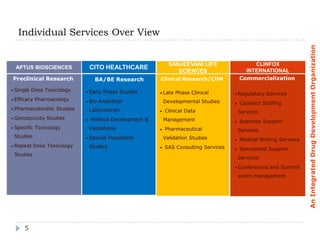
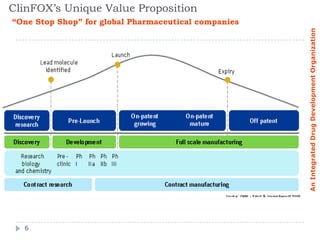
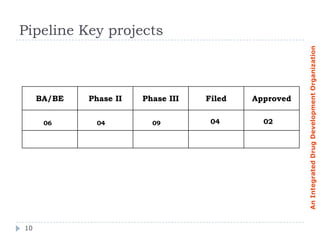
ClinFOX International is an integrated drug development organization comprising several subsidiary companies that specialize in different areas of the drug development process. The management team has extensive experience in the industry. ClinFOX uses a Split Business Technique to divide its operations into five collaborative but independently operated units covering preclinical research, bioanalytical studies, clinical research, commercialization, and business support services. This allows ClinFOX to offer clients a one-stop-shop for their drug development needs from preclinical through commercialization.