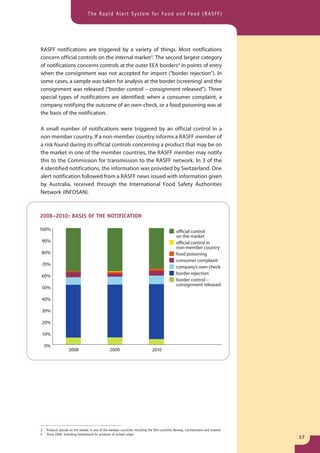
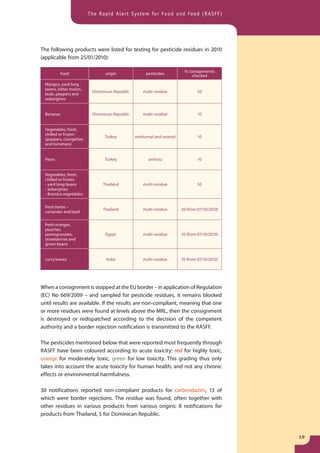
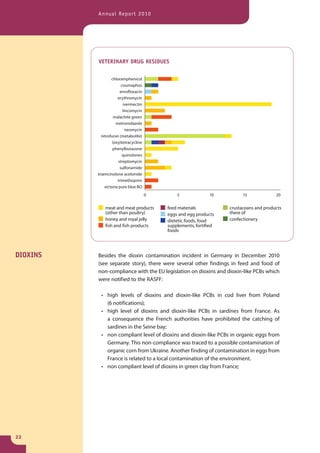
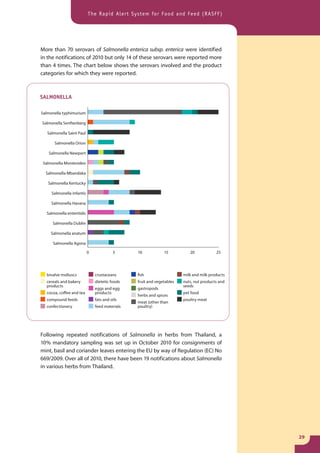
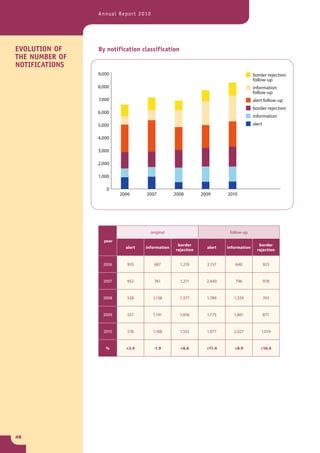
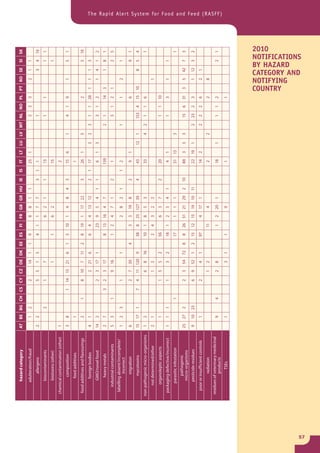
The document provides information about the Rapid Alert System for Food and Feed (RASFF). It describes the legal basis, members, and system of RASFF. Key points include:
- RASFF was established to facilitate the rapid exchange of information between EU countries about serious risks detected in food and feed.
- The legal basis is Regulation (EC) No 178/2002, which requires EU countries to notify the Commission of measures taken in response to food/feed risks.
- Members include EU countries, EFTA countries, the European Commission, and the European Food Safety Authority.
- The system facilitates quick action in response to food/feed hazards through a network of national contact points that