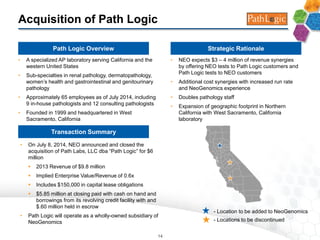
This company presentation provides an overview of a cancer genetic testing services company. Key points include:
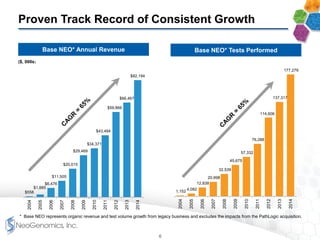
- The company has experienced strong and consistent growth in revenue and test volume over the past 10 years through both organic growth and acquisitions.
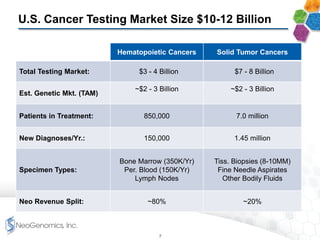
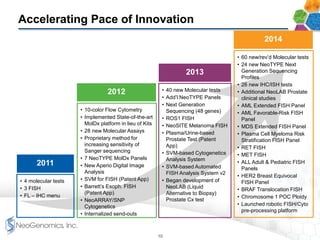
- It provides a broad menu of genetic and molecular testing services including next generation sequencing and is developing liquid biopsy tests.
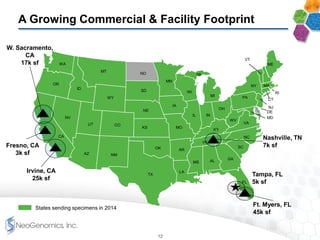
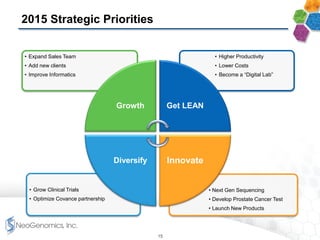
- The company has an experienced management team with experience in large reference laboratories. It is focused on innovation, cost management, and expanding its commercial footprint.
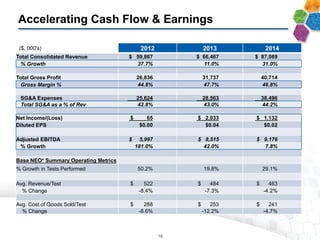
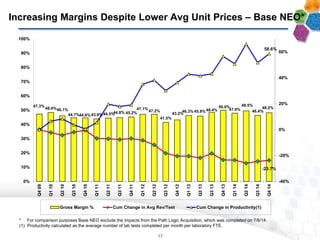
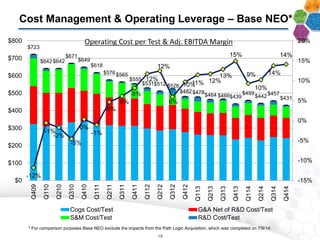
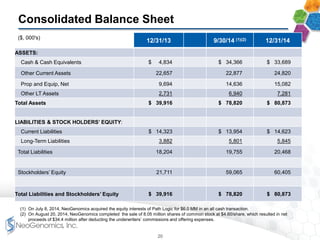
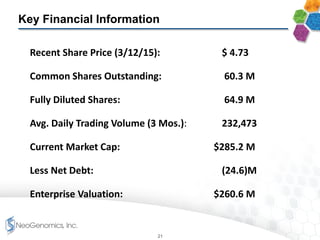
- Financial results show increasing margins, productivity, and adjusted EBITDA despite lower average prices per test, demonstrating strong operating leverage as the business scales.