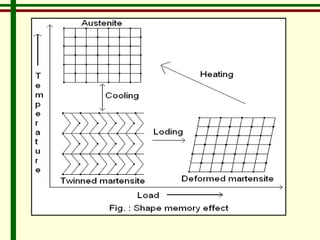
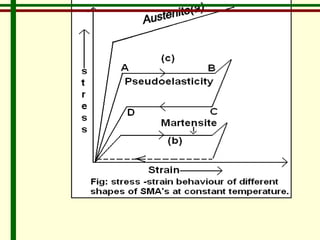
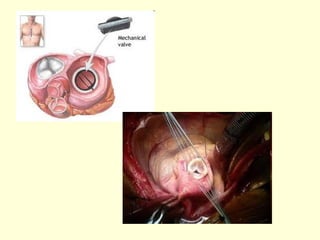
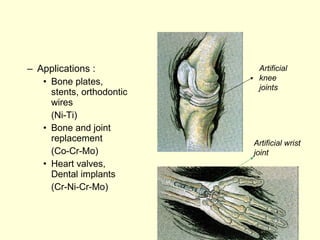
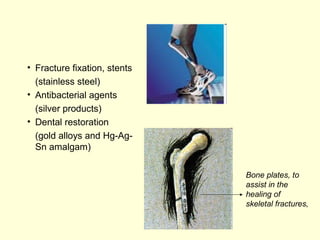
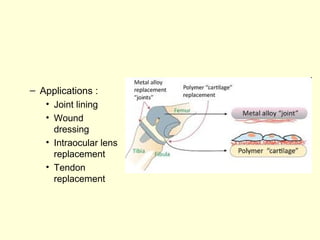
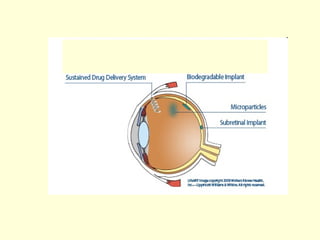
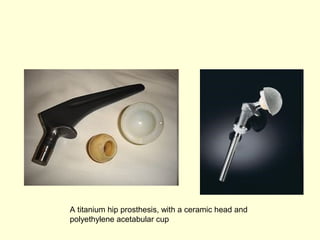
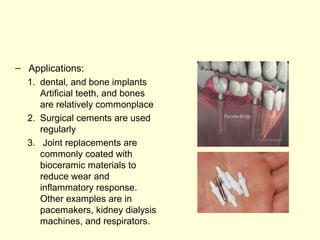
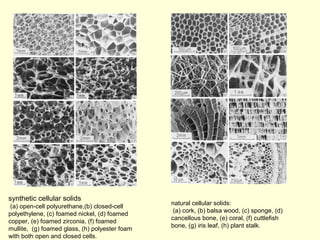
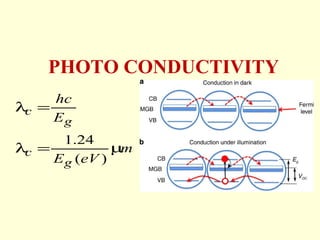
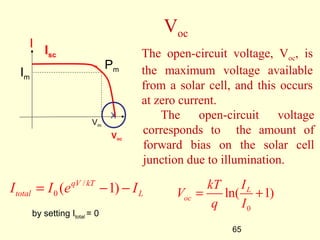
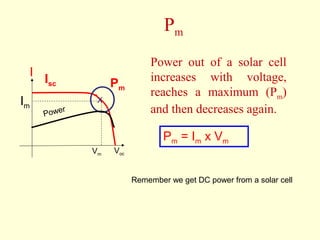
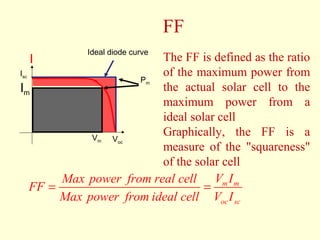
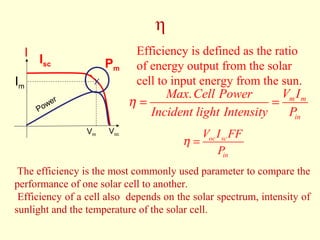
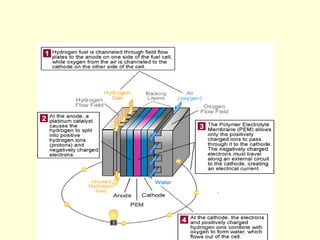
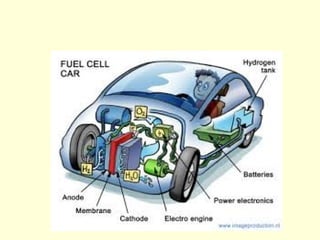
The document discusses new engineering materials called metallic glasses. Metallic glasses are metal alloys that are non-crystalline, or amorphous, meaning they lack long-range atomic order. They can be produced by rapidly cooling molten metals to rates of over 2 million degrees Celsius per second. This prevents crystal nucleation and results in a glassy solid. Metallic glasses have advantages like strength and corrosion resistance. Applications include use in transformers, sensors, and as surgical instruments. Shape memory alloys that can revert to their original shape after deformation are also discussed.