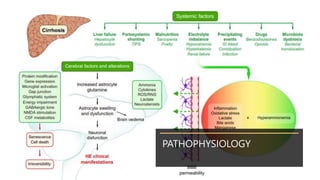
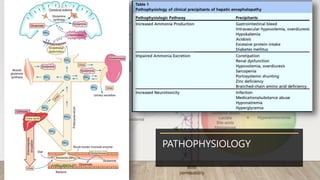
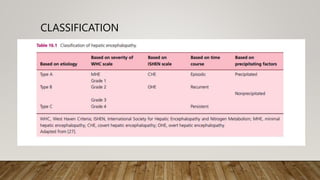
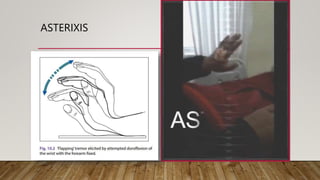
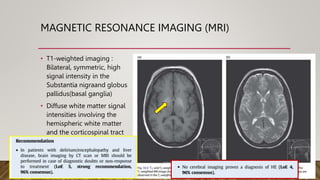
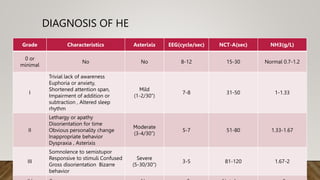
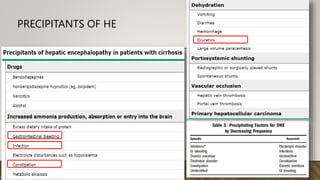
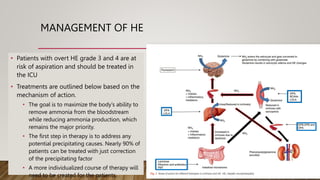
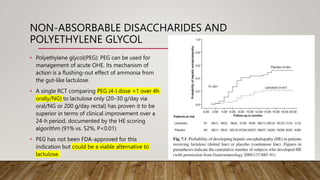
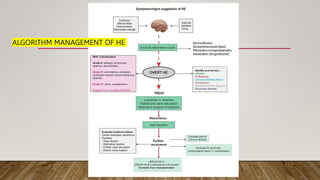
Hepatic encephalopathy (HE) is a brain dysfunction caused by liver insufficiency. It is characterized by alterations in personality, consciousness, cognition and motor function. The document discusses the definition, epidemiology, pathophysiology, classification, diagnosis and treatment of HE. For treatment, non-absorbable disaccharides, antibiotics, L-ornithine L-aspartate, branched-chain amino acids and other novel drugs are used to reduce ammonia levels and symptoms of HE.