GSIPA2M, Parallel session 2, Making compulsory licenses routine - Nimit Tienudom
•Download as PPTX, PDF•
0 likes•56 views
Thailand achieved several key milestones in expanding access to HIV/AIDS treatment between 1999-2010: 1) In 1999, civil society called for compulsory licensing of ddI, starting Thailand down the path of using compulsory licensing to increase access to antiretrovirals. 2) Between 2000-2005 the government committed to including antiretrovirals in the Universal Coverage Scheme and began local production of generic antiretrovirals. 3) By October 2005, antiretroviral therapy was officially included in the Universal Coverage Scheme, marking a major milestone in achieving universal treatment access in Thailand.
Report
Share
Report
Share
Recommended
Recommended
Patent Oppositions: Perspective from the Patent Offices.
Presented by Menna-t-allah M. El Kotamy.GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...

GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...MakeMedicinesAffordable
More Related Content
More from MakeMedicinesAffordable
Patent Oppositions: Perspective from the Patent Offices.
Presented by Menna-t-allah M. El Kotamy.GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...

GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...MakeMedicinesAffordable
More from MakeMedicinesAffordable (20)
GSIPA2M, Roundtable 4, Challenging unmerited patents - Tahir Amin

GSIPA2M, Roundtable 4, Challenging unmerited patents - Tahir Amin
GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...

GSIPA2M, Roundtable 4, Challenging unmerited patents - Menna-t-allah M. El Ko...
GSIPA2M, Roundtable 4, Challenging unmerited patents - Georgina Gerde

GSIPA2M, Roundtable 4, Challenging unmerited patents - Georgina Gerde
GSIPA2M, Roundtable 4, Challenging unmerited patents - Jose Maria di Bello

GSIPA2M, Roundtable 4, Challenging unmerited patents - Jose Maria di Bello
GSIPA2M, Roundtable 4, Challenging unmerited patents - Sergey Kondratyuk

GSIPA2M, Roundtable 4, Challenging unmerited patents - Sergey Kondratyuk
GSIPA2M, Roundtable 3, Pro-health law reform - Judit Rius Sanjuan

GSIPA2M, Roundtable 3, Pro-health law reform - Judit Rius Sanjuan
GSIPA2M, Rountable 3, Pro-health law reform - Oksana Kashyntseva

GSIPA2M, Rountable 3, Pro-health law reform - Oksana Kashyntseva
GSIPA2M, Roundtable 2, Local production - Padmashree Gehl Sampath

GSIPA2M, Roundtable 2, Local production - Padmashree Gehl Sampath
GSIPA2M, Roundtable 2, Local production - Jorge Bermudez

GSIPA2M, Roundtable 2, Local production - Jorge Bermudez
GSIPA2M, Roundtable 2, Local production - Mia Lahlou Filali 

GSIPA2M, Roundtable 2, Local production - Mia Lahlou Filali
GSIPA2M, Roundtable 1, TRIPS-minus - Yunus Mohammed

GSIPA2M, Roundtable 1, TRIPS-minus - Yunus Mohammed
GSIPA2M, Roundtable 1, TRIPS-minus - Chikosa Banda

GSIPA2M, Roundtable 1, TRIPS-minus - Chikosa Banda
GSIPA2M, Roundtable 1, TRIPS-minus - Henry Zakumumpa

GSIPA2M, Roundtable 1, TRIPS-minus - Henry Zakumumpa
GSIPA2M, Plenary 3, Leaving TRIPS behind - Mohammed El Said

GSIPA2M, Plenary 3, Leaving TRIPS behind - Mohammed El Said
GSIPA2M, Plenary 3, Leaving TRIPS behind - Sergey Kondratyuk

GSIPA2M, Plenary 3, Leaving TRIPS behind - Sergey Kondratyuk
GSIPA2M, Plenary 3, Leaving TRIPS behind - Oksana Kashyntseva

GSIPA2M, Plenary 3, Leaving TRIPS behind - Oksana Kashyntseva
GSIPA2M, Plenary 3, Leaving TRIPS behind - Marcela Vieira

GSIPA2M, Plenary 3, Leaving TRIPS behind - Marcela Vieira
GSIPA2M, Plenary 3, Leaving TRIPS behind - Lorena di Giano

GSIPA2M, Plenary 3, Leaving TRIPS behind - Lorena di Giano
GSIPA2M, Parallel session 2, Making compulsory licenses routine - James Love

GSIPA2M, Parallel session 2, Making compulsory licenses routine - James Love
Recently uploaded
Recently uploaded (20)
Advancing Impact Measurement | Public Good App House

Advancing Impact Measurement | Public Good App House
sarkarijobswork.online-bsf-si-vehicle-mechanic-and-constable-technical-may24.pdf

sarkarijobswork.online-bsf-si-vehicle-mechanic-and-constable-technical-may24.pdf
#Bepartoftheplan on International Day For Biological Diversity 2024

#Bepartoftheplan on International Day For Biological Diversity 2024
Low Atmospheric Pressure Stunning is not a humane alternative to Carbon Dioxi...

Low Atmospheric Pressure Stunning is not a humane alternative to Carbon Dioxi...
Ghana High Commission on list of diplomats including US & China who owe £143m...

Ghana High Commission on list of diplomats including US & China who owe £143m...
“Be part of the Plan” International Day For Biological Diversity 2024.

“Be part of the Plan” International Day For Biological Diversity 2024.
Proposed Facility Types: Chesapeake Trails and Connectivity Plan

Proposed Facility Types: Chesapeake Trails and Connectivity Plan
GSIPA2M, Parallel session 2, Making compulsory licenses routine - Nimit Tienudom
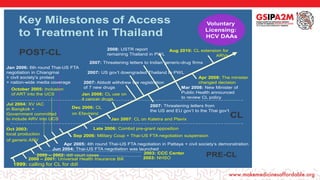
- 1. Key Milestones of Access to Treatment in Thailand 1999: calling for CL for ddI 2000 – 2001: Universal Health Insurance Bill 2000 – 2002: ddI court cases Oct 2003: local production of generic ARV Jun 2004: Thai-US FTA negotiation was launched Jul 2004: XV IAC in Bangkok + Government committed to include ARV into UCS Apr 2005: 4th round Thai-US FTA negotiation in Pattaya + civil society’s demonstration October 2005: Inclusion of ART into the UCS Jan 2006: 6th round Thai-US FTA negotiation in Chiangmai + civil society’s protest + nation-wide media coverage Sep 2006: Military Coup + Thai-US FTA negotiation suspension Dec 2006: CL on Efavirenz Late 2006: Combid pre-grant opposition Jan 2007: CL on Kaletra and Plavix Jan 2008: CL use on 4 cancer drugs Apr 2008: The minister changed decision Mar 2008: New Minister of Public Health announced to review CL policy Aug 2010: CL extension for ARVs 2007: Abbott withdrew the registration of 7 new drugs 2007: US gov’t downgraded Thailand to PWL 2007: Threatening letters from the US and EU gov’t to the Thai gov’t 2007: Threatening letters to Indian generic-drug firms 2008: USTR report remaining Thailand in PWL PRE-CL POST-CL CL Voluntary Licensing: HCV DAAs 2003: CCC Center 2003: NHSO