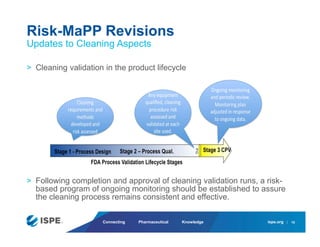
This document summarizes ISPE's guides on cleaning and cleaning validation. It discusses ISPE's Risk-MaPP guide and how it introduces health-based limits for residues that are applied to cleaning. It also provides an overview of the status of ISPE's Cleaning Baseline Guide, explaining that it did not receive sufficient regulatory review to be published as a Baseline Guide. Finally, it announces that ISPE is forming a team to develop a new Good Practice Guide on cleaning that will provide specific guidance and examples on cleaning practices and validation.