Nora
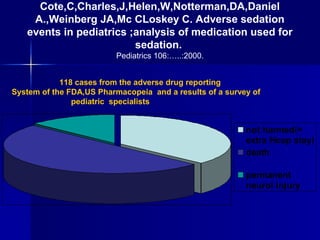
- 1. Cote,C,Charles,J,Helen,W,Notterman,DA,Daniel A.,Weinberg JA,Mc CLoskey C. Adverse sedation events in pediatrics ;analysis of medication used for sedation. Pediatrics 106:…..:2000. 118 cases from the adverse drug reporting System of the FDA,US Pharmacopeia and a results of a survey of pediatric specialists not harmed(+ extra Hosp stay) death permanent neurol injury
- 2. Allocation of medication related adverse events drug interaction drug overdose premature discharge prescr/transcripti on error idadeq understanding of adm.medications adm by unsupervised
- 3. Relationship of interest of negative outcome • • • • No with general category of drug No with route of administration Yes with 3 or > sedation medications 12 pts suffered at home or in auto;chloral hydrate most frequently involved • Dental specialists overrepresented:39%!
- 4. Sedation and general anaesthesia in children undergoing MRI and CT: adverse events and outcomes. Malvija S. et al. Br. J. Anaesth. 84:743-8, 2000 • RCT • MRI and CT (01/ ’97-01/ ’98 from 0 to 18 yr) • University of Michigan Health Care Systems • Sedation (922) / GA (140) Sedation/Age Not Adequate sedation/ASA 6 30% 4 20% 2 10% 0% ASA 1-2 ASA 3-4 0 Adequate Not Adequate Sedation
- 5. Sedative agents and adverse events Sedative Mean Dosag e (SD) Oxygen Airway (=9) Inadequat e sedation (=146) Oversedatio n (=4) desaturation management (=27) 69 (9.9) mg/kg Failed 21 (3%) 7 (1%) 63 (9%) 4 (<!%) 26 (4%) 1 (1%) 0 17 (19%) 0 8 (9%) 0 0 0 0 0 2 (2%) 1 (<1%) 59 (50%) 0 28 (24%) 1 (14%) 0 1 (14%) 0 0 procedure s (=65) Single agent • Chloral Hydrate (679) • BDZ (90) • Barbiturate (2) Multiple agents • Anxiolytic combination (117) • Analgesicanxiolytic combination (7) Malvija S. et al. Br. J. Anaesth. 84:743-8, 2000
- 6. Sedation procedures in MRI imaging: safety, effectiveness, and nursing effect on examinations. Bluemke D. et al. Radiology 216:645-52, 2000 • • John Hopkins Hospital, Baltimore 1991-1998 6.093 patients, 4.761 (78,1%) received conscious sedation by the MR conscious sedation service • Complications: 20 patients of 4.761 (0,42%), no death. Age range 400 300 164 N° 200 195 183 84 125 84 Dose Indication 0-3 325 Medication Chloral Hydrate 80-100 mg/kg Sedativehypnotic Sedativehypnotic 3-7 Pentobarbital 4-6 mg/kg 7-18 Diazepam 0.2 mg/kg Antianxietysedative >18 Alprazolam 0.5 mg/23kg Antianxietysedative 100 0 10-20 20-30 30-40 40-50 Age 50-60 60-70 70-100
- 7. Sedation procedures in MRI imaging: safety, effectiveness, and nursing effect on examinations. Bluemke D. et al. Radiology 216:645-52, 2000 2500 CH: Chloral hydrate 4,8% D: Diazepam 2000 P: Pentobarbital 4,9% A: Alprazolam 1500 1000 O: Other M: Midazolam 2081 13,1% 1498 500 0 6,2% 588 D i.v. D os 16% 497 48 CH Failed sedation N° pts 9,4% P A 49 O Adverse Event during Conscious Sedation N° patients 6 Agitation 5 Bronchospasm 4 Congestion and coughing Hypotension 3 2 Desaturation 1 Seizure 0 CH M P Vomiting • Complete MR examination: 93,5% • Mean time to sedate patients: • 23,6 ± 15,2 min for specialized MR nurses • 26,8 ± 20,1 min for general radiology nurses (p<0.001)
- 8. MacIntyre PA,Sury MRI.Is propofol infusion better than inhalational anesthesia for paediatric MRI? Anaesthesia 51;517,1996 43 children 6 difficult veins 3 intrascan airway manipulations 3 abandonement of technique Haloth induction SaO2 88-89% in 6 9 repetition of scan sequences Induction with propofol 1-3 mg/kg Maint:propofol 5 mg/kg/h 30 required additional boluses of propofol 1 mg/kg; In 5, 2-3 mg/kg Recovery < 3 min,but in 9 >10 min
- 9. Vangerven M.,Van Hemelriick J,Wouters P,Vandermeersch E,Van Aken,H.Light anesthesia with propofol for paediatric MRI Anaesthesia 47;706-7,1992 • 20 children,1 mo-12 years from neuropediatrics • Atropine syrup 0.02 mg/kg • Propofol induct 1 + 8 mg/kg/hr
- 10. Vangerven M.,Van Hemelriick J,Wouters P,Vandermeersch E,Van Aken,H.Light anesthesia with propofol for paediatric MRI Anaesthesia 47;706-7,1992 • 7 SaO2 99% 6 5 4 PECO2 additional boluses mean tot prop dose 3 2 1 0 3 10 20 min 30
- 11. Initial experience with i.v. pentobarbital sedation for children undergiong MRI at a tertiary care pediatric hospital: the learning curve Greenberg et al. Pediatr Radiol 30:689-691, 2000 • • • 100 children in MRI (ASA I-II); 2-15 ys with mean age of 6,8 ys Weight from 11,4 to 70 kg with a mean of 28 kg Max 6 mg/kg pentobarbital in 3 divided doses with total dose ≤ 200 mg administrated by radiologists (PALS certificated) 100 8 80 3 > 12 y old* 60 40 92 3 > 50 kg* (*p < 0.05) 20 0 Failed Successful Sedation
- 12. Initial experience with i.v. pentobarbital sedation for children undergoing MRI at a tertiary care pediatric hospital: the learning curve Greenberg et al. Pediatr Radiol 30:689-691, 2000 Number of cases Adverse reaction to Pentobarbital in 100 children 10 9 8 7 6 5 4 3 2 1 0 Hyperreaction Desaturation 7 6 Cough 3 2 1 Adverse reaction 1 Prolonged sedation Nasal congestion Vomiting
- 13. Intravenous sedation for MR Imaging of the brain and spine in children: Pentobarbital vs Propofol Bloomfield E.L. et al. Radiology 186:93-97, 1993 • • • RCT from April 1991 to February 1992 (Cleveland Clinic Foundation) 30 pts received i.v. Pentobarbital 2,5 mg/kg (to a max of 7,5 mg/kg) by radiologists 31 pts received i.v. Propofol 1-2 mg/kg + 6-10 mg/kg/h (premed. 0.05/12 Kg glycopyrrolate) first 10 by anesthesiologists then by radiologists (Control- A-Flow Regulator Extension Set, Baxter) Group Data Age (y) (mean/range) Sex (M/F) Weight (kg) (mean/range) Original diagnoses: • Seizures • Neurofibromatosis I • Developmental delay • Cerebral palsy • Dysmyelination syndrome • Spinal dysraphism • Chiari malformation • Brain tumor • Infarct • Diskitis • Headache Propofol (n=31) Pentobarbital (n=30) 5 / 2 – 11 15 / 16 20 / 11.5-34.0 4/2–8 18 / 12 17.9 / 9-50 10 2 3 4 0 0 2 9 0 0 1 11 4 3 2 2 2 2 1 1 1 1
- 14. Intravenous sedation for MR Imaging of the brain and spine in children: Pentobarbital vs Propofol Bloomfield E.L. et al. Radiology 186:93-97, 1993 120 p=0,05 100 80 Pulse SpO2 MAP RR 60 40 20 0 Mean t=0 % drop % rise Mean Mean % drop % rise Propofol Mean t=0 % drop % rise Mean Mean % drop % rise Pentobarbital
- 15. Intravenous sedation for MR Imaging of the brain and spine in children: Pentobarbital vs Propofol Bloomfield E.L. et al. Radiology 186:93-97, 1993 50 45 Pentobarbital Propofol Time (min) 40 35 30 34 25 20 15 21,5 10 5 0 12,5 5 Arousal Discharge P=0.0005 P=0.0001
- 16. Anaesthesia for magnetic resonance imaging: a survey of current practice in the UK and Ireland McBrien M. E. et al. Anaesthesia 55:737-743, 2000 • Postal questionnaire was sent to 120 MRI units in UK and 6 in Republic of Ireland 100 (79%) responses: • – 46 units had an anaesthetic service (36 units on a regular basis, 10 on demand) Sedation ever provided by nonanaesthetic personnel Sedation provided by anaesthetic consultants 12 25 10 20 6 N° Units N° Units 8 12 10 21 8 4 15 5 2 0 15 5 0 Radiologists Paediatricians Both 9 GAA GPA NAP 7 NA GAA: General Adult Anaesthetist GPA: General Paediatric Anaesthetist NAP: Neuroanaesthetist with regular Paediatric practice NA: Neuroanaesthetist with no regular paediatric practice
- 17. Patient demographics Preoperative assesment 30 25 25 20 15 N° Units N° Units 20 28 15 10 23 10 5 0 2 Only Adults 9 Mainly Adults 6 Only Children 1 13 5 8 2 Mainly Not Children answered 0 A B C D A: Only at arrival on scan B: On ward prior to scan C: Seen at assesment clinic prior to scan D: Questionnaire returned by referring clinician • • • Written consent for anaesthesia was obtained in 40 units but only 20 units had written information about anaesthesia 44 of 46 units providing anaesthesia always had anaesthetic assistance, 2 had occasional assistance Remote monitoring in 35 units McBrien M. E. et al. Anaesthesia 55:737-743, 2000
- 18. Ventilator Anaesthetic machine 2 2 2 15 27 A B C D A: MR-compatible anaesthetic machine inside the scanning room B: Long breathing system tubing fed into the scanning room from a remote anaesthetic machine C: Non compatible gas delivery system bolted to the wall inside D: Nothing (only sedation and never GA) Ventilation 12 15 A B C A: remote ventilator in the control room with circuit tubing fed into SR B: Non compatible ventilator attached to the wall inside C: Ambu bags attached to an oxygen supply if ventilator was required • 15 units had not scavenging of gases in the induction area 1 16 • 13 units had not scavenging of gases in the scanning room 29 SB IPPV Both • 18 units had a separate recovery room equipped with NIBP, SpO2, ECG McBrien M. E. et al. Anaesthesia 55:737-743, 2000
- 19. Adverse events and factor associated with the sedation of children by nonanesthesiologists Shobha Malviya et al. Anaesth Analg 85:1207-13,1997 • • • • • • • • • • • University of Michigan Hospital, october 1995-1996 1.140 children mean age (2,96±3,7) ASA I-II 848 (81%) ASA III-IV 199 (19%) MRI 48% TC 27% Cardiac catheterization 2% Echocardiogram 20% Other 3% Conscious sedation 604, deep sedation 294
- 20. Sedation Respiratory events Inadequate Oversedation Failed procedures 46 (5,3%) 77 (10%) 11 (1,3%) 26 (3%) 0 14 (19%) 0 4 (5%) Anxiolytic combinatios (123) 3 (2,4%) 39 (32%)* 3 (2,4%) 16 (13%)* Analgesic / anxiolytic combinations (24) 2 (8,3%) 0 0 0 Opioid alone 2 (100%) 1 (50%) 1 (50%) 2 (100%) Diphenhydramine alone (2) 2 (100%) 0 0 0 Sedative (n) Chloral hydrate (854) BDZ (72) * All included the use of chloral hydrate Shobha Malviya et al. Anaesth Analg 85:1207-13,1997
- 21. Relationship of age to the incidence of adverse events in sedated children. *P<0.05 100 % Children 80 All events Respiratory events 60 * 40 20 0 ASA I-II * ASA III-IV Shobha Malviya et al. Anaesth Analg 85:1207-13,1997
- 22. Relationship of age to the incidence of adverse events in sedated children. *P<0.05 **P<0.0001 100 All events Respiratory events % Children 80 60 40 20 0 * <1 mo ** 1-12 mo 13-24 mo 25 mo-12 yr >12-18 yr Shobha Malviya et al. Anaesth Analg 85:1207-13,1997
- 23. Summary • Respiratory depression: – 5,5% experienced hypoxemia, 2 became apneic and required resuscitation. – Several reports of mild hypoxemia and airway obstruction requiring IOT in children who received chloral hydrate as a sole sedative for CT scan. – Chloral hydrate may selectively depress genioglossus activity >>>> airway obstruction – In this study, chloral hydrate in recommended doses (38-83 mg/kg) as a sole drug is associated with a significant risk of oxygen desaturation
- 24. Concluding • Importance of diligent monitoring throughout the sedation experience until the child has met discharge criteria, regardless of the sedative administered or its route of administration >>>> early detection and early intervention • Cotè reported an anecdotal incident of death from the practice of administering chloral hydrate to an infant at home before arrival in the hospital • Inadequate sedation with chloral hydrate (5-15%) resulted in either a prolonged or a failed procedure. • A larger portion of children who had received combinations of sedatives that included chloral hydrate experienced inadequate sedation compared with those who had received chloral hydrate alone. • The individual response to chloral hydrate is varied and unpredictable.
- 25. • Older children had a greater failure rate (15%) compared with younger children (5%) for chloral hydrate sedation. • In older children, it may be preferable to consider alternative sedation regimens that combine chloral hydrate with another sedative. • Prolonged or aborted procedures may be costly to the institution, as well as inconvenient for the patient, who may be required to return for a repeated procedure with general anesthesia. • Neonates and infants less than one year old, and ASA III-IV all age, are at greater risk of adverse events related to sedation.
- 26. Chloral hydrate toxicity • Chloral hydrate is a reactive metabolite of trichloroethylene, an industrial solvent. Is it responsible for the carcinogenicity of trichloroethylene? • The carcinogenicity of richloroethylene is due to a reactive intermediate epoxide metabolite. • Trichloroethylene is carcinogenic in some laboratory animal species but not in others. • Multiple epidemiologic studies in humans have failed to document an increase in cancer incidence.
- 27. Randomised double-blind clinical trial of intermediate vs high-dose chloral hydrate for neuroimaging of children Martì-Bonmatì et al. Neuroradiology 1995;37:687-691 • Background: – Recommended paediatric dose is 50 mg/kg orally. – Previous study shown a successfull of sedation in 90% of cases with 70 mg/kg orally. – Higher doses did not seem justified (risk of prolonged sedation, rising of adverse reaction) – Some studies have concluded that 100 mg/kg provides effective and safe sedation (Greenberg 1993).
- 28. Randomised double-blind clinical trial of intermediate vs high-dose chloral hydrate for neuroimaging of children Martì-Bonmatì et al. Neuroradiology 1995;37:687-691 • Materials and methods: – – – – 97 children undergoing MRI (Valencia University) Mean age 38 ± 31 month Mean weight 14,7 ± 6,4 kg Randomized (double-blind) in two groups: • A Group: 70 mg/kg of orally syrup 30 min before scanning • B Group: 100 mg/kg of orally syrup 30 min before scanning – O2 supplementation during exam – Indications: • Congenital craniocerebral abnormalities (33) • Epilepsy (21) • White matter disease (11) • Development delay (11) • Congenital spine disease (10) • Cerebral-spinal tumor (11)
- 29. Percentage of effectiveness after the initial dose and the total dose Initial dose Total dose p < 0,05 % Successful examination 100 92 100 87 80 64 60 40 20 0 Group A Group B (70 mg/kg) (100 mg/kg) Martì-Bonmatì et al. Neuroradiology 1995;37:687-691
- 30. Degree of of acceptance of the two syrups by the children Bad Medium Good Degree of acceptance % 60 Chi-squared: p < 0,05 55 50 43 40 30 28,5 28,5 30 20 15 10 0 70 mg/ml 100 mg/kg Martì-Bonmatì et al. Neuroradiology 1995;37:687-691
- 31. Initial and the total dose in patients with failed and successful axamination Failure Successful examination 90 p < 0,01 p < 0,05 Dose (mg/kg) 80 70 60 50 Initial dose Total dose Martì-Bonmatì et al. Neuroradiology 1995;37:687-691
- 32. Adverse reactions (21%) was similar in both groups A-B Vomiting Excitement Nausea Stomach pain 5% 5% 10% 80% Martì-Bonmatì et al. Neuroradiology 1995;37:687-691
- 33. Anaesthesia with midazolam and S-(+)-ketamine in spontaneously breathing paediatric patients during MRI Haeseler G, et al. Paediatric Anaesthesia 2000;10:513-519 • RCT study • Department of Anaesthesiology, Hannover Medical School • MRI from april 1998 to march 1999 Materials and methods: • Prospective study of 34 patients (8 months – 7 years) undergoing MRI – Randomized in two groups: • 1st Group: Premedication with 0,5 mg/kg of rectal midazolam Induction with 3-5 mg/kg of methohexital, 0,5 mg/kg atracurium IOT and IPPV (FiO2 0,4, End tidal Isoflurane 1 ± 0,3%)
- 34. •2nd Group: premedication with 0,5 mg/kg of rectal midazolam and 5 mg/kg of S-(+)ketamine O2 supply 2-3 L/min by two nasal prongs Continuous nasopharyngeal EtCO2 monitoring 0,05 mg/kg midazolam and 0,25 mg/kg S-(+)-ketamine until adequate immobilization All patients received 0,01 mg/kg atropine i.v. and 0,05% xylometazoline nasal Standard operating room monitoring Haeseler G, et al. Paediatric Anaesthesia 2000;10:513-519
- 35. P E tC O du in g 120 at ur at io n du r 130 D es S pO 2 a) g (k P ri n 0 be fo re ri ng g fo re du be ri n fo re du be 10 S pO 2 2 N IB P N IB P H R H R Results * P < 0,05 140 Sedation G.A. 110 100 90 80 70 60 50 40 30 20 *
- 36. na es th es io lo gi st i n ch ar ge re co ve ry * A R Sedation G.A. Diff.means Co m pl er e R 100 to tim e 120 Di sc ha rg e Sc an ni ng rt 20 st a tim e 40 De la y In du ct io n Time (min) 140 * P < 0,05 * 80 60 * 0 -20 -40
- 37. S-(+)-ketamine Clinical superiority in anaesthetic potency compared to its optical enantiomer R-(-)-ketamine Superiority in recovery time Superiority in neuroprotective effects Shorter induction and discharge times
- 38. THANK YOU FOR YOUR ATTENTION
- 39. • D:Anesth outside OR Gurman • D:bis e tci modena • D:propofol tci e bis monitoring dentro bis monit and consciousness • RMN • Propofol tci e anest reg modena;il + completo per la metodica……………. • Modena 12403komplet • Macmida per le scale di sedaz