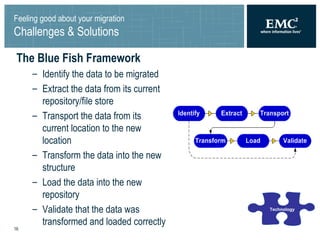
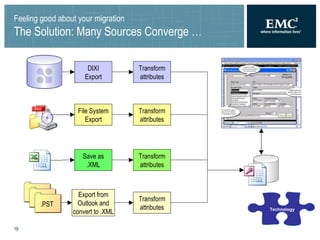
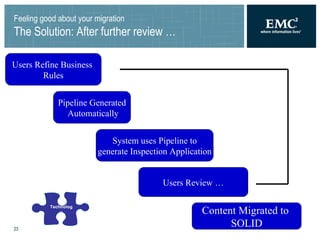
The document summarizes a framework for performing validated Documentum migrations that was used by Solvay Pharmaceuticals and Blue Fish Development Group. It describes challenges faced such as organizational issues, project management difficulties, technological hurdles, and quality assurance problems. These were addressed through solutions like forming a global migration team, precisely defining scope, using common tools for validation, and allowing business users to validate documents. The end result was a successful migration completed on time and within budget that satisfied users and found no issues during the transition to the new system.