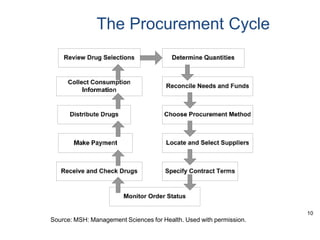
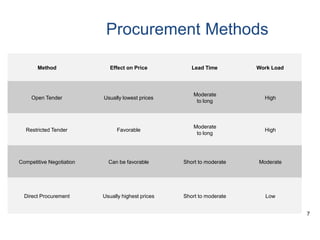
This document discusses best practices for pharmaceutical procurement. It outlines key objectives of an effective procurement program such as selecting cost-effective essential drugs, quantifying needs, procuring the right quantities at low cost from reliable suppliers, and monitoring supplier performance. Good practices include using generic names, bulk purchasing, competitive bidding, quality assurance programs, and transparency. The procurement cycle and various procurement methods are described along with advantages of pooled procurement and common challenges faced.