Generic product development with QbD pathway
•
1 like•152 views
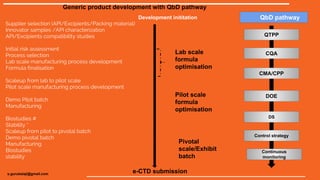
This document outlines the key steps for generic drug product development using a Quality by Design (QbD) pathway, including supplier selection, API and excipient compatibility studies, process development from lab to pilot to pivotal scale, formula optimization at each stage, manufacturing demonstration batches, biostudies, stability testing, and e-CTD regulatory submission with defined quality target product profile, critical quality attributes, critical material attributes, and control strategy.
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
What's hot
What's hot (19)
Role of quality by design (qb d) in quality assurance of pharmaceutical product

Role of quality by design (qb d) in quality assurance of pharmaceutical product
Quality by design for Pharmaceutical Industries: An introduction

Quality by design for Pharmaceutical Industries: An introduction
PAT and QbD concepts in designing the LiMS and other Electronic systems in La...

PAT and QbD concepts in designing the LiMS and other Electronic systems in La...
Talk on QbD by Nitin Kadam at International Conference on Novel Formulation S...

Talk on QbD by Nitin Kadam at International Conference on Novel Formulation S...
Key Components of Pharmaceutical QbD, an Introduction

Key Components of Pharmaceutical QbD, an Introduction
Considering Quality by Design (QbD) in Analytical Development for Protein The...

Considering Quality by Design (QbD) in Analytical Development for Protein The...
More from Guru Balaji .S
More from Guru Balaji .S (20)
Basic Candlestick Pattern in stock market, finance

Basic Candlestick Pattern in stock market, finance
Specifications of process parameters and general ipc and finish product controls

Specifications of process parameters and general ipc and finish product controls
Points to be considered in topical formulation design

Points to be considered in topical formulation design
Recently uploaded
Recently uploaded (20)
Ahmedabad Call Girls CG Road 🔝9907093804 Short 1500 💋 Night 6000

Ahmedabad Call Girls CG Road 🔝9907093804 Short 1500 💋 Night 6000
Call Girls Whitefield Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Whitefield Just Call 7001305949 Top Class Call Girl Service Available
Hi,Fi Call Girl In Mysore Road - 7001305949 | 24x7 Service Available Near Me

Hi,Fi Call Girl In Mysore Road - 7001305949 | 24x7 Service Available Near Me
Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai

Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai
Kesar Bagh Call Girl Price 9548273370 , Lucknow Call Girls Service

Kesar Bagh Call Girl Price 9548273370 , Lucknow Call Girls Service
Call Girls Chennai Megha 9907093804 Independent Call Girls Service Chennai

Call Girls Chennai Megha 9907093804 Independent Call Girls Service Chennai
VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...

VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...
Vip Call Girls Anna Salai Chennai 👉 8250192130 ❣️💯 Top Class Girls Available

Vip Call Girls Anna Salai Chennai 👉 8250192130 ❣️💯 Top Class Girls Available
Sonagachi Call Girls Services 9907093804 @24x7 High Class Babes Here Call Now

Sonagachi Call Girls Services 9907093804 @24x7 High Class Babes Here Call Now
VIP Call Girls Indore Kirti 💚😋 9256729539 🚀 Indore Escorts

VIP Call Girls Indore Kirti 💚😋 9256729539 🚀 Indore Escorts
Call Girls Colaba Mumbai ❤️ 9920874524 👈 Cash on Delivery

Call Girls Colaba Mumbai ❤️ 9920874524 👈 Cash on Delivery
VIP Call Girls Pune Vrinda 9907093804 Short 1500 Night 6000 Best call girls S...

VIP Call Girls Pune Vrinda 9907093804 Short 1500 Night 6000 Best call girls S...
Call Girls Yelahanka Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Yelahanka Just Call 7001305949 Top Class Call Girl Service Available
CALL ON ➥9907093804 🔝 Call Girls Hadapsar ( Pune) Girls Service

CALL ON ➥9907093804 🔝 Call Girls Hadapsar ( Pune) Girls Service
Call Girls Service Surat Samaira ❤️🍑 8250192130 👄 Independent Escort Service ...

Call Girls Service Surat Samaira ❤️🍑 8250192130 👄 Independent Escort Service ...
Call Girls Service Noida Maya 9711199012 Independent Escort Service Noida

Call Girls Service Noida Maya 9711199012 Independent Escort Service Noida
CALL ON ➥9907093804 🔝 Call Girls Baramati ( Pune) Girls Service

CALL ON ➥9907093804 🔝 Call Girls Baramati ( Pune) Girls Service
Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service

Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service
Russian Call Girls in Chennai Pallavi 9907093804 Independent Call Girls Servi...

Russian Call Girls in Chennai Pallavi 9907093804 Independent Call Girls Servi...
Generic product development with QbD pathway
- 1. Supplier selection (API/Excipients/Packing material) Innovator samples /API characterization API/Excipients compatibility studies Initial risk assessment Process selection Lab scale manufacturing process development Formula finalisation Scaleup from lab to pilot scale Pilot scale manufacturing process development Demo Pilot batch Manufacturing Biostudies # Stability * Scaleup from pilot to pivotal batch Demo pivotal batch Manufacturing Biostudies stability Lab scale formula optimisation Pilot scale formula optimisation Pivotal scale/Exhibit batch Development inititation e-CTD submission Generic product development with QbD pathway QbD pathway QTPP CQA CMA/CPP DOE DS Control strategy Continuous monitoring s.gurubalaji@gmail.com
