Facts about Generics Drugs
•
0 likes•283 views
Facts about generic drugs
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
Similar to Facts about Generics Drugs
Similar to Facts about Generics Drugs (20)
Consumer Preference towards Generic and Branded medicines.pptx

Consumer Preference towards Generic and Branded medicines.pptx
Recently uploaded
TEST BANK For Porth's Essentials of Pathophysiology, 5th Edition by Tommie L Norris, Verified Chapters 1 - 52, Complete Newest VersionTEST BANK For Porth's Essentials of Pathophysiology, 5th Edition by Tommie L ...

TEST BANK For Porth's Essentials of Pathophysiology, 5th Edition by Tommie L ...rightmanforbloodline
PEMESANAN OBAT ASLI : +6287776558899
Cara Menggugurkan Kandungan usia 1 , 2 , bulan - obat penggugur janin - cara aborsi kandungan - obat penggugur kandungan 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 bulan - bagaimana cara menggugurkan kandungan - tips Cara aborsi kandungan - trik Cara menggugurkan janin - Cara aman bagi ibu menyusui menggugurkan kandungan - klinik apotek jual obat penggugur kandungan - jamu PENGGUGUR KANDUNGAN - WAJIB TAU CARA ABORSI JANIN - GUGURKAN KANDUNGAN AMAN TANPA KURET - CARA Menggugurkan Kandungan tanpa efek samping - rekomendasi dokter obat herbal penggugur kandungan - ABORSI JANIN - aborsi kandungan - jamu herbal Penggugur kandungan - cara Menggugurkan Kandungan yang cacat - tata cara Menggugurkan Kandungan - obat penggugur kandungan di apotik kimia Farma - obat telat datang bulan - obat penggugur kandungan tuntas - obat penggugur kandungan alami - klinik aborsi janin gugurkan kandungan - ©Cytotec ™misoprostol BPOM - OBAT PENGGUGUR KANDUNGAN ®CYTOTEC - aborsi janin dengan pil ©Cytotec - ®Cytotec misoprostol® BPOM 100% - penjual obat penggugur kandungan asli - klinik jual obat aborsi janin - obat penggugur kandungan di klinik k-24 || obat penggugur ™Cytotec di apotek umum || ®CYTOTEC ASLI || obat ©Cytotec yang asli 200mcg || obat penggugur ASLI || pil Cytotec© tablet || cara gugurin kandungan || jual ®Cytotec 200mcg || dokter gugurkan kandungan || cara menggugurkan kandungan dengan cepat selesai dalam 24 jam secara alami buah buahan || usia kandungan 1_2 3_4 5_6 7_8 bulan masih bisa di gugurkan || obat penggugur kandungan ®cytotec dan gastrul || cara gugurkan pembuahan janin secara alami dan cepat || gugurkan kandungan || gugurin janin || cara Menggugurkan janin di luar nikah || contoh aborsi janin yang benar || contoh obat penggugur kandungan asli || contoh cara Menggugurkan Kandungan yang benar || telat haid || obat telat haid || Cara Alami gugurkan kehamilan || obat telat menstruasi || cara Menggugurkan janin anak haram || cara aborsi menggugurkan janin yang tidak berkembang || gugurkan kandungan dengan obat ©Cytotec || obat penggugur kandungan ™Cytotec 100% original || HARGA obat penggugur kandungan || obat telat haid 1 bulan || obat telat menstruasi 1-2 3-4 5-6 7-8 BULAN || obat telat datang bulan || cara Menggugurkan janin 1 bulan || cara Menggugurkan Kandungan yang masih 2 bulan || cara Menggugurkan Kandungan yang masih hitungan Minggu || cara Menggugurkan Kandungan yang masih usia 3 bulan || cara Menggugurkan usia kandungan 4 bulan || cara Menggugurkan janin usia 5 bulan || cara Menggugurkan kehamilan 6 Bulan
________&&&_________&&&_____________&&&_________&&&&____________
Cara Menggugurkan Kandungan Usia Janin 1 | 7 | 8 Bulan Dengan Cepat Dalam Hitungan Jam Secara Alami, Kami Siap Meneriman Pesanan Ke Seluruh Indonesia, Melputi: Ambon, Banda Aceh, Bandung, Banjarbaru, Batam, Bau-Bau, Bengkulu, Binjai, Blitar, Bontang, Cilegon, Cirebon, Depok, Gorontalo, Jakarta, Jayapura, Kendari, Kota Mobagu, Kupang, LhokseumaweCara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...Cara Menggugurkan Kandungan 087776558899
Recently uploaded (20)
HISTORY, CONCEPT AND ITS IMPORTANCE IN DRUG DEVELOPMENT.pptx

HISTORY, CONCEPT AND ITS IMPORTANCE IN DRUG DEVELOPMENT.pptx
Physicochemical properties (descriptors) in QSAR.pdf

Physicochemical properties (descriptors) in QSAR.pdf
Circulatory Shock, types and stages, compensatory mechanisms

Circulatory Shock, types and stages, compensatory mechanisms
VIP ℂall Girls Kothanur {{ Bangalore }} 6378878445 WhatsApp: Me 24/7 Hours Se...

VIP ℂall Girls Kothanur {{ Bangalore }} 6378878445 WhatsApp: Me 24/7 Hours Se...
See it and Catch it! Recognizing the Thought Traps that Negatively Impact How...

See it and Catch it! Recognizing the Thought Traps that Negatively Impact How...
TEST BANK For Porth's Essentials of Pathophysiology, 5th Edition by Tommie L ...

TEST BANK For Porth's Essentials of Pathophysiology, 5th Edition by Tommie L ...
Dr. A Sumathi - LINEARITY CONCEPT OF SIGNIFICANCE.pdf

Dr. A Sumathi - LINEARITY CONCEPT OF SIGNIFICANCE.pdf
ANATOMY AND PHYSIOLOGY OF REPRODUCTIVE SYSTEM.pptx

ANATOMY AND PHYSIOLOGY OF REPRODUCTIVE SYSTEM.pptx
Obat Aborsi Ampuh Usia 1,2,3,4,5,6,7 Bulan 081901222272 Obat Penggugur Kandu...

Obat Aborsi Ampuh Usia 1,2,3,4,5,6,7 Bulan 081901222272 Obat Penggugur Kandu...
Jual Obat Aborsi Di Dubai UAE Wa 0838-4800-7379 Obat Penggugur Kandungan Cytotec

Jual Obat Aborsi Di Dubai UAE Wa 0838-4800-7379 Obat Penggugur Kandungan Cytotec
Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...
MOTION MANAGEMANT IN LUNG SBRT BY DR KANHU CHARAN PATRO

MOTION MANAGEMANT IN LUNG SBRT BY DR KANHU CHARAN PATRO
Cardiac Output, Venous Return, and Their Regulation

Cardiac Output, Venous Return, and Their Regulation
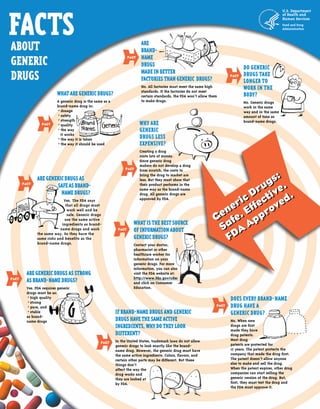
Facts about Generics Drugs
- 1. Why are generic drugS less expensive? Creating a drug costs lots of money. Since generic drug makers do not develop a drug from scratch, the costs to bring the drug to market are less. But they must show that their product performs in the same way as the brand-name drug. All generic drugs are approved by FDA. What is the best source of information about generic drugs? Contact your doctor, pharmacist or other healthcare worker for information on your generic drugs. For more information, you can also visit the FDA website at: http://www.fda.gov/cder and click on Consumer Education. If brand-name drugs and generic drugs have the same active ingredients, why do they look different? In the United States, trademark laws do not allow generic drugs to look exactly like the brand-name drug. However, the generic drug must have the same active ingredients. Colors, flavors, and certain other parts may be different. But these things don’t affect the way the drug works and they are looked at by FDA. What are generic drugs? A generic drug is the same as a brand-name drug in: • dosage • safety • strength • quality • the way it works • the way it is taken • the way it should be used FACT Are generic drugs as safe as brand-name drugs? Yes. The FDA says that all drugs must work well and be safe. Generic drugs use the same active ingredients as brand-name drugs and work the same way. So they have the same risks and benefits as the brand-name drugs. Are brand-name drugs made in BETTER factories than generic drugs? No. All factories must meet the same high standards. If the factories do not meet certain standards, the FDA won’t allow them to make drugs. Do generic drugs take longer to work in the body? No. Generic drugs work in the same way and in the same amount of time as brand-name drugs. U.S. Department of Health and Human Services Food and Drug Administration Does every BRAND-NAME drug have a generic DRUG? No. When new drugs are first made they have drug patents. Most drug patents are protected for 17 years. The patent protects the company that made the drug first. The patent doesn’t allow anyone else to make and sell the drug. When the patent expires, other drug companies can start selling the generic version of the drug. But, first, they must test the drug and the FDA must approve it. Are generic drugs as strong as brand-name drugs? Yes. FDA requires generic drugs must be as: • high quality • strong • pure, and • stable as brand-name drugs FACT FACT FACT FACT FACT FACT FACT FACT Generic Drugs: Safe. Effective. FDA Approved.