Aminoglycoside SAR one page.doc
•
0 likes•144 views
Aminoglycoside SAR one page
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Chemistry of Anti Anginal Drugs by Professor Beubenz

Chemistry of Anti Anginal Drugs by Professor Beubenz
Penicillins by Dr. Panchumarthy Ravisankar M.Pharm., Ph.D.

Penicillins by Dr. Panchumarthy Ravisankar M.Pharm., Ph.D.
Aminoglycosides(medicinal chemistry by p.ravisankar)

Aminoglycosides(medicinal chemistry by p.ravisankar)
Sulphonamides (Sulfonamides) and Sulfones || B.Pharm VI Semester || Medicinal...

Sulphonamides (Sulfonamides) and Sulfones || B.Pharm VI Semester || Medicinal...
Chloramphenicol: Unclassified Antibiotics-Shahare HV

Chloramphenicol: Unclassified Antibiotics-Shahare HV
Similar to Aminoglycoside SAR one page.doc
PROTECTING GROUPS (PROTECTION FOR THE CARBOXYLIC GROUP & PROTECTION FOR THE AMINO GROUPProtection for carboxylic group & Protection for the Amino group

Protection for carboxylic group & Protection for the Amino groupCollege of Pharmacy,Sri Ramakrishna Institute of Paramedical Sciences,Coimbatore
Similar to Aminoglycoside SAR one page.doc (7)
Protection for carboxylic group & Protection for the Amino group

Protection for carboxylic group & Protection for the Amino group
More from Mahavir Ghante
More from Mahavir Ghante (12)
Neuroglia, nerve fiber, action potential, synapse, neurotransmitters.ppt

Neuroglia, nerve fiber, action potential, synapse, neurotransmitters.ppt
Recently uploaded
☑️░ 9630942363 ░ CALL GIRLS ░ VIP ░ ESCORT ░ SERVICES ░ AGENCY ░
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...

Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...GENUINE ESCORT AGENCY
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In Hyderabad Escorts Service
Escorts Service Available
Whatsapp Chaya ☎️ : [+91-9352852248 ]
Escorts Service Hyderabad are always ready to make their clients happy. Their exotic looks and sexy personalities are sure to turn heads. You can enjoy with them, including massages and erotic encounters.#P12Our area Escorts are young and sexy, so you can expect to have an exotic time with them. They are trained to satiate your naughty nerves and they can handle anything that you want. They are also intelligent, so they know how to make you feel comfortable and relaxed
SERVICE ✅ ❣️
⭐➡️HOT & SEXY MODELS // COLLEGE GIRLS HOUSE WIFE RUSSIAN , AIR HOSTES ,VIP MODELS .
AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME
★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE
★
SATISFACTION,UNLIMITED ENJOYMENT.
★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE PROFILes Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & HOTEL Star Hotel Service .In Call & Out call
SeRvIcEs :
★ A-Level (star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without condom
★ANAL SERVICES.
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...

🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
☑️░ 9630942363 ░ CALL GIRLS ░ VIP ░ ESCORT ░ SERVICES ░ AGENCY ░
9630942363 THE GENUINE ESCORT AGENCY VIP LUXURY CALL GIRLS
HIGH CLASS MODELS CALL GIRLS GENUINE ESCORT BOOK
BOOK APPOINTMENT - 9630942363 THE GENUINE ESCORT AGENCY
BEST VIP CALL GIRLS & ESCORTS SERVICE 9630942363 VIP CALL GIRLS ALL TYPE WOMEN AVAILABLE
INCALL & OUTCALL BOTH AVAILABLE BOOK NOW
9630942363 VIP GENUINE INDEPENDENT ESCORT AGENCY
VIP PRIVATE AUNTIES
BEAUTIFUL LOOKING HOT AND SEXT GIRLS AND PARTY TYPE GIRLS YOU WANT SERVICE THEN CALL THIS NUMBER 9630942363
ROOM ALSO PROVIDE HOME & HOTELS SERVICE
FULL SAFE AND SECURE WORK
WITHOUT CONDOMS, ORAL, SUCKING, LIP TO LIP, ANAL, BACK SHOTS, SEX 69, WITHOUT BLOWJOB AND MUCH MORE
FOR BOOKING
9630942363Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available

Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service AvailableGENUINE ESCORT AGENCY
Recently uploaded (20)
Call Girls Kolkata Kalikapur 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...

Call Girls Kolkata Kalikapur 💯Call Us 🔝 8005736733 🔝 💃 Top Class Call Girl Se...
Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...

Call Girls Vasai Virar Just Call 9630942363 Top Class Call Girl Service Avail...
Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available
🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...

🌹Attapur⬅️ Vip Call Girls Hyderabad 📱9352852248 Book Well Trand Call Girls In...
Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...

Saket * Call Girls in Delhi - Phone 9711199012 Escorts Service at 6k to 50k a...
Top Rated Hyderabad Call Girls Erragadda ⟟ 9332606886 ⟟ Call Me For Genuine ...

Top Rated Hyderabad Call Girls Erragadda ⟟ 9332606886 ⟟ Call Me For Genuine ...
Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available
Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...

Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...
Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available

Trichy Call Girls Book Now 9630942363 Top Class Trichy Escort Service Available
Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...

Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...
Low Rate Call Girls Bangalore {7304373326} ❤️VVIP NISHA Call Girls in Bangalo...

Low Rate Call Girls Bangalore {7304373326} ❤️VVIP NISHA Call Girls in Bangalo...
Call Girls Rishikesh Just Call 9667172968 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 9667172968 Top Class Call Girl Service Available
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
Call Girls Hyderabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Hyderabad Just Call 8250077686 Top Class Call Girl Service Available
Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...

Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...
8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad

8980367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad
Manyata Tech Park ( Call Girls ) Bangalore ✔ 6297143586 ✔ Hot Model With Sexy...

Manyata Tech Park ( Call Girls ) Bangalore ✔ 6297143586 ✔ Hot Model With Sexy...
Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...

Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...
Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...

Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...
Aminoglycoside SAR one page.doc
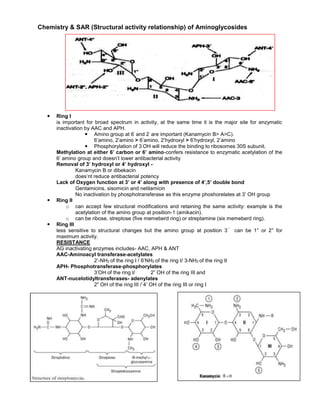
- 1. Chemistry & SAR (Structural activity relationship) of Aminoglycosides Ring I is important for broad spectrum in activity, at the same time it is the major site for enzymatic inactivation by AAC and APH. Amino group at 6΄ and 2΄ are important (Kanamycin B> A>C). 6’amino, 2’amino > 6’amino, 2’hydroxyl > 6’hydroxyl, 2’amino Phosphorylation of 3΄OH will reduce the binding to ribosomes 30S subunit. Methylation at either 6’ carbon or 6’ amino-confers resistance to enzymatic acetylation of the 6’ amino group and doesn’t lower antibacterial activity Removal of 3’ hydroxyl or 4’ hydroxyl - Kanamycin B or dibekacin does’nt reduce antibacterial potency Lack of Oxygen function at 3’ or 4’ along with presence of 4’,5’ double bond Gentamicins, sisomicin and netilamicin No inactivation by phosphotransferase as this enzyme phoshorelates at 3’ OH group Ring II o can accept few structural modifications and retaining the same activity: example is the acetylation of the amino group at position-1 (amikacin). o can be ribose, streptose (five memeberd ring) or streptamine (six memeberd ring). Ring III less sensitive to structural changes but the amino group at position 3΄΄ can be 1° or 2° for maximum activity. RESISTANCE AG inactivating enzymes includes- AAC, APH & ANT AAC-Aminoacyl transferase-acetylates 2’-NH2 of the ring I / 6’NH2 of the ring I/ 3-NH2 of the ring II APH- Phosphotransferase-phosphorylates 3’OH of the ring I/ 2” OH of the ring III and ANT-nucelotidyltransferases- adenylates 2” OH of the ring III /`4’ OH of the ring III or ring I