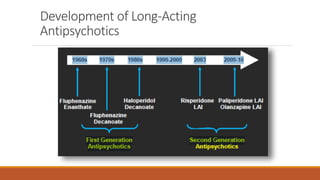
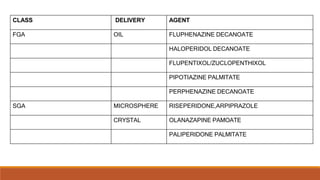
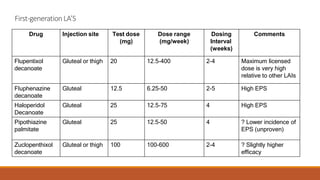
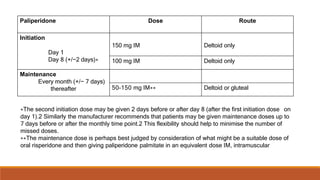
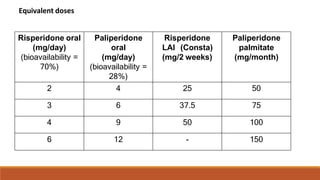
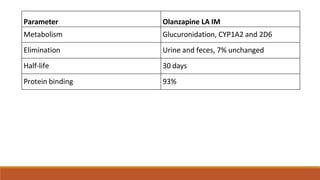
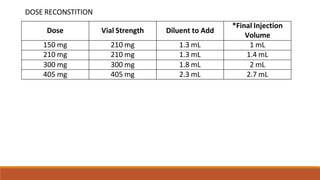
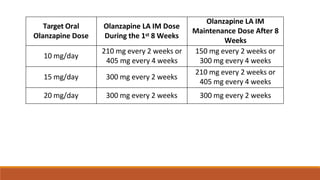
Long Acting Antipsychotics can be administered as injectable compounds that are released slowly over weeks. This helps improve compliance and reduces relapse rates compared to short acting oral medications. There are first generation LAIs based on esters of older antipsychotics combined with fatty acids, and second generation LAIs including risperidone microspheres. LAIs provide consistent drug levels and avoid issues with non-adherence to oral medications. They require less frequent dosing than tablets and have benefits for patients with schizophrenia or other psychoses.