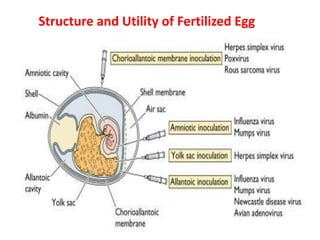
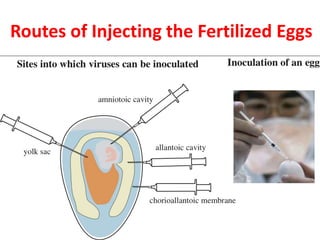
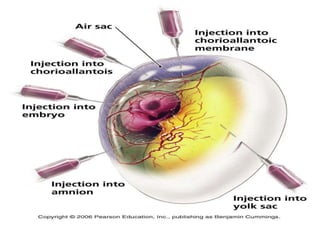
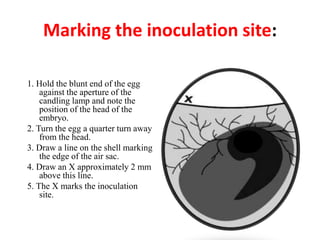
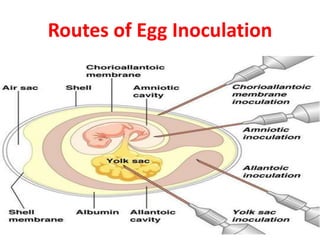
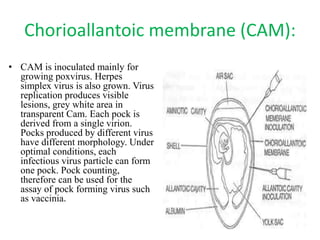
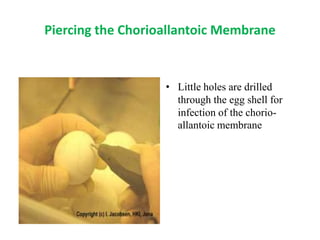
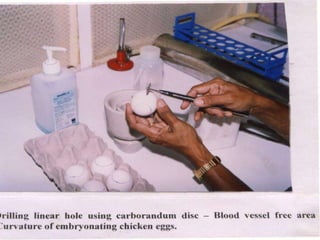
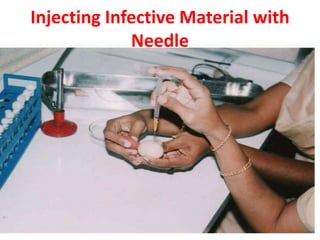
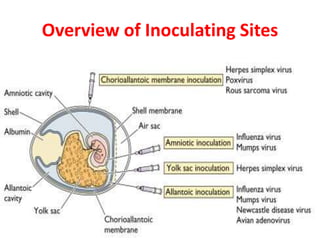
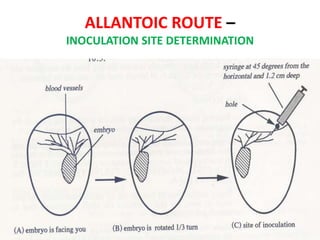
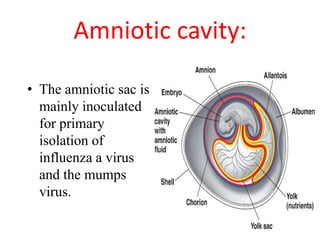
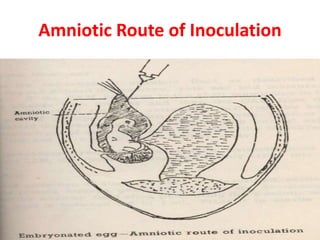
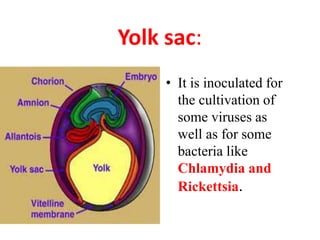
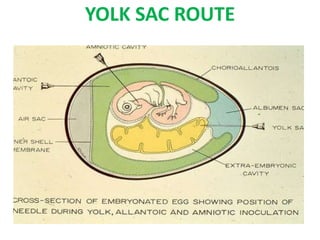
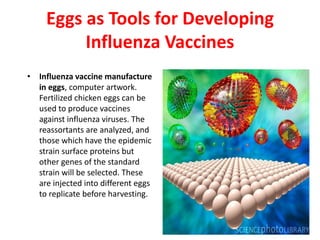
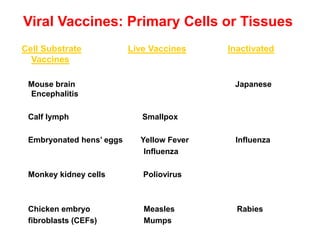
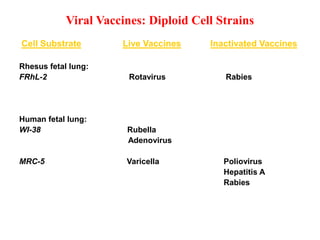
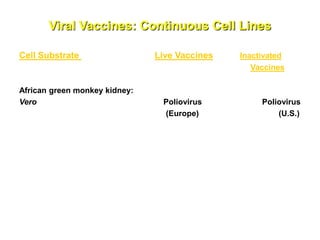
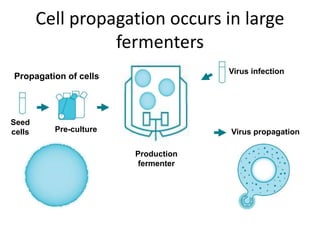
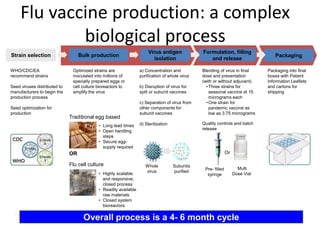
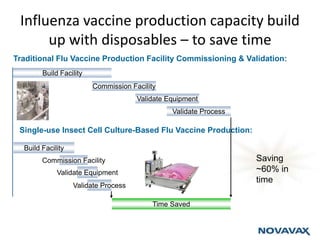
The document outlines the cultivation of viruses, particularly focusing on methods using embryonated hens' eggs and cell culture. It details the advantages of using embryonated eggs for virus isolation and vaccine production, as well as the procedures for inoculation and incubation. Additionally, it discusses alternative cell culture methods for vaccine production and highlights challenges and considerations associated with both egg-based and cell-based systems.