Generics drug approval and timelines in china
•Download as PPTX, PDF•
0 likes•368 views
pharmaceuticals#validation#parameters#quality#China#pharma #health #medicine #research #manufacturing #innovation #research #development #Healthcare #quality #pharmacist #technology #CRO #compliance #counterfeits #gurubalaji
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Tabular summary of New Drugs & Clinical Trials Rules, 2019 [INDIA]![Tabular summary of New Drugs & Clinical Trials Rules, 2019 [INDIA]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Tabular summary of New Drugs & Clinical Trials Rules, 2019 [INDIA]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Tabular summary of New Drugs & Clinical Trials Rules, 2019 [INDIA]
Quality regulation for biological products current and future

Quality regulation for biological products current and future
Regulatory affairs and Intellectual Property Rights

Regulatory affairs and Intellectual Property Rights
Good clinical practices tutorial-june-21-09 shehnaz-v7.0

Good clinical practices tutorial-june-21-09 shehnaz-v7.0
Similar to Generics drug approval and timelines in china
Similar to Generics drug approval and timelines in china (20)
Clinical Trials in India | Veeda Clinical Research

Clinical Trials in India | Veeda Clinical Research
Regulation Governing Clinical Trials In India,USA and Europe. 

Regulation Governing Clinical Trials In India,USA and Europe.
More from Guru Balaji .S
More from Guru Balaji .S (20)
Basic Candlestick Pattern in stock market, finance

Basic Candlestick Pattern in stock market, finance
Specifications of process parameters and general ipc and finish product controls

Specifications of process parameters and general ipc and finish product controls
Points to be considered in topical formulation design

Points to be considered in topical formulation design
Recently uploaded
sauth delhi call girls in Bhajanpura 🔝 9953056974 🔝 escort Service

sauth delhi call girls in Bhajanpura 🔝 9953056974 🔝 escort Service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Escort Service Call Girls In Sarita Vihar,, 99530°56974 Delhi NCR

Escort Service Call Girls In Sarita Vihar,, 99530°56974 Delhi NCR9953056974 Low Rate Call Girls In Saket, Delhi NCR
Call Girls Service Navi Mumbai Samaira 8617697112 Independent Escort Service ...

Call Girls Service Navi Mumbai Samaira 8617697112 Independent Escort Service ...Call girls in Ahmedabad High profile
Recently uploaded (20)
sauth delhi call girls in Bhajanpura 🔝 9953056974 🔝 escort Service

sauth delhi call girls in Bhajanpura 🔝 9953056974 🔝 escort Service
College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...

College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...
VIP Call Girls Tirunelveli Aaradhya 8250192130 Independent Escort Service Tir...

VIP Call Girls Tirunelveli Aaradhya 8250192130 Independent Escort Service Tir...
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
Low Rate Call Girls Pune Esha 9907093804 Short 1500 Night 6000 Best call girl...

Low Rate Call Girls Pune Esha 9907093804 Short 1500 Night 6000 Best call girl...
♛VVIP Hyderabad Call Girls Chintalkunta🖕7001035870🖕Riya Kappor Top Call Girl ...

♛VVIP Hyderabad Call Girls Chintalkunta🖕7001035870🖕Riya Kappor Top Call Girl ...
Call Girls Darjeeling Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Darjeeling Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available
Bangalore Call Girls Hebbal Kempapura Number 7001035870 Meetin With Bangalor...

Bangalore Call Girls Hebbal Kempapura Number 7001035870 Meetin With Bangalor...
Call Girls Colaba Mumbai ❤️ 9920874524 👈 Cash on Delivery

Call Girls Colaba Mumbai ❤️ 9920874524 👈 Cash on Delivery
💎VVIP Kolkata Call Girls Parganas🩱7001035870🩱Independent Girl ( Ac Rooms Avai...

💎VVIP Kolkata Call Girls Parganas🩱7001035870🩱Independent Girl ( Ac Rooms Avai...
Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...

Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...
VIP Call Girls Indore Kirti 💚😋 9256729539 🚀 Indore Escorts

VIP Call Girls Indore Kirti 💚😋 9256729539 🚀 Indore Escorts
Escort Service Call Girls In Sarita Vihar,, 99530°56974 Delhi NCR

Escort Service Call Girls In Sarita Vihar,, 99530°56974 Delhi NCR
Vip Call Girls Anna Salai Chennai 👉 8250192130 ❣️💯 Top Class Girls Available

Vip Call Girls Anna Salai Chennai 👉 8250192130 ❣️💯 Top Class Girls Available
VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...

VIP Mumbai Call Girls Hiranandani Gardens Just Call 9920874524 with A/C Room ...
Call Girls Service Navi Mumbai Samaira 8617697112 Independent Escort Service ...

Call Girls Service Navi Mumbai Samaira 8617697112 Independent Escort Service ...
Bangalore Call Girl Whatsapp Number 100% Complete Your Sexual Needs

Bangalore Call Girl Whatsapp Number 100% Complete Your Sexual Needs
Bangalore Call Girls Nelamangala Number 7001035870 Meetin With Bangalore Esc...

Bangalore Call Girls Nelamangala Number 7001035870 Meetin With Bangalore Esc...
Call Girl Number in Panvel Mumbai📲 9833363713 💞 Full Night Enjoy

Call Girl Number in Panvel Mumbai📲 9833363713 💞 Full Night Enjoy
Generics drug approval and timelines in china
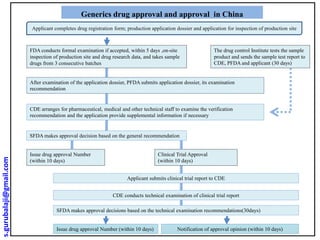
- 1. Applicant completes drug registration form; production application dossier and application for inspection of production site FDA conducts formal examination if accepted, within 5 days ,on-site inspection of production site and drug research data, and takes sample drugs from 3 consecutive batches CDE arranges for pharmaceutical, medical and other technical staff to examine the verification recommendation and the application provide supplemental information if necessary SFDA makes approval decision based on the general recommendation Issue drug approval Number (within 10 days) Clinical Trial Approval (within 10 days) The drug control Institute tests the sample product and sends the sample test report to CDE, PFDA and applicant (30 days) Applicant submits clinical trial report to CDE CDE conducts technical examination of clinical trial report SFDA makes approval decisions based on the technical examination recommendations(30days) Issue drug approval Number (within 10 days) Notification of approval opinion (within 10 days) After examination of the application dossier, PFDA submits application dossier, its examination recommendation Generics drug approval and approval in China s.gurubalaji@gmail.com
