Eco-Friendly Ball Milling for Organic Synthesis
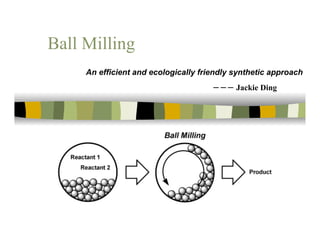
- 1. Ball Milling An efficient and ecologically friendly synthetic approach ‐‐‐ Jackie Ding
- 2. Content Ball milling – solvent free approach Application in organic synthesis Conclusion 2
- 3. The 12 Principles of Green Chemistry Prevention of waste Reduce Derivatives Atom Economy Use Renewable Feedstocks Less Hazardous Chemical Catalysis Syntheses Design for Degradation Design Safer Chemicals Real-time Analysis for Safer Solvents and Pollution Prevention Auxiliaries Inherently Safer Chemistry Design for Energy Efficiency for Accident Prevention 3
- 4. Do I need to use a solvent? 4
- 5. The best solvent is no solvent Alternative solvents: water supercritical CO2 ionic liquids Solventless Chemistry Mortar and pestle Ball milling 5
- 6. Content Principles of green chemistry Application in organic synthesis Conclusion 6
- 7. What is ball milling? A ball mill is a type of grinder used to grind materials into extremely fine powder. 7
- 8. Major parameters for ball milling Temperature Size and Number of the balls Nature of the balls Rotation speed 8
- 9. Types of Ball Mills Drum ball mills Jet-mills Bead-mills Horizontal rotary ball mills Vibration ball mills Planetary ball mills 9
- 10. Vibration Mills 10
- 11. Mixer Mill MM 400 Feed material: hard, medium-hard, soft, brittle, elastic, fibrous Material feed size: ≤ 8 mm Final fineness: ~ 5 µm Setting of vibrational frequency: digital, 3 - 30 Hz (180 - 1800 min-1) 11
- 12. Planetary Mills 12
- 13. Planetary Ball Mill PM 400 Feed material: soft, hard, brittle, fibrous (dry or wet); Material feed size: < 10 mm; Final fineness < 1 µm; No. of grinding stations: 4 / 2 13
- 14. Content Principles of green chemistry Ball milling – solvent free approach Conclusion 14
- 15. Application in Organic Synthesis Aldol Condensations Knoevenagel Condensations Baylis-Hillman Reaction Michael Reaction Wittig Reaction Fullerene Cycloadditions Suzuki Reaction Heck-Jeffery Reaction Radical Additions Mediated by Mn (III) Etc. 15
- 16. Suzuki reaction [Pd(PPh3)4] (5mol%) K2CO3!(3 equivs.) B(OH)2 + Br-Ar Ar NaCl ball milling 30-60 min Up to 96% yield S. F. Nielsen, O. Axelsson, Synth. Commun. 2000, 30, 3501. Br KF-Al2O3/ Pd(OAc)2 + Ar Ac ball milling B(OH)2 Ac Entry Rpm T (min) Yield% 1 400 10 92 2 800 5 94 Franziska Schneider, Org. Proc. Res. & Develop., 2009, 13,44 16
- 17. Aldol Condensation O O O OH (S)-proline H 10 mol% + R R Ball milling (A) or stirring (B) Entry R= Method t/h Yield% anti/syn ee% 1 4-NO2 A 5.5 99 89:11 94 2 4-NO2 B 24 95 89:11 94 3 3-NO2 A 7 94 88:12 >99 4 3-NO2 B 16 89 82:18 98 5 2-NO2 A 7 97 93:7 97 6 2-NO2 B 36 89 91:9 97 B. Rodr´ıguez, Angew. Chem., Int. Ed., 2006, 45, 6924 17
- 18. Oxidation O O O R2 N N O R2 R1 + Mn(OAc)3.2H2O ball milling O R1 O X X 30Hz, 1h R1= CH3 X=CH or N R1 R2 = -C6H5, -C6H5NO2, -C6H5CN, etc. 18 examples R1 up to 91% yield Conventional heating in organic solvent: a. Acetic acid, 80oC, argon atmosphere, yield 32% b. Ethanol, refluxing temperature for 12h, yield 48% Guan-Wu Wang, J. Org. Chem. 73, 7088 (2008). 18
- 19. Other applications of ball milling Fine particles Inorganic solids 19
- 20. Drawbacks of ball milling Purification of byproducts • Solution Benign Solvents It is not a magic • Solution Under development wand. 20
- 21. Conclusion Ball milling should be considered as a potentially attractive solution for solvent-free synthesis. One-pot Tip of Solvent free process iceberg 21
- 22. Reference Lance Frazer, Environmental health perspectives, 2003, 111, 10 Belen Rodríguez, Adv. Synth. Catal. 2007, 349, 2213 Ana Lazuen Garay, Chem. Soc. Rev., 2007, 36, 846–855 Raphael Janot, Progress in Materials Science 50 (2005) 1–92 Andreas Theisen, “Green Chemistry”in the Laboratory, TECHNICAL REPORT, www.retsch.com G Kaupp, Solvent-free organic synthesis, Wiley-VCH, 2009. S. F. Nielsen, O. Axelsson, Synth. Commun. 2000, 30, 3501. Franziska Schneider, Org. Proc. Res. & Develop., 2009, 13,44 B. Rodr´ıguez, Angew. Chem., Int. Ed., 2006, 45, 6924 Guan-Wu Wang, J. Org. Chem. 73, 7088 (2008). G.Kaupp, Top. Curr. Chem. 254, 95 (2005). M. Reza Naimi-Jamal, Eur. J. Org. Chem. 2009, 3567–3572. 22
- 23. 23