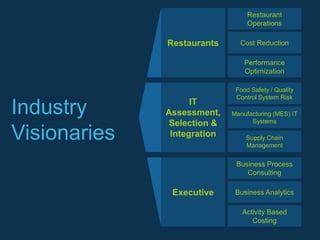
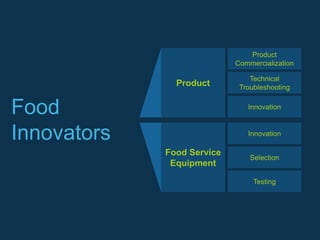
Food Technical Consulting (FTC) specializes in innovative food and beverage technology solutions that enhance clients' competitive edge through various consulting services, including operational performance optimization, product commercialization, and food safety systems. They offer expertise in manufacturing, supply chain management, and technical troubleshooting, along with workshops and seminars on food innovation and quality control. FTC aims to push the boundaries of the food industry by supporting clients in new product development and compliance with industry standards.