Chapter 21 alkylating agents,platins,antimetabolites
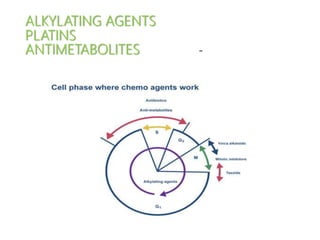
- 2. ALKYLATING AGENTS • cell cycle–phase nonspecific. • DNA and RNA and proteins are alkylated (N-7 position of guanine) the O6 group of guanine is alkylated by nitrosoureas. • Alkylation abnormal nucleotide sequences, miscoding of messenger RNA, cross-linked DNA strands that cannot replicate, breakage of DNA strandsdamage to the transcription and translation of genetic material. • cross-linking of DNA strands & breaks in DNA. not able to complete the replication cyclecytotoxicity. • Tumor resistance ↑capacity of cells to repair nucleic acid damage and to inactivate the drugs by conjugation with glutathione.
- 3. Altretamine Indication.- Recurrent ca ovary Pharmacology a. M/A- unknown. b. Metabolism. Rapidly demethylated and hydroxylated in the liver by the microsomal P450 system. Excreted in urine and hepatobiliary tract as metabolites Toxicity a. Dose limiting. Nausea and vomiting, b. Common. • Neurotoxicity (25%), hallucinations,hypoesthesia, hyperreflexia, motor weakness, confusion,lethargy,including paresthesias, agitation, coma;depression, • myelosuppression (mild) • Nausea , vomiting c. Occasional. • Abnormal LFTs, flulike syndrome; abdominal cramps, diarrhea d. Rare. • Alopecia, skin rashes, cystitis
- 4. Bendamustine Indications.CLL, low-grade B-cell NHL that has progressed within 6 mths of t/t with a rituximab-containing regimen Pharmacology. Bendamustine is a bifunctional mechlorethamine derivative containing a purine-like benzimidazole ring. About 90% of the drug is excreted in the feces. Toxicity a. Dose limiting. -Hematosuppression b. Common. • Nausea, vomiting, diarrhea, fever, fatigue, headache, stomatitis, rash, infusion reactions (consider administering an antihistamine,acetaminophen, prophylactically) c. Occasional. Anaphylactic reactions, severe skin reactions, acute renal failure; peripheral edema, dizziness; myelodysplasia; dysgeusia
- 5. Busulfan Indications. Part of conditioning regimen for BMT, CML palliation Pharmacology. Acts directly; catabolized to inactive products that are excreted in the urine. Toxicity a. Dose limiting. Reversible and irreversible myelosuppression with slow recovery b. Common. Mild GI upset, sterility c. Occasional. Skin hyperpigmentation,alopecia, rash,gynecomastia, cataracts, LFT abnormalities; seizures d. Rare. Pulmonary fibrosis (“busulfan lung”), retroperitoneal fibrosis, endocardial fibrosis; addisonian-like asthenia (without biochemical evidence of adrenal insufficiency); hypotension, impotence, hemorrhagic cystitis, secondary neoplasms
- 6. Chlorambucil Indications. CLL, Waldenström macroglobulinemia, lymphomas Pharmacology. It acts directly; spontaneously hydrolyzed to inactive and active products ; also is extensively metabolized by the hepatic P450 microsomal system. The drug and metabolic products are excreted in urine. Toxicity. Least toxic alkylating agent a. Dose limiting. Myelosuppression b. Occasional. GI upset , mild LFT alter, sterility, rash c. Rare. Rash, alopecia, fever; cachexia, pulmonary fibrosis, neurologic or ocular toxicity, cystitis; acute leukemia
- 7. Cyclophosphamide Indications.HL, lymphocytic lymphoma, mixed cell–type lymphoma, histiocytic lymphoma, Burkitt lymphoma; multiple myeloma, leukemias, mycosis fungoides, neuroblastoma, adenocarcinoma of ovary, retinoblastoma, breast carcinoma, conditioning regimen for BMT Pharmacology. drug is inactive activation by liver P450 system acrolein and an alkylating metabolite (e.g., phosphoramide mustard). Active and inactive metabolites are excreted in the urine. Toxicity a. Dose limiting (1) Myelosuppression. (2)hemorrhagic cystitis- o prevented by maintaining a high UOP. o more common and can be severe when massive doses are used (e.g.,BMT); under these circumstances, the use of mesna can be preventative. o UB fibrosis with telangiectasia of the mucosa can occur without episodes of cystitis. o Bladder carcinoma. b. Side effects (1) Common. Alopecia, stomatitis, aspermia, amenorrhea; headache Nausea and vomiting. (2) Occasional. Skin or fingernail hyperpigmentation; metallic taste during injection; sneezing or a cold sensation in the nose after injection; abnormal LFTs, dizziness; allergy, fever (3) Rare. Transient SIADH, hypothyroidism, cataracts,jaundice,pulmonary fibrosis; cardiac necrosis and acute myopericarditis; secondary neoplasms
- 9. Dacarbazine [dimethyl-triazeno-imidazole-carboxamide (DTIC, DIC) Indications. HL,MM, sarcomas, Neuroblastoma Pharmacology a. Mechanisms. Dacarbazine acts as a purine analog and inhibits DNA synthesis; it is an alkylating agent and it interacts with SH groups. b. Metabolism. liveroxidative N methylation by CYP 450activation Excreted in urine predominantly, minor HB & pulmonary excretion Toxicity a. Dose limiting. Myelosuppression b. Common. Nausea and vomiting (often severe), anorexia; pain along the injection site c. Occasional. Alopecia, facial flushing, photosensitivity, abnormal LFTs. Flulike syndrome d. Rare. Diarrhea, stomatitis; cerebral dysfunction; hepatic necrosis; azotemia; anaphylaxis
- 10. Ifosfamide Indications. Lymphomas, sarcomas, relapsed testicular tumors, and various carcinomas Pharmacology a. M/A.- produces phosphotriesters as the predominant rexn products. The t/t of intact cell nuclei may also result in the formation of DNA–DNA cross-links. b. Metabolism. hepatic activation acrolein and its alkylating metabolite. Acrolein is highly toxic to urothelial mucosa. The chloroacetaldehyde neurotoxic effects, particularly in patients with renal dysfunction. Drug and metabolites are excreted in urine.
- 11. Toxicity a. Dose limiting. Myelosuppression, hemorrhagic cystitis, encephalopathy b. Common. Alopecia; anorexia, constipation, nausea, and vomiting; amenorrhea, oligospermia, and infertility c. Neurotoxicity d. Occasional. Salivation, stomatitis, diarrhea; urticaria, hyperpigmentation, nail ridging; abnormal LFTs, phlebitis, fever; hypotension, hypertension, hypokalemia; renal tubular acidosis (at high doses); SIADH e. Rare. Coma; renal tubular acidosis, or Fanconi-like syndrome
- 13. MESNA • Inj- 100 mg /ml • Tablet- 400 mg • Prevention of ifosfamide HC 240 mg/m2 IV over 15 min before & 4 & 8 hrs after 240 mg/m2 IV 15 min before & 480 mg/m2 of mesna tab 2 & 6 hrs after For cyclophosphamide-20% when injected, 4 & 8 hrs after
- 14. Melphalan Indications. MM , BMT Pharmacology a. M/A.- A phenylalanine derivative of nitrogen mustard, an alkylating agent b. Metabolism. Acts directly. Melphalan is excreted in the urine (about 30%) as unchanged drug and metabolites, remainder in feces. Toxicity a. Dose limiting. Myelosuppression b. Occasional. Anorexia, nausea, vomiting, mucositis, sterility c. Rare. Alopecia, pruritus, rash, hypersensitivity; secondary malignancies (acute leukemia); pulmonary fibrosis, vasculitis, cataracts
- 15. Nitrogen mustard (mechlorethamine) Indication. HL; T-cell lymphoma Pharmacology a. M/A -alkylating agents b. Metabolism. In water or body fluids, mechlorethamine undergoes rapid chemical transformation ,drug is no longer present in active form a few minutes after administration. Metabolites are mostly excreted in urine. Toxicity a. Dose limiting. Myelosuppression b. Common. Severe nausea and vomiting; skin necrosis if extravasated; burning at IV injection site and facial flushing; metallic taste; discoloration of the infused vein; abnormal LFTs. c. Occasional. Alopecia, sterility, diarrhea, thrombophlebitis, gynecomastia d. Rare. Neurotoxicity (including hearing loss), angioedema, 2nd cancers
- 16. Carmustine [BCNU, bischlorethyl nitrosourea (BiCNU)] Indications. Brain tumors, myeloma, HL & NHL. In high doses for BMT. In the form of implantable wafers: GBM Pharmacology a. M/A. Forms interstrand cross-links in DNA preventing DNA replication and transcription. b. Metabolism. Highly lipid-soluble drug that enters the brain. Rapid spontaneous decomposition to active and inert product. Most of the intact drug and metabolic products are excreted in urine. Toxicity a. Dose limiting. Myelosuppression ,aggravated by concurrent RT. b. Common. Nausea and vomiting. Local pain during injection. c. Occasional. Stomatitis, esophagitis, diarrhea, LFT abnormalities; alopecia, facial flushing, brown discoloration of skin; interstitial lung disease with pulmonary fibrosis ,dizziness, optic neuritis, ataxia, organic brain syndrome; renal insufficiency d. Rare. 2nd cancer Lomustine [CCNU, cyclohexyl chlorethyl nitrosourea] Indications. Brain tumors & HL
- 17. Procarbazine Indications. HL Pharmacology a. M/A- cessation of protein synthesis. Direct DNA damage b. Metabolism. Hepatic metabolism and activation. Readily enters the CSF. Toxicity a. Dose limiting. Myelosuppression b. Common. Nausea and vomiting; flulike syndrome; sensitizes tissues to radiation; amenorrhea and azoospermia, sterility c. Occasional. Dermatitis, hyperpigmentation, photosensitivity; stomatitis, dysphagia, diarrhea; hypotension, tachycardia; urinary frequency, hematuria; gynecomastia d. Neurologic. disorders of consciousness or mild peripheral neuropathies (10%) ,sedation, depression, agitation, psychosis, decreased deep tendon reflexes, paresthesias, myalgias, and ataxia. e. Rare. Xerostomia, retinal hemorrhage, photophobia, papilledema; hypersensitivity pneumonitis, 2nd cancers
- 18. Streptozocin (streptozotocin) Indications. Islet cell cancer of the pancreas, carcinoid tumors Pharmacology a. M/A. Alkylating agent. A cell cycle–nonspecific nitrosourea analog. Inhibits DNA synthesis and the DNA repair enzyme, guanine-O6-methyltransferase; affects pyrimidine nucleotide metabolism and inhibits enzymes involved in gluconeogenesis. Selectively targets pancreatic β cells, presumably due to the glucose moiety on the molecule. b.Metabolism. Hepatic metabolism to active metabolites and has a short plasma half-life (<1 hour). Crosses the BBB. Excreted in urine as metabolites and unchanged drug Toxicity a. Dose limiting. Nephrotoxicity proteinuria ,glycosuria, aminoaciduria, proximal RTA, nephrogenic DI, and renal failure b. Common. Nausea and vomiting, myelosuppression, hypoglycemia after infusion, hypoglycemia or hyperglycemia c. Occasional. Diarrhea, abdominal cramps, LFT abnormalities d. Rare. CNS toxicity, fever, 2ND cancer.
- 19. Temozolomide Indications. Brain tumors; metastatic melanoma Pharmacology. similar to dacarbazine a. M/A. Activated to MTIC by nonenzymatic hydrolysis in tumors. Inhibits DNA, RNA, and protein synthesis but does not cross-link DNA strands. b. Metabolism. Renal excretion. Lipophilic crosses the BBB. Toxicity a. Dose limiting. Myelosuppression b. Common. nausea and vomiting, diarrhea, headache, fatigue c. Occasional. Photosensitivity, myalgias, fever d. Rare. Prolonged cytopenia, myelodysplastic syndrome (MDS)
- 20. Thiotepa (triethylenethiophosphoramide) Indications. Intracavitary for malignant effusions, intravesicular for urinary bladder, and intrathecal use for meningeal metastasis; severe thrombocytosis. Also can be used for breast and ovarian cancers and for HSCT Pharmacology. Ethylenimine analog, chemically related to nitrogen mustard a. M/A. Alkylates the N-7 position of guanine, which severs the linkage between the purine base and the sugar and liberates alkylated guanines. b. Metabolism. Rapidly decomposed in plasma and excreted in urine. Extensively metabolized by the hepatic P450 microsomal system to active and inactive metabolites Toxicity a. Dose limiting. Myelosuppression b. Common (for intravesicular administration). Chemical cystitis, abdominal pain, hematuria, dysuria, frequency, urgency, ureteral obstruction; nausea and vomiting 6 hours after treatment c. Occasional. GI upset, abnormal LFTs, hypersensitivity d. Rare. Alopecia, fever, angioedema, secondary malignancies
- 21. Trabectedin Indications. LS or LMS after an anthracycline containing regimen Pharmacology a. M/A. binds guanine residues in the minor groove of DNAadductsaffects binding of transcription factors and DNA repair. b. Metabolism. It is metabolized in the liver, and only negligible amount is excreted in urine. The drug is delivered as a 24-hour continuous infusion. Toxicity a. Dose limiting. Rhabdomyolysis (monitor the level of CPK before each dose), severe and fatal cardiomyopathy (ECG f/u), severe neutropenia (40%), febrile neutropenia (5%) b. Common. Elevation of liver enzymes, thrombocytopenia, anemia, nausea, fatigue, vomiting, constipation, decreased appetite, diarrhea, peripheral edema, dyspnea, and headache c. Rare. Liver failure, peripheral neuropathy
- 22. PLATINS- M/A--- • Enters the cells by diffusion and covalently binds to DNA N-7 position of guanine and adenine. • Reacts with two different sites on DNA to produce cross-links, either intrastrand (>90%) or interstrand (<5%). • Formation of DNA adducts results in inhibition of DNA synthesis and function as well as inhibition of transcription • not cell cycle specific. M/RESISTANCE--- • alterations in cellular transport. • Increased inactivation by glutathione and related enzymes. • Increased DNA repair enzymes activity • Deficiency in mismatch repair (MMR) enzymes
- 23. CISPLATIN • INDICATIONS • Testicular cancer • Ovarian cancer • Bladder cancer • Head and neck cancer • Esophageal cancer • Small cell and non-small cell lung cancer • Non-Hodgkin’s lymphoma • Trophoblastic neoplasms
- 24. From Peres’
- 25. DOSAGE RANGE • Ovarian cancer - 75mg/m2 IV on day 1 every 21 days as part of the cisplatin / paclitaxel regimen, and 100mg/m2 on day 1 every 21 days as part of cisplatin / cyclophosphamide regimen. • Testicular cancer - 20 mg/m2 IV on days 1-5 every 21 days as part of the BEP regimen. • Bladder cancer : administered as a single agent at a dose of 50-70 mg/m2 IV per cycle repeated every 4 weeks • Non - small cell lung cancer : 60-100 mg/m2 IV on day 1 every 21 days as part of the cisplatin / etoposide or cisplatin / gemcitabine regimens. • Head and neck cancer : 20 mg/m2/day IV continuous infusion * 4 days.
- 26. Toxicity a. Dose limiting 1) renal insufficiency- 5% with adequate hydration 25%-45% without 2) Peripheral sensory neuropathy >200 mg/m2 and can become dose limiting when the cumulative cisplatin dose exceeds 400 mg/m2. Symptoms may progress after treatment is discontinued and include loss of proprioception and vibratory senses, hyporeflexia. Symptoms may resolve slowly after many months. 3) Ototoxicity- tinnitus and high-frequency hearing loss (5%) >100 mg/m2 by rapid infusion or high cumulative doses. b. Common. Severe nausea and vomiting; preventative antiemetic regimens are required. ↓K+↓Mg++ mild myelosuppression alopecia; azoospermia, sterility, impotence. c. Occasional. Alopecia, loss of taste, vein irritation, transiently abnormal LFTs, SIADH, hypophosphatemia, myalgia, fever; optic neuritis d. Rare. Altered color perception and reversible focal encephalopathy that often causes cortical blindness. Raynaud phenomenon, bradycardia, bundle-branch block, congestive heart failure; anaphylaxis, tetany
- 27. Prevention of nephrotoxicity • Pre-hydration • Cisplatin dose < 50mg/m2 : N/Saline in 1L × 1 hour. If another drug is being given in the regimen e.g. etoposide then this would be used as prehydration i.e. etoposide in 1L N/Saline. • Cisplatin dose 50-75mg/m2 : N/Saline + KCl 20mmol/L in 1L × 1hr+ 100mls 10% mannitol (or equivalent) • Cisplatin dose > 75mg/m2 < 100mg/m2 : N/Saline + KCl 20mmol/L in 1L × 1hr + 200mls 10% mannitol (or equivalent) • Cisplatin should not be given if UOP is < 100ml/hr. • If urine output is insufficient, give 500ml N/Saline × 30mins. Consider giving further mannitol. Do not administer cisplatin until urine output is > 100ml/hr.
- 28. • Post-hydration • Cisplatin dose < 50mg/m2 : no post-hydration, recommend oral intake of 8 glasses of water. • Cisplatin dose > 50mg/m2 < 75mg/m2 : N/Saline + KCl 20mmol/L + MgSO4 10mmol/L in 1L × 1 hr. • Cisplatin dose > 75mg/m2 < 100mg/m2 : N/Saline + KCl 20mmol/L + MgSO4 10mmol/L in 1L × 1 hr x 2. Monitor BP as required
- 31. contraindications • History of hypersensitivity to cisplatin or other platinum containing compounds. • Pre-existing renal impairment, myelosuppressed patients or patients with hearing impairment. • Pregnancy, breastfeeding
- 32. CARBOPLATIN • INDICATIONS • Ovarian cancer • Germ cell tumors • Head and neck cancer • Small cell and non small cell lung cancer • Bladder cancer • Relapsed and refractory acute leukemia • Endometrial cancer Pharmacology a. M/A. Heavy metal alkylating- like agent with mechanisms very similar to cisplatin, but with different toxicity profile b. Metabolism. Plasma half-life of only 2 to 3 hours. Excreted in urine as unchanged drug (70%) and metabolites
- 33. Dosage range • Dose of carboplatin is usually calculated to a target area under the curve (AUC) based on the glomerular filtration rate (GFR). • Calvert formula is used to calculate dose – Total dose (mg) = ( target AUC ) x ( GFR + 25 ) • Note: dose is in mg, not mg/m2 • Target AUC is usually between 5 and 7 mg/mL/min for previously untreated patients. In previously treated patients , lower AUCs (between 4 and 6 mg/mL/min) are recommended . • AUCs >7 are not associated with improved response rates.
- 34. • CrCl + 25 =AUC • FOR WEEKLY DOSING ~2 AUC • FOR 3 WEEKLY DOSING 5-6 AUC OUR SET UP PRACTICE
- 35. Toxicity a. Dose limiting. Myelosuppressionespecially thrombocytopenia. b. Common. Nausea, vomiting, myalgias, weakness, and nephrotoxicity pain at injection site; cation electrolyte imbalance c. Occasional. Reversible abnormal LFTs, azotemia; peripheral neuropathy (5%), visual disturbance; hypersensitivity reactions; amenorrhea, azoospermia, impotence, and sterility d. Rare. Alopecia, rash, flulike syndrome, hematuria, hyperamylasemia; hearing loss, optic neuritis; alopecia C/I— • History of hypersensitivity to cisplatin or other platinum containing compounds. • Severe bone marrow depression or significant bleeding • Pregnancy category D; breastfeeding should be avoided
- 36. OXALIPLATIN • It is available as 50 mg/ 100 mg vials. • Reconstituted by using 10 ml (for 50 mg vial) and 20 ml (for 100 mg vial) of Water or 5% Dextrose injection. • It is further diluted in 250-500 ml of 5% Dextrose injection. • Reconstitution of final dilution must never be performed with NaCl solution or other chloride containing solutions. Indications. Colorectal, pancreatic, and gastric cancers Pharmacology a. Mechanisms. Binds covalently to DNA -intrastrand and interstrand cross-links. b. Metabolism. Undergoes extensive nonenzymatic conversion to its active cytotoxic species; >50% -kidneys. Only 2% - feces.
- 37. INDICATIONS • Metastatic colorectal cancer : FDA - approved in combination with infusional 5-FU/LV in patients with advanced, metastatic disease. • Early stage colon cancer : FDA - approved as adjuvant therapy in combination with infusional 5 –FU/LV in patients with stage III colon cancer and also effective in patients with high risk stage II disease. • Metastatic pancreatic cancer • Metastatic gastric cancer and gastroesophageal cancer • DOSAGE-- • Recommended dose is 85 mg/m2 IV over 2 hours, on an every 2 weeks schedule. • Can also administer 100-130 mg/m2 IV on an every 3 - weeks schedule.
- 38. Oxaliplatin Toxicity a. Dose limiting (1) Acute dysesthesias in the hands, feet, perioral area, or throat develop within hrs or up to 2 days after dosing and may be precipitated or exacerbated by exposure to cold -usually resolves within 2 weeks-ameliorated by prolonging the infusion to 6 hrs. (2) Persistent peripheral sensory neuropathy usually characterized by paresthesias, dysesthesias, and hypesthesia, including deficits in proprioception, which is usually reversible within 4 mths of discontinuing oxaliplatin. b. Common. Anorexia, nausea, vomiting, constipation, diarrhea, • abdominal pain; fever, fatigue; mild to moderate myelosuppression; mild to moderate LFT abnormalities c. Occasional. Allergic reactions, mild nephrotoxicity, headache, stomatitis, taste alteration; back pain, arthralgias d. Rare. Pulmonary fibrosis
- 39. contraindications • Known history of hypersensitivity to oxaliplatin • Pregnant • Breast feeding • Myelosuppression • Peripheral sensory neuropathy with functional impairment prior to first course • Severely impaired renal function
- 40. Toxicity profiles of platinum analogs in use TOXICITY CISPLATIN CARBOPLATIN OXALIPLATIN MYELOSUPPRESSION + NEPHROTOXICITY + NEUROTOXICITY + + OTOTOXICITY + NAUSEA AND VOMITTING + + +
- 41. ANTI-METABOLITE CLASSIFIACTION •ANTIFOLATE • METHOTRAXATE • PEMETREXED • PRALATREXATE •FLUOROPYRIMIDINES • 5-FU • CAPACITABINE •DEOXYCYTIDINE ANALOGS • CYTARABINE • GEMCITABINE •PURINE ANALOGS • 6-MP • 6-TG • FLUDARABINE • CLADRIBINE • CLOFARABINE
- 42. ANTIMETABOLITES • General pharmacology of antimetabolites 1. structure resemblance to purine or pyrimidine precursors or because they interfere with purine or pyrimidine synthesis. 2. Greatest activity in the S phase 3. most effective when cell proliferation is rapid. Azacitidine Indication. Myelodysplastic syndromes (MDS) Pharmacology a. M/A-cytidine analogincorporated into DNA and RNAinhibiting protein synthesis; also inhibits pyrimidine synthesis and DNA methylation. Toxicity a. Dose limiting. Myelosuppression; nausea and vomiting. b. Common. Hepatic dysfunction, fatigue, headache, diarrhea, alopecia, fever, injection site erythema c. Occasional. Neurotoxicity ,azotemia, arthralgias, hypophosphatemia with myalgia, stomatitis, phlebitis, rash d. Rare. Progressive lethargy and coma, renal tubular acidosis, rhabdomyolysis, hypotension
- 43. Cladribine Indications. Hairy cell leukemia Pharmacology. a. M/A. An analog of the purine deoxyadenosine. Inhibits ribonucleotide reductase. Depletes ATP. Induces apoptosis. Active against both dividing and resting cells Toxicity. Patients are at increased risk for opportunistic infections. a. Dose limiting. Myelosuppression b. Common. Immunosuppression with decreases in CD4+ and CD8+ cells; nausea, skin reactions at injection site; fever , chills, flulike syndrome c. Occasional. Neurotoxicity, hypersensitivity reactions, fatigue d. Rare. Severe neurotoxicity, pancreatitis
- 44. Clofarabine Indications. Relapsed or refractory ALL Pharmacology. Purine antimetabolite Toxicity a. Dose limiting. (1) Capillary leak syndrome (CLS)/SIRS –due to cytokine release (2) Hematosuppression (90%) (3) Hepatotoxicity and nephrotoxicity b. Common. Tachycardia, hypotension, flushing; headache, fever, chills, fatigue; pruritus, rash; nausea, vomiting, diarrhea; abnormal LFTs (80%); increased creatinine (50%), limb pain c. Occasional. Hypertension, edema, dyspnea, pleural, or pericardial effusion; mucositis; myalgia, arthralgia; irritability, somnolence, agitation; cecitis; CLS (4%), SIRS (2%) d. Rare. Hepatic venoocclusive disease, Stevens-Johnson syndrome, hallucination
- 45. Cytarabine Indications. Acute leukemia, lymphoma, meningeal involvement with tumor Pharmacology. An analog of deoxycytidine a. M/A. Antimetabolite.Intracellular activation ara-CTPinhibits DNA polymerases; some are incorporated into DNA. Ara-CTP inhibits ribonucleotide reductase DNA synthesis and function. b. Metabolism. In patients with renal insufficiencyhigh concentrations of ara- CTP, which may result in CNS toxicity. Toxicity a. Dose limiting. Myelosuppression b. Common. Nausea, vomiting, mucositis, diarrhea; conjunctivitis; hydradenitis, arachnoiditis with intrathecal administration c. Neurotoxicity (cerebellar ataxia, lethargy, confusion) d. Occasional. Alopecia, stomatitis, metallic taste, esophagitis, hepatic dysfunction, pancreatitis, severe GI ulceration; thrombophlebitis; headache; rash, transient skin erythema without exfoliation.
- 46. Decitabine Indications. MDS Pharmacology. analogue of 2′-decoxycytidine. a. M/A- inhibits DNA methyltransferase, causing hypomethylation of DNA and cellular differentiation or apoptosis. Toxicity a. Dose limiting. Hematosuppression b. Common. Hematosuppression, fatigue, fever; nausea, constipation (35%), diarrhea; headache, arthralgias, rigors, edema, cough; hyperglycemia, ↓K+, ↓ Mg++
- 47. Fludarabine Indications. CLL, low-grade lymphomas Pharmacology. analog of ara-A (arabinofuranosyladenosine). drug is resistant to adenosine deaminase activity(compare with cytarabine). high specificity for lymphoid cells. It has activity against both dividing and resting cells and induces apoptosis. Toxicity a. Dose limiting. Myelosuppression; AIHA b. Common. Immunosuppression -risk for opportunistic infections; mild nausea and vomiting; fever; cough, weakness, arthralgia/myalgias c. Occasional. Alopecia (mild), abnormal LFTs, tumor lysis syndrome
- 48. 5-Fluorouracil A fluoropyrimidine analog M/A- blocking thymidylate synthetase (TS)interfere with DNA syn Incorporation of another metabolite (FdUTP) into DNA results in inhibition of DNA synthesis and function. It is cell–cycle S-phase specific but acts in other cell cycle phases as well. 5-FU rapidly enters all tissues, including spinal fluid and malignant effusions. Toxicity is more common and more severe in patients with dihydropyrimidine dehydrogenase deficiency.
- 51. TOXICITY a. Dose limiting. Myelosuppression; mucositis; diarrhea b. Common. Nasal discharge; eye irritation and excessive lacrimation due to dacryocystitis and lacrimal duct stenosis; dry skin, photosensitivity, and pigmentation of the infused vein c. Neurologic. Reversible cerebellar dysfunction, somnolence, confusion, or seizures occurs in about 1% of patients. d. Occasional. Esophagitis; hand–foot syndrome; coronary vasospasm; thrombophlebitis; nausea, vomiting e. Rare. Alopecia, dermatitis, loss of nails, dark bands on nails; blurred vision, “black hairy tongue” (hypertrophy of filiform papillae), anaphylaxis, fever
- 52. Capecitabine Indications. breast or colon ca Pharmacology. Capecitabine is a fluoropyrimidine carbamate that is a systemic prodrug of 5′-deoxy-5-fluorouridine (5′-DFUR), which is converted in vivo to 5-FU. M/A & metabolism & toxicity- same as 5-FU. Toxicity. a. Dose limiting. Diarrhea (50%), hand–foot syndrome b. Common. Hand–foot syndrome -15% to 50%; nausea, vomiting, hematosuppression; fatigue c. Occasional. Abnormal LFTs, neurotoxicity; cardiac ischemia in patients with a prior history of coronary artery disease; tear duct stenosis, conjunctivitis, blepharitis; confusion, cerebellar ataxia
- 55. Gemcitabine Indications. Carcinoma of pancreas, bladder, lung, ovary; STS Pharmacology. A fluorine-substituted deoxycytidine analog a. M/A. Cell-phase specific-S phase -blocking the progression of cells through the G1-S-phase also. Inhibits ribonucleotide reductase; competes with deoxycytidine triphosphate (dCTP) for incorporation into DNA Toxicity a. Dose limiting. Myelosuppression b. Common. Nausea, vomiting, diarrhea, stomatitis; fever with flulike symptoms (40%); macular or maculopapular rash; transient LFT elevations; mild proteinuria and hematuria c. Occasional. Hair loss, rash, edema d. Rare. Hemolytic–uremic syndrome; pulmonary drug toxicity; hypersensitivity reactions; alopecia
- 57. Hydroxyurea Indications. MPD, refractory ca ovary, sickle cell disease Pharmacology. An analog of urea a. M/A. Antimetabolite. inhibiting nucleotide reductaseinterfere with DNA syn Inhibits DNA repair and thymidine incorporation into DNA. Cell cycle S-phase specific but acts in other phases as well b. Metabolism. Crosses the BBB. Half of the drug is rapidly degraded into inactive compounds by the liver. Inactive products and unchanged drug (50%) are excreted in urine. Toxicity a. Dose limiting. Myelosuppression b. Occasional. Nausea, vomiting, diarrhea; skin rash, facial erythema, hyperpigmentation; azotemia, proteinuria; transient LFT abnormalities; c. Rare. Alopecia, mucositis, diarrhea, constipation; neurologic events; pulmonary edema; flulike syndrome; painful perimalleolar ulcers; possible acute leukemia in myeloproliferative disorders
- 58. 6-Mercaptopurine Indication. Acute lymphoblastic leukemia (maintenance therapy) a. M/A- Purine analog The parent drug is inactive. Requires intracellular phosphorylation – HGPRTase b. Metabolism-by xanthine oxidase. Allopurinol- XO inhibitortoxicity. Toxicity a. Dose limiting. Myelosuppression b. Common. Mild nausea, vomiting, anorexia (25%); usually reversible cholestasis (30%); dry skin, photosensitivity; immunosuppression c. Rare. Stomatitis, diarrhea, dermatitis, fever, hematuria, BuddChiari– like syndrome, hepatic necrosis
- 59. Methotrexate Indications. A wide variety of conditions a.M/A- interfere with DNA syn. The drug also inhibits RNA and protein synthesis and prevents cells from entering the S phase of the cell cycle. b. Metabolism. Renal dysfunction results in dangerous blood levels of MTX and possible further renal damage.
- 60. Toxicity. Leucovorin can reverse the immediate cytotoxic effects of MTX; generally, 1 mg of leucovorin is given for each 1 mg of MTX. a. Dose limiting. Myelosuppression, stomatitis, renal dysfunction b. High-dose regimens. Nausea, vomiting, renal tubular necrosis, cortical blindness c. Skin erythema, pulmonary fibrosis, transverse myelitis, cerebritis d. Chronic therapy. Liver cirrhosis; osteoporosis e. Neurotoxicity. acute aseptic meningitis,subacute encephalopathy and myelopathy. irreversible leukoencephalopathy f. Occasional. Nausea, vomiting, dermatitis, photosensitivity, altered pigmentation, furunculosis; conjunctivitis, photophobia, excessive lacrimation, cataracts; fever, reversible oligospermia, flank pain g. Rare. Alopecia, MTX pneumonitis
- 61. Pemetrexed Indications. Mesothelioma (with cisplatin) & NSCLC a. M/A. antifolate analog activity in the S phase of the cell cycle. Inhibition of TS & DHFRase. b. Metabolism- cleared by the kidneys. About 90% of the drug is excreted unchanged in the urine within 24 hours. Toxicity. a. Dose limiting. Myelosuppression. All patients are given 350 μg/d PO of folic acid and 1,000 mg of vitamin B12 SC every 3 weeks to reduce drug toxicity. b. Common. Skin rash (usually as the hand–foot syndrome), mucositis, nausea, vomiting, diarrhea; mild dyspnea, fatigue; transient elevation of LFTs c. Occasional. Myalgia/arthralgia, fever
- 62. Pentostatin Indications. CLL, hairy cell leukemia, and cutaneous T-cell lymphoma Pharmacology. A fermentation product of Streptomyces antibioticus a. M/A- Antimetabolite. Both cell cycle specific and cell cycle nonspecific. Inhibitor of adenine deaminase., ribonucleotide reductase b. Metabolism. excreted unchanged in urine. Toxicity a. Dose limiting. Myelosuppression b. Common. Immunosuppression; mild nausea and vomiting, diarrhea, altered taste; fatigue, fever; erythematous, papular, vesiculobullous rashes c. Occasional. Chills, myalgia, arthralgia; abnormal LFTs; keratoconjunctivitis, photophobia; cough, renal failure
- 63. Pralatrexate Indications. Relapsed or refractory peripheral T-cell lymphoma Pharmacology: An antineoplastic folate analog a. M/A. Competitively inhibits DHFRase and polyglutamylation by the enzyme folylpolyglutamyl synthetase b. Metabolism. Approximately 33% of the drug is excreted unchanged in the urine. Toxicity a. Dose limiting. Thrombocytopenia, neutropenia, and mucositis b. Common. Anorexia, nausea, vomiting, diarrhea, constipation; fatigue, fever, edema; rash d. Rare. Hepatitis; pulmonary infiltrates and insufficiency
- 64. 6-Thioguanine Indication. AML a. M/A. Purine analog with cell cycle–specific activity in the S phase. The drug requires intracellular phosphorylation by HGPRT The drug is incorporated extensively into DNA, resulting in miscoding of transcription and DNA replication, and into RNA. b. Metabolism. can be given in full doses with allopurinol. Clearance of the drug is primarily hepatic, but also renal. Toxicity a. Dose limiting. Myelosuppression b. Common. Stomatitis, diarrhea c. Occasional. Nausea and vomiting, hepatic dysfunction, hepatic venoocclusive disease; decreased vibratory sensation, unsteady gait
