2013 WCU's Poster Presentation(ModifiedII)
•Download as PPT, PDF•
1 like•719 views
Report
Share
Report
Share
Recommended
Hydrothermal Assisted Microwave Pyrolysis of Water Hyacinth for Electrochemic...

We develop ‘green’ approach to prepare conductive carbon material from water hyacinth (Eichhornia crassipes) powder for use in electrochemical capacitor device. The features on morphology, crystallography and surface functionality were analyzed based on SEM, XRD and FTIR instrumentation, respectively. The electrical conductivities were measured using four-point probe. Electrochemical properties were studied using cyclic voltammetry. SEM analyses indicated the existence of nanoparticles in the carbon samples. XRD analysis showed that carbon sample had sharp peaks indicating crystallite carbon and sylvite. FTIR analysis showed that the carbon have common surface functionalities which also can be found in other conductive carbon samples. The electrical conductivities test showed that the carbon had 0.001–1.5 S cm-1 of conductivity. The shape of the cyclic voltammograms were typical for carbon electrode that use in electrochemical capacitor.
M.Sc. Chemical Engineering Thesis Defense (Omer Farooqi)

This is the presentation for my M.Sc. research thesis. I worked on a novel electrode preparation method to carry out voltammetry in order to detect heavy metals in water.
Growth and Characterization of Barium doped Potassium Hydrogen Phthalate Sing...

The Non Linear Optical materials have acquired new significance with the advent of a large number of devices
utilizing solid state Laser sources. Potassium Hydrogen Phthalate one of the Non Linear Optical material having
superior non linear optical properties has been exploited for variety of application. In the present work, KHP
single crystals were grown by slow evaporation technique with Barium metal ion as a dopant. The grown
crystals were subjected to powder XRD analysis and the result shows that the Ba2+ ions does not alter the crystal
structure, but it enter into the crystal lattice of pure KHP. The optical transparency of the grown crystal was
studied by UV-Visible spectroscopy, the molecular structure was confirmed by FTIR analysis and its thermal
stability by TG/DTA analysis. The improved SHG efficiency of barium doped Potassium Hydrogen Phthalate
crystal could enhance the nonlinearity behaviour. In addition to this, the electrical parameter such as dielectric
constant was studied in detail.
Amyloid–carbon hybrid membranes for universal water purification 

Seminar for the Environmental Engineering Group, University of Oxford, 2017
Synthesis of flower-like magnetite nanoassembly: Application in the efficient...

A facile approach for the synthesis of magnetite microspheres with flower-like morphology is reported
that proceeds via the reduction of iron(III) oxide under a hydrogen atmosphere. The ensuing magnetic
catalyst is well characterized by XRD, FE-SEM, TEM, N2 adsorption-desorption isotherm, and
Mössbauer spectroscopy and explored for a simple yet efficient transfer hydrogenation reduction of a
variety of nitroarenes to respective anilines in good to excellent yields (up to 98%) employing hydrazine
hydrate. The catalyst could be easily separated at the end of a reaction using an external magnet and
can be recycled up to 10 times without any loss in catalytic activity.
Recommended
Hydrothermal Assisted Microwave Pyrolysis of Water Hyacinth for Electrochemic...

We develop ‘green’ approach to prepare conductive carbon material from water hyacinth (Eichhornia crassipes) powder for use in electrochemical capacitor device. The features on morphology, crystallography and surface functionality were analyzed based on SEM, XRD and FTIR instrumentation, respectively. The electrical conductivities were measured using four-point probe. Electrochemical properties were studied using cyclic voltammetry. SEM analyses indicated the existence of nanoparticles in the carbon samples. XRD analysis showed that carbon sample had sharp peaks indicating crystallite carbon and sylvite. FTIR analysis showed that the carbon have common surface functionalities which also can be found in other conductive carbon samples. The electrical conductivities test showed that the carbon had 0.001–1.5 S cm-1 of conductivity. The shape of the cyclic voltammograms were typical for carbon electrode that use in electrochemical capacitor.
M.Sc. Chemical Engineering Thesis Defense (Omer Farooqi)

This is the presentation for my M.Sc. research thesis. I worked on a novel electrode preparation method to carry out voltammetry in order to detect heavy metals in water.
Growth and Characterization of Barium doped Potassium Hydrogen Phthalate Sing...

The Non Linear Optical materials have acquired new significance with the advent of a large number of devices
utilizing solid state Laser sources. Potassium Hydrogen Phthalate one of the Non Linear Optical material having
superior non linear optical properties has been exploited for variety of application. In the present work, KHP
single crystals were grown by slow evaporation technique with Barium metal ion as a dopant. The grown
crystals were subjected to powder XRD analysis and the result shows that the Ba2+ ions does not alter the crystal
structure, but it enter into the crystal lattice of pure KHP. The optical transparency of the grown crystal was
studied by UV-Visible spectroscopy, the molecular structure was confirmed by FTIR analysis and its thermal
stability by TG/DTA analysis. The improved SHG efficiency of barium doped Potassium Hydrogen Phthalate
crystal could enhance the nonlinearity behaviour. In addition to this, the electrical parameter such as dielectric
constant was studied in detail.
Amyloid–carbon hybrid membranes for universal water purification 

Seminar for the Environmental Engineering Group, University of Oxford, 2017
Synthesis of flower-like magnetite nanoassembly: Application in the efficient...

A facile approach for the synthesis of magnetite microspheres with flower-like morphology is reported
that proceeds via the reduction of iron(III) oxide under a hydrogen atmosphere. The ensuing magnetic
catalyst is well characterized by XRD, FE-SEM, TEM, N2 adsorption-desorption isotherm, and
Mössbauer spectroscopy and explored for a simple yet efficient transfer hydrogenation reduction of a
variety of nitroarenes to respective anilines in good to excellent yields (up to 98%) employing hydrazine
hydrate. The catalyst could be easily separated at the end of a reaction using an external magnet and
can be recycled up to 10 times without any loss in catalytic activity.
2017 ACS Spring National Meeting Presentation Slides

Improving the Capacitance of Graphene Aerogel Electrodes via a Facile Ion-intercalation Strategy (oral presentation, ENFL #162, 253rd ACS National Meeting, San Francisco, CA, 2017)
2017 MRS Spring Meeting Presentation Slides

Three-dimensional Nitrogen-doped Graphene Aerogels Enhance Power Density of Microbial Fuel Cells (oral presentation, NM3.1.06, 2017 MRS Spring Meeting and Exhibit, Phoenix, AZ, 2017)
2017 ACS Spring National Meeting Presentation Slides

Manipulate the Chemical Composition and Substrate Nature to Push the Cycling Stability Limit of Polypyrrole (oral presentation, ENFL #114, 253rd ACS National Meeting, San Francisco, CA, 2017)
Ir3615081517

International Journal of Engineering Research and Applications (IJERA) is an open access online peer reviewed international journal that publishes research and review articles in the fields of Computer Science, Neural Networks, Electrical Engineering, Software Engineering, Information Technology, Mechanical Engineering, Chemical Engineering, Plastic Engineering, Food Technology, Textile Engineering, Nano Technology & science, Power Electronics, Electronics & Communication Engineering, Computational mathematics, Image processing, Civil Engineering, Structural Engineering, Environmental Engineering, VLSI Testing & Low Power VLSI Design etc.
H0354854

IOSR Journal of Applied Chemistry (IOSR-JAC) is an open access international journal that provides rapid publication (within a month) of articles in all areas of applied chemistry and its applications. The journal welcomes publications of high quality papers on theoretical developments and practical applications in Chemical Science. Original research papers, state-of-the-art reviews, and high quality technical notes are invited for publications.
Synthesis and applications of graphene based ti o2 photocatalysts

This gives insight on Graphene and TiO2 as a photocatalyst. This presentation has been prepared for personal purpose only.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used
for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after
24 h irradiation was 9934 mmol g1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a
sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride
145 mmol g1cat under identical conditions. The presence of triethylamine was found to be vital for the
higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for
subsequent six runs without significant loss in photo activity.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used
for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after
24 h irradiation was 9934 mmol g1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a
sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride
145 mmol g1cat under identical conditions. The presence of triethylamine was found to be vital for the
higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for
subsequent six runs without significant loss in photo activity.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after 24 h irradiation was 9934 μmol g−1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride 145 μmol g−1cat under identical conditions. The presence of triethylamine was found to be vital for the higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for subsequent six runs without significant loss in photo activity.
Water-splitting photoelectrodes consisting of heterojunctions of carbon nitri...

Quinary and senary non-stoichiometric double perovskites such as Ba2Ca0.66Nb1.34-xFexO6-δ (BCNF) have been utilized for gas sensing, solid oxide fuel cells and thermochemical CO2 reduction. Herein, we examined their potential as narrow bandgap semiconductors for use in solar energy harvesting. A cobalt co-doped BCNF, Ba2Ca0.66Nb0.68Fe0.33Co0.33O6-δ (BCNFCo), exhibited an optical absorption edge at ~ 800 nm, p-type conduction and a distinct photoresponse upto 640 nm while demonstrating high thermochemical stability. A nanocomposite of BCNFCo and g-C3N4 (CN) was prepared via a facile solvent assisted exfoliation/blending approach using dichlorobenzene and glycerol at a moderate temperature. The exfoliation of g-C3N4 followed by wrapping on perovskite established an effective heterojunction between the materials for charge separation. The conjugated 2D sheets of CN enabled better charge migration resulting in increased photoelectrochemical performance. A blend composed of 40 wt% perovskite and CN performed optimally, whilst achieving a photocurrent density as high as 1.5 mA cm-2 for sunlight-driven water-splitting with a Faradaic efficiency as high as ~ 88%.
228 presentation iit bombay new

228 presentation iit bombay new4th International Conference on Advances in Energy Research (ICAER) 2013
Visible light assisted reduction of nitrobenzenes using Fe(bpy)3+2/rGOnanocom...

Visible-light-induced photocatalytic reduction of aromatic nitrobenzenes to the corresponding anilinesat room temperature using reduced graphene oxide (rGO) immobilized iron(II) bipyridine complex asphotocatalyst is described. The rGO-immobilized iron catalyst exhibited superior catalytic activity thanhomogeneous iron(II) bipyridine complex and much higher than metal free rGO photocatalysts. Theheterogeneous photocatalyst was found to be robust and could easily be recovered and reused for severalruns without any significant loss in photocatalytic activity.
Band edge engineering of composite photoanodes for dye sensitized solar cells

Band edge engineering of composite photoanodes for dye-sensitized solar cells
Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...

Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...Aremu Emmanuel Olugbemisola
LiMn2O4 - Spinel cathode material for lithium ion secondary batteryElectrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...

Electrochemical oxidation of methylene blue using carbon composite electrode in sodium chloride
solution have been done. The aim of this work was to degradation of methylen blue using the direct oxidation in
undivided electrolysis cell. Carbon composite electrode was prepare by C powder and PVC in 4 mL
tetrahydrofuran (THF) solvent and swirled flatly to homogeneous followed by drying in an oven at 100 °C for 3
h. The mixture was placed in stainless steel mould and pressed at 10 ton/cm2
. Sodium chloride was used
electrolyte solution. The effects of the current and electrolysis time were investigated using cyclic voltammetry,
spectrophotometer UV-Visible and gas chromatography mass spectrometry (GC-MS). The experimental results
showed that the carbon composite electrode have higher effect in the electrochemical degradation of methylene
blue. Based on UV-visible spectra and GC-MS analysis also shows current and electrolysis time for
degradation of methylene blue optimum at 1,5 A and 60 minute, respectively. As a conclusions is this method its
simple equipment, easy operation and friendly for environmental.
More Related Content
What's hot
2017 ACS Spring National Meeting Presentation Slides

Improving the Capacitance of Graphene Aerogel Electrodes via a Facile Ion-intercalation Strategy (oral presentation, ENFL #162, 253rd ACS National Meeting, San Francisco, CA, 2017)
2017 MRS Spring Meeting Presentation Slides

Three-dimensional Nitrogen-doped Graphene Aerogels Enhance Power Density of Microbial Fuel Cells (oral presentation, NM3.1.06, 2017 MRS Spring Meeting and Exhibit, Phoenix, AZ, 2017)
2017 ACS Spring National Meeting Presentation Slides

Manipulate the Chemical Composition and Substrate Nature to Push the Cycling Stability Limit of Polypyrrole (oral presentation, ENFL #114, 253rd ACS National Meeting, San Francisco, CA, 2017)
Ir3615081517

International Journal of Engineering Research and Applications (IJERA) is an open access online peer reviewed international journal that publishes research and review articles in the fields of Computer Science, Neural Networks, Electrical Engineering, Software Engineering, Information Technology, Mechanical Engineering, Chemical Engineering, Plastic Engineering, Food Technology, Textile Engineering, Nano Technology & science, Power Electronics, Electronics & Communication Engineering, Computational mathematics, Image processing, Civil Engineering, Structural Engineering, Environmental Engineering, VLSI Testing & Low Power VLSI Design etc.
H0354854

IOSR Journal of Applied Chemistry (IOSR-JAC) is an open access international journal that provides rapid publication (within a month) of articles in all areas of applied chemistry and its applications. The journal welcomes publications of high quality papers on theoretical developments and practical applications in Chemical Science. Original research papers, state-of-the-art reviews, and high quality technical notes are invited for publications.
Synthesis and applications of graphene based ti o2 photocatalysts

This gives insight on Graphene and TiO2 as a photocatalyst. This presentation has been prepared for personal purpose only.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used
for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after
24 h irradiation was 9934 mmol g1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a
sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride
145 mmol g1cat under identical conditions. The presence of triethylamine was found to be vital for the
higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for
subsequent six runs without significant loss in photo activity.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used
for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after
24 h irradiation was 9934 mmol g1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a
sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride
145 mmol g1cat under identical conditions. The presence of triethylamine was found to be vital for the
higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for
subsequent six runs without significant loss in photo activity.
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

A novel heteroleptic iridium complex supported on graphitic carbon nitride was synthesized and used for photoreduction of carbon dioxide under visible light irradiation. The methanol yield obtained after 24 h irradiation was 9934 μmol g−1cat (TON 1241 with respect to Ir) by using triethylamine (TEA) as a sacrificial donor, which was significantly higher as compared to the semiconductor carbon nitride 145 μmol g−1cat under identical conditions. The presence of triethylamine was found to be vital for the higher methanol yield. After the reaction, the photocatalyst could easily be recovered and reused for subsequent six runs without significant loss in photo activity.
Water-splitting photoelectrodes consisting of heterojunctions of carbon nitri...

Quinary and senary non-stoichiometric double perovskites such as Ba2Ca0.66Nb1.34-xFexO6-δ (BCNF) have been utilized for gas sensing, solid oxide fuel cells and thermochemical CO2 reduction. Herein, we examined their potential as narrow bandgap semiconductors for use in solar energy harvesting. A cobalt co-doped BCNF, Ba2Ca0.66Nb0.68Fe0.33Co0.33O6-δ (BCNFCo), exhibited an optical absorption edge at ~ 800 nm, p-type conduction and a distinct photoresponse upto 640 nm while demonstrating high thermochemical stability. A nanocomposite of BCNFCo and g-C3N4 (CN) was prepared via a facile solvent assisted exfoliation/blending approach using dichlorobenzene and glycerol at a moderate temperature. The exfoliation of g-C3N4 followed by wrapping on perovskite established an effective heterojunction between the materials for charge separation. The conjugated 2D sheets of CN enabled better charge migration resulting in increased photoelectrochemical performance. A blend composed of 40 wt% perovskite and CN performed optimally, whilst achieving a photocurrent density as high as 1.5 mA cm-2 for sunlight-driven water-splitting with a Faradaic efficiency as high as ~ 88%.
228 presentation iit bombay new

228 presentation iit bombay new4th International Conference on Advances in Energy Research (ICAER) 2013
Visible light assisted reduction of nitrobenzenes using Fe(bpy)3+2/rGOnanocom...

Visible-light-induced photocatalytic reduction of aromatic nitrobenzenes to the corresponding anilinesat room temperature using reduced graphene oxide (rGO) immobilized iron(II) bipyridine complex asphotocatalyst is described. The rGO-immobilized iron catalyst exhibited superior catalytic activity thanhomogeneous iron(II) bipyridine complex and much higher than metal free rGO photocatalysts. Theheterogeneous photocatalyst was found to be robust and could easily be recovered and reused for severalruns without any significant loss in photocatalytic activity.
Band edge engineering of composite photoanodes for dye sensitized solar cells

Band edge engineering of composite photoanodes for dye-sensitized solar cells
Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...

Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...Aremu Emmanuel Olugbemisola
LiMn2O4 - Spinel cathode material for lithium ion secondary batteryWhat's hot (20)
2017 ACS Spring National Meeting Presentation Slides

2017 ACS Spring National Meeting Presentation Slides
2021 influence of basic carbon additives on the electrochemical performance ...

2021 influence of basic carbon additives on the electrochemical performance ...
A closed loop ammonium salt system for recovery of high-purity lead tetroxide...

A closed loop ammonium salt system for recovery of high-purity lead tetroxide...
2017 ACS Spring National Meeting Presentation Slides

2017 ACS Spring National Meeting Presentation Slides
Electrodeposited Ru Nanoparticles for Electrochemical Reduction of NAD+ to NADH

Electrodeposited Ru Nanoparticles for Electrochemical Reduction of NAD+ to NADH
Synthesis and applications of graphene based ti o2 photocatalysts

Synthesis and applications of graphene based ti o2 photocatalysts
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...
Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...

Metal-organic hybrid: Photoreduction of CO2 using graphitic carbon nitride su...
Water-splitting photoelectrodes consisting of heterojunctions of carbon nitri...

Water-splitting photoelectrodes consisting of heterojunctions of carbon nitri...
Visible light assisted reduction of nitrobenzenes using Fe(bpy)3+2/rGOnanocom...

Visible light assisted reduction of nitrobenzenes using Fe(bpy)3+2/rGOnanocom...
Band edge engineering of composite photoanodes for dye sensitized solar cells

Band edge engineering of composite photoanodes for dye sensitized solar cells
Zr doped TiO2 nanocomposites for dye sensitized solar cells

Zr doped TiO2 nanocomposites for dye sensitized solar cells
Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...

Li mn2o4 spinel cathode material for lithium-ion secondary battery by emman...
Similar to 2013 WCU's Poster Presentation(ModifiedII)
Electrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...

Electrochemical oxidation of methylene blue using carbon composite electrode in sodium chloride
solution have been done. The aim of this work was to degradation of methylen blue using the direct oxidation in
undivided electrolysis cell. Carbon composite electrode was prepare by C powder and PVC in 4 mL
tetrahydrofuran (THF) solvent and swirled flatly to homogeneous followed by drying in an oven at 100 °C for 3
h. The mixture was placed in stainless steel mould and pressed at 10 ton/cm2
. Sodium chloride was used
electrolyte solution. The effects of the current and electrolysis time were investigated using cyclic voltammetry,
spectrophotometer UV-Visible and gas chromatography mass spectrometry (GC-MS). The experimental results
showed that the carbon composite electrode have higher effect in the electrochemical degradation of methylene
blue. Based on UV-visible spectra and GC-MS analysis also shows current and electrolysis time for
degradation of methylene blue optimum at 1,5 A and 60 minute, respectively. As a conclusions is this method its
simple equipment, easy operation and friendly for environmental.
Treatment of Hospital Wastewater by Electro-Coagulation Technique

https://www.irjet.net/archives/V10/i5/IRJET-V10I5228.pdf
IRJET- Electrocoagulation Treatment of Electroplating Industrial Effluent

https://www.irjet.net/archives/V6/i7/IRJET-V6I7470.pdf
(LiCoO2, LiMn2O4, LiNixMnyCozO2 (Three (3) composite system), Li(LiwNixMnyCoz...

(LiCoO2, LiMn2O4, LiNixMnyCozO2 (Three (3) composite system), Li(LiwNixMnyCoz...Aremu Emmanuel Olugbemisola
A-Z (LiCoO2, LiMn2O4, LiNixMnyCozO2 (Three (3) composite system), Li(LiwNixMnyCoz)O2 (Li-rich system), LiFePO4) Cathode materials for battery production TREATMENT OF HOSPITAL WASTEWATER BY USING ELECTROCOAGULATION

https://www.irjet.net/archives/V10/i10/IRJET-V10I1038.pdf
TREATING WASTE WATER USING ELECTROCOAGULATION APPROACH

Performance of Electro coagulation method is evaluated by treating various
sources of water. The electrodes of stainless steel are used for arranging electrolytic
cell. The treatment efficiency is assessed by varying electrical potential and
concentration time. The parameters namely pH, Total Dissolved Solids (TDS), Total
Hardness, Electrical Conductivity, Biochemical Oxygen Demand (BOD) and
Chemical Oxygen Demand (COD) were tested and results were compared to assess
the performance of treatment process. Effects of operating parameters such as
varying electrical potential (3V-12V) and concentration time (10 minutes- 30 minutes)
were evaluated for optimum operating conditions. The result shown that identified
electrolytic arrangement is significantly efficient for treating BOD and COD when
compared to other water quality parameters especially for grey and industrial water
samples. The highest removal efficiency under optimum operating condition for COD
and BOD removal were obtained with 58% and 67% in grey water sample whereas in
industrial waste water sample, the efficiencies are 53% and 57%. Further, the study
suggested the various simulations in the operating parameters with the economic
considerations to optimize the findings and for upcoming progress in the application
of this advanced technology.
Manufacture of caustic soda and chlorine using electrolysis process ...

PROJECT PRESENTATION - PHASE 1
Phase 2
http://www.slideshare.net/AnkushGupta40/phase23
Removal of Heavy Metals from Water using Electrocoagulation

https://www.irjet.net/archives/V9/i6/IRJET-V9I6446.pdf
Removal of turbidity using electrocoagulation

Abstract Electrocoagulation (EC) is a technique involving the electrolytic addition of coagulating metal ions directly from electrodes, this electrode also known as sacrificial electrode. These ions work as a coagulating agent in the water, similar manner to the addition of chemicals such as alum and ferric chloride. Electrode allows the easier removal of the turbidity. In this study experimental investigation is carried out to removal of turbidity using electrocoagulation method with the help of aluminum electrodes in a batch reactor. Several working parameters, such as pH, current density, and operating time were varied to achieve a higher removal efficiency of turbidity. Two type of water sample were used1) synthetic turbid water and 2) back wash water synthetic water was made from crushed sand with initial turbidity of 200 NTU whereas back wash water was collected from Nigadi water treatment plant . Current intensity was varied from 2 to 6 A. and detention time between 5 to 20 minutes with pH range from pH 3 to pH 11. The removal efficiencies for synthetic turbidity were found about 91% at 6A for 20 minutes at neutral pH, whereas removal efficiencies for backwash water were found about 89% at 6A for 20 minutes at neutral pH. The results of the study indicated that the turbidity can be removed effectively by EC process using Al electrodes. Keywords: Electrocoagulation, sacrificial electrode, Turbidity, Water Treatment
Similar to 2013 WCU's Poster Presentation(ModifiedII) (20)
Electrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...

Electrochemical Degradation of Methylen Blue Using Carbon Composite Electrode...
Treatment of Hospital Wastewater by Electro-Coagulation Technique

Treatment of Hospital Wastewater by Electro-Coagulation Technique
IRJET- Electrocoagulation Treatment of Electroplating Industrial Effluent

IRJET- Electrocoagulation Treatment of Electroplating Industrial Effluent
(LiCoO2, LiMn2O4, LiNixMnyCozO2 (Three (3) composite system), Li(LiwNixMnyCoz...

(LiCoO2, LiMn2O4, LiNixMnyCozO2 (Three (3) composite system), Li(LiwNixMnyCoz...
01 Judd Sundine - Mine Water Solutions in Extreme Environments 2015

01 Judd Sundine - Mine Water Solutions in Extreme Environments 2015
TREATMENT OF HOSPITAL WASTEWATER BY USING ELECTROCOAGULATION

TREATMENT OF HOSPITAL WASTEWATER BY USING ELECTROCOAGULATION
TREATING WASTE WATER USING ELECTROCOAGULATION APPROACH

TREATING WASTE WATER USING ELECTROCOAGULATION APPROACH
A high-capacity lithium-air battery with Pd modified carbon nanotube sponge c...

A high-capacity lithium-air battery with Pd modified carbon nanotube sponge c...
Manufacture of caustic soda and chlorine using electrolysis process ...

Manufacture of caustic soda and chlorine using electrolysis process ...
Removal of Heavy Metals from Water using Electrocoagulation

Removal of Heavy Metals from Water using Electrocoagulation
2013 WCU's Poster Presentation(ModifiedII)
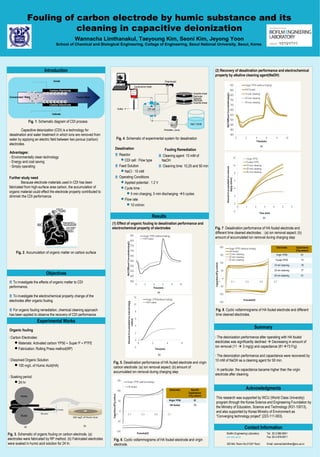
- 1. Poster template by ResearchPosters.co.za Fouling of carbon electrode by humic substance and its cleaning in capacitive deionization Wannacha Limthanakul, Taeyoung Kim, Seoni Kim, Jeyong Yoon School of Chemical and Biological Engineering, College of Engineering, Seoul National University, Seoul, Korea Introduction Objectives Experimental Works Results Summary Carbon Electrode Carbon Electrode + - + + + +- - - - - - - - - + + + + + + + + - - Treated WaterConcentrated Water Anode Cathode Fig. 1. Schematic diagram of CDI process Capacitive deionization (CDI) is a technology for desalination and water treatment in which ions are removed from water by applying an electric field between two porous (carbon) electrodes. Advantages: - Environmentally clean technology - Energy and cost saving - Simply operating Further study need Because electrode materials used in CDI has been fabricated from high-surface area carbon, the accumulation of organic material could effect the electrode property contributed to diminish the CDI performance. Fig. 2. Accumulation of organic matter on carbon surface Source: wateensolutions.com.au ⧄ To investigate the effects of organic matter to CDI performance. ⧄ To investigate the electrochemical property change of the electrodes after organic fouling. ⧄ For organic fouling remediation, chemical cleaning approach has been applied to observe the recovery of CDI performance. ⧄ Carbon Electrodes: Materials: Activated carbon YP50 + Super P + PTFE Fabrication: Rolling Press method(RP) ⧄ Dissolved Organic Solution 100 mg/L of Humic Acid(HA) ⧄ Soaking period 24 hr 100 mg/L of Humic Acid Roller Roller Carbon mixture 50 mm 0.3 mm (a) (b) Fig. 3. Schematic of organic fouling on carbon electrode. (a) electrodes were fabricated by RP method. (b) Fabricated electrodes were soaked in humic acid solution for 24 hr. Desalination ⧄ Reactor CDI cell : Flow type ⧄ Feed Solution NaCl : 10 mM ⧄ Operating Conditions Applied potential : 1.2 V Cycle time : 5 min charging, 5 min discharging 5 cycles Flow rate 10 ml/min Organic fouling Potentiostat Outlet Conductivity meter CDI cell NaCl 10mM Peristaltic pump Graphite sheet Graphite sheet Electrode Electrode Spacers Fig. 4. Schematic of experimental system for desalination ⧄ Cleaning agent: 10 mM of NaOH ⧄ Cleaning time: 10,25 and 50 min Fouling Remediation (1) Effect of organic fouling to desalination performance and electrochemical property of electrodes (a) (b) Fig. 5. Desalination performance of HA fouled electrode and virgin carbon electrode: (a) ion removal aspect; (b) amount of accumulated ion removal during charging step Electrodes Specific Capacitance (F/g carbon) Virgin YP50 81 HA fouled 73 Fig. 6. Cyclic voltammograms of HA fouled electrode and virgin electrode. (2) Recovery of desalination performance and electrochemical property by alkaline cleaning agent(NaOH) (a) (b) Fig. 7. Desalination performance of HA fouled electrode and different time cleaned electrodes. : (a) ion removal aspect; (b) amount of accumulated ion removal during charging step Electrodes Capacitance (F/g carbon) Virgin YP50 81 Fouled YP50 73 10 min cleaning 78 25 min cleaning 77 50 min cleaning 91 Fig. 8. Cyclic voltammograms of HA fouled electrode and different time cleaned electrodes. ⧄ The deionization performance after operating with HA fouled electrodes was significantly declined Decreasing in amount of ion removal (11 3 mg/g) and capacitance (8173 F/g) ⧄ The deionization performance and capacitance were recovered by 10 mM of NaOH as a cleaning agent for 50 min. ⧄ In particular, the capacitance became higher than the virgin electrode after cleaning. Contact Information Biofilm Engineering Laboratory env.snu.ac.kr 302 Bld. Room No.513(5th floor) Tel: 82-2-880-8941 Fax: 82-2-876-8911 Email: wannachalimthan@snu.ac.kr Acknowledgments This research was supported by WCU (World Class University) program through the Korea Science and Engineering Foundation by the Ministry of Education, Science and Technology (R31-10013), and also supported by Korea Ministry of Environment as “Converging technology project” (223-111-003).
