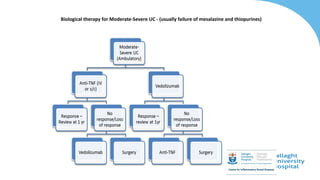
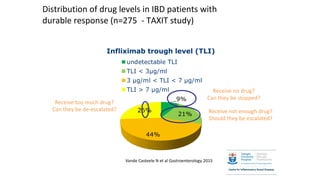
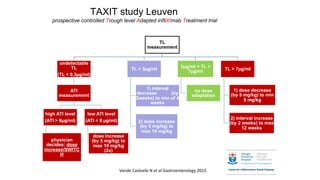
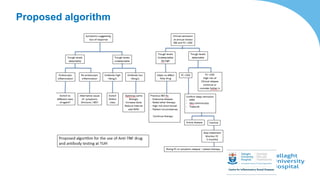
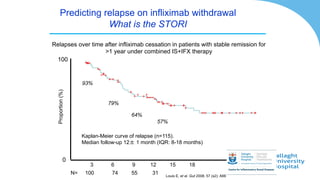
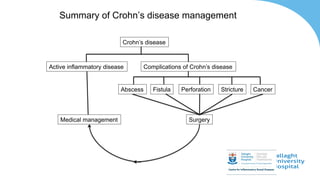
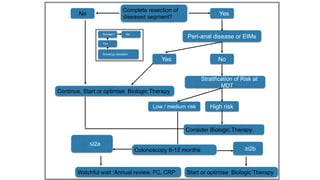
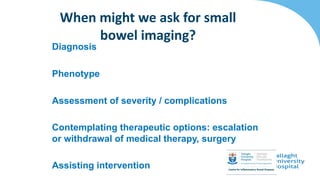
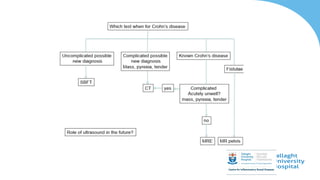
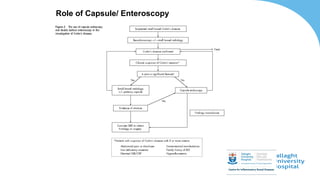
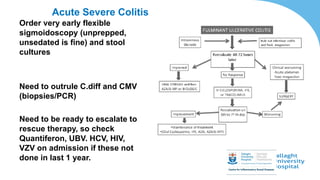
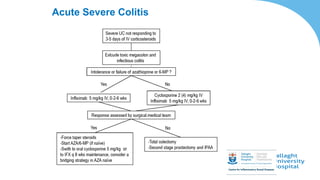
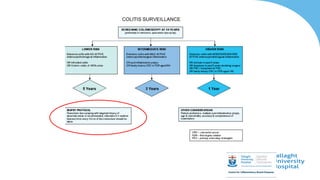
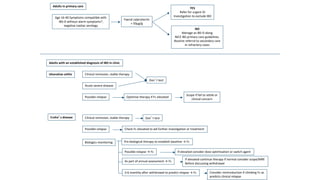
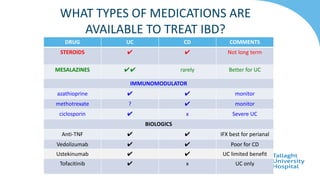
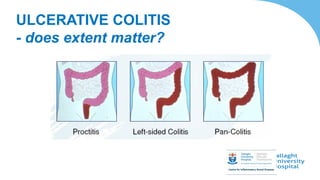
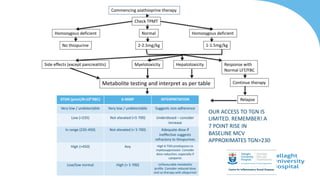
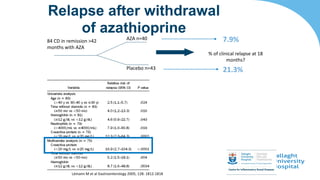
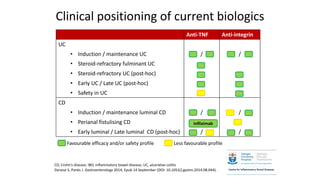
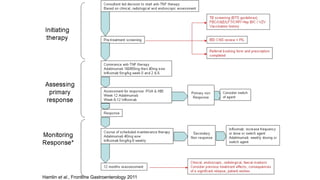
This document discusses the management of inflammatory bowel disease (IBD) including monitoring disease activity, initiating and monitoring therapies, and practical considerations. It provides guidelines on using fecal calprotectin to monitor ulcerative colitis and Crohn's disease. It also summarizes the types of medications available to treat IBD including mesalazines, immunomodulators, biologics, and their effectiveness and monitoring. Trial results are presented comparing medications for inducing remission and preventing relapse in IBD.


















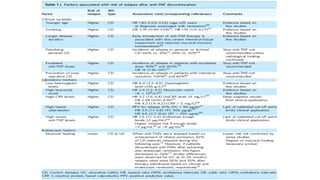
![TRIAL DESIGN OUTCOMES COMMENTS
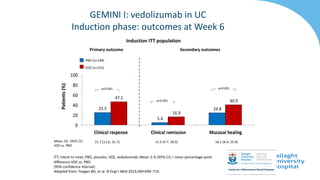
GEMINI I
Feagan et al., 2013 NEJM
Cohort 1 374 pts 300mg v
placebo IV wk 0,2
Cohort 2: open label wk 0,2
assess wk 6
RESPONSE WEEK 6
47.1% V 25.5%
P<0.001
Cohort 2 open label therefore
bias
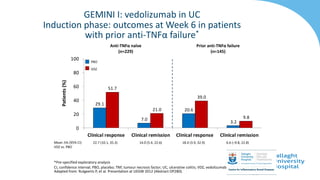
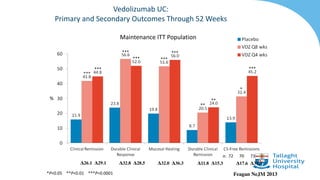
GEMINI 1 maintenance
Feagan et al., 2013 NEJM
Responders from cohorts 1 or 2
randomised to placebo / 4
weekly or 8 weekly Vedo for 52
weeks
REMISSION WK 52
15.9% v 44.8% v 41.8%
P<0.001
Only randomise responders
therefore biased (but real life!)
NNT=4 for both schedules
Parikh et al., IBD 2013 After placebo controlled study
38 UC – 2,6 or 10mg/kg at
1,15,43 days then q8. 34 rx
niave (15 UC, 19CD) same
regimen
UC
21/53 (39%) response
38/53 (58%) remission
Safe
Improved IBDQ
Parikh et al., IBD 2012 Dose ranging RCT
2,6 or 10 mg/kg or placebo days
1,15,29 and 85
[drug], safety etc
Response (Mayo), FC
Well tolerated,safe
VEDOLIZUMAB TRIAL DATA IN UC](https://image.slidesharecdn.com/ibdfinalmedsprofanthonyoconnor-230219121905-89056b5e/85/Inflammatory-Bowel-Disease-29-320.jpg)