This document provides an overview of diabetes mellitus (DM), including:
1) It defines DM as a metabolic disorder characterized by chronic hyperglycemia resulting from defects in insulin secretion or action.
2) It classifies the main types of DM as type 1, type 2, and gestational diabetes and discusses their characteristics.
3) It discusses the etiology, pathophysiology, clinical manifestations, diagnosis, and management of type 1 and type 2 DM.
















![Diabetes Mellitus (DM)
The term diabetes mellitus describes a metabolic
disorder of multiple etiology, characterized by chronic
hyperglycemia with disturbances of carbohydrate, fat
and protein metabolism resulting from an imbalance
b/w insulin availability and insulin need.
OR
Diabetes mellitus is a group of metabolic diseases
characterized by elevated levels of glucose in the
blood (hyperglycemia) resulting from defects in
insulin secretion, insulin action, or both (American
Diabetes Association [ADA],](https://image.slidesharecdn.com/dm-240102074149-5ec50102/85/DM-pptx-19-320.jpg)


























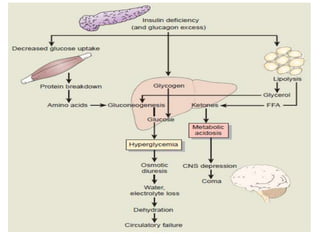
![Diabetic Ketoacidosis (DKA)
The three major metabolic derangements in DKA are
Hyperglycemia, (blood glucose levels >250 mg dL [13.8
mmol/L])
ketosis, [ketonemia and moderate Ketonuria] and
Metabolic acidosis. (low serum bicarbonate (<15 mEq/L [15
mmol/L]), and low pH <7.3).](https://image.slidesharecdn.com/dm-240102074149-5ec50102/85/DM-pptx-47-320.jpg)



![Hyperosmolar Hyperglycemic State
HHS is characterized by hyperglycemia (blood glucose
>600 mg/dL [33.3 mmol/L]), hyperosmolarity (plasma
osmolarity >320 mOsm/L) and dehydration, the absence
of ketoacidosis, and depression of the sensorium.
HHS may occur in various conditions, including type 2
diabetes, acute pancreatitis, severe infection, myocardial
infarction, and treatment with oral or parenteral nutrition
solutions.
It is seen most frequently in people with type 2 diabetes.](https://image.slidesharecdn.com/dm-240102074149-5ec50102/85/DM-pptx-52-320.jpg)












































