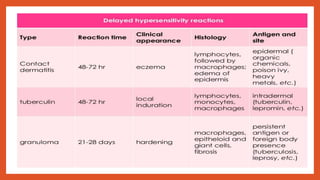
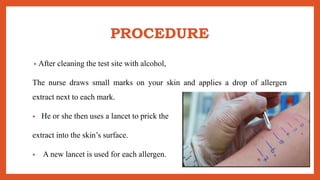
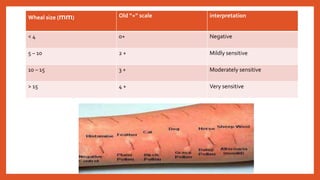
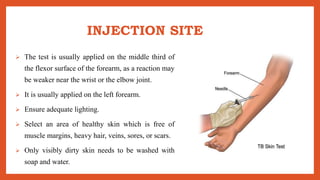
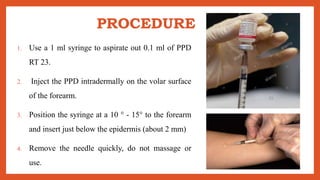
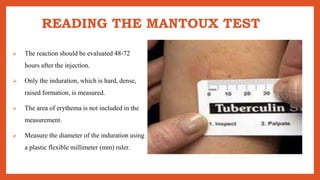
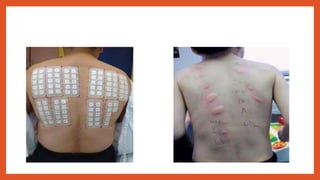
Cell-mediated immunity involves activation of phagocytes, cytotoxic T-lymphocytes, and cytokines in response to antigens, rather than antibodies. Effector cells include cytotoxic T cells, NK cells, and those performing ADCC. Cell-mediated immunity can be assessed using tests such as mixed lymphocyte reaction, cell-mediated lympholysis, and graft versus host. Delayed-type hypersensitivity is a type of hypersensitivity reaction mediated by T cells rather than antibodies, and can be assessed using skin tests such as Mantoux, patch, and intradermal tests. These tests measure induration resulting from local cytokine and chemokine production.