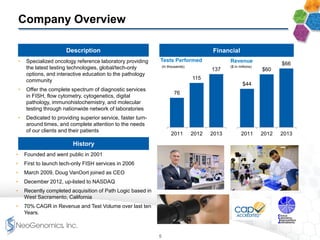
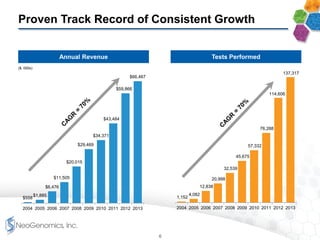
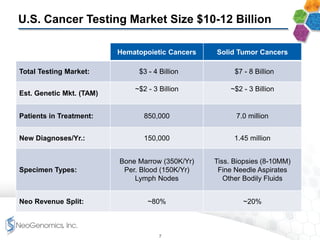
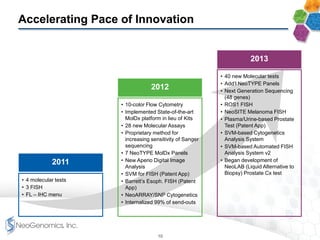
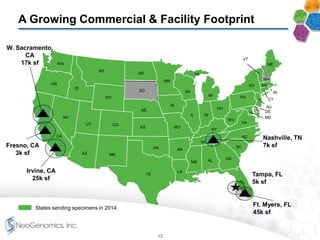
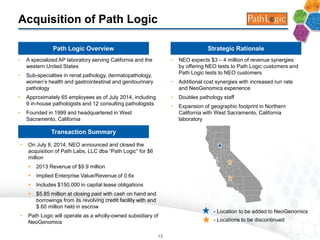
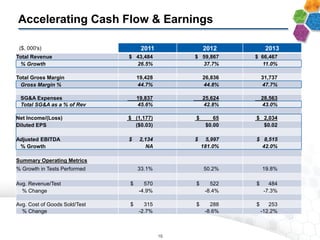
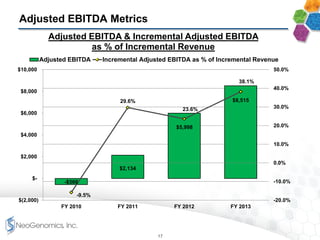
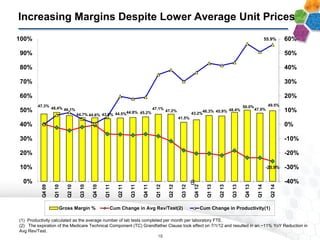
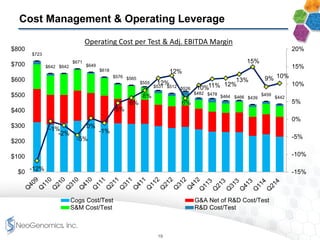
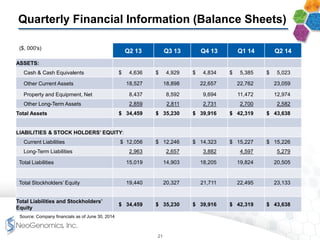
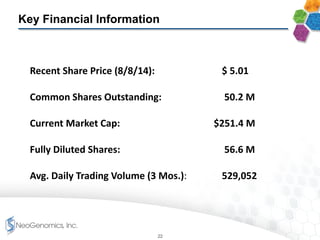
The document provides an overview of a fast-growing cancer genetics laboratory that focuses on advanced genetic testing services for oncologists, pathologists, and hospitals. It emphasizes the company’s significant revenue growth, innovative testing methodologies, and strategic acquisitions aimed at enhancing service offerings and market reach. The presentation also highlights a highly experienced management team and outlines financial performance metrics, anticipating ongoing growth and expansion in the cancer testing market.