Acr 2014 poster
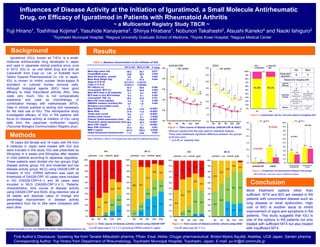
- 1. Influences of Disease Activity at the Initiation of Iguratimod, a Small Molecule Antirheumatic Drug, on Efficacy of Iguratimod in Patients with Rheumatoid Arthritis Yuji Hirano1, Toshihisa Kojima2, Yasuhide Kanayama3, Shinya Hirabara1, Nobunori Takahashi2, Atsushi Kaneko4 and Naoki Ishiguro2 Background 1Toyohashi Municipal Hospital, 2Nagoya University Graduate School of Medicine, 3Toyota Kosei Hospital, 4Nagoya Medical Center Iguratimod (IGU), known as T-614, is a small-molecule antirheumatic drug developed in Japan and used in Japanese clinical practice since June in 2012. IGU is an oral tablet drug and sold as Careram® from Eisai co. Ltd. or Kolbet® from Taisho Toyama Pharmaceutical Co. Ltd. in Japan. IGU is known to inhibit nuclear factor-kappa B activation in cultured human synovial cells. Although biological agents (BIO) have good efficacy to treat rheumatoid arthritis (RA), they costs very much. IGU is not comparatively expensive and used as monotherapy or combination therapy with methotrexate (MTX). Data in clinical practice is lacking and necessary for the best use of IGU. This retrospective study investigated efficacy of IGU in RA patients with focus on disease activity at initiation of IGU using data from the Japanese multicenter registry (Tsurumai Biologics Communication Registry plus). Methods ~ a Multicenter Registry Study TBCR ~ Results Figure 4: Continuation rate for 24w and reasens of stopping IGU Figure 5: Comparison of improvement between two groups Mann-Whitney U-test was used for statistical analysis. Conclusion 78 cases (62 female and 16 male) with RA from 9 institutes in Japan were treated with IGU and were included in this study. IGU was prescribed as 25mg/day in 4 weeks and 50mg/day after 4weeks in most patients according to Japanese regulation. These patients were divided into two groups (high disease activity group; HG and moderate and low disease activity group; MLG) using DAS28-CRP at initiation of IGU. IORRA definition was used as thresholds of DAS28-CRP. 42 cases were included in HG (DAS28-CRP>4.1) and 36 cases were included in MLG (DAS28-CRP ≦ 4.1). Patients’ characteristics, time course of disease activity using DAS28-CRP and SDAI, drug retention rate at 24 weeks and absolute value of change and percentage improvement in disease activity parameters from 0w to 24w were compared with each other. Table 1: Baseline characteristics at the initiation of IGU HG (n=42) MLG (n=36) Mean Age (years old) Female/Male (case) Mean RA duration (month) Mean Body Weight (kg) Stage (I/ II/ III/ IV) Class (1/ 2/ 3/ 4) RF>15IU/ml (%) Concomitant MTX (%) Mean MTX dose in all (mg/week) MTX dose in Only MTX-treated Concomitant PSL (%) Mean PSL dose (mg/day) DMARDs numbers including IGU Biologics-concomitant (case) Mean DAS28-CRP Mean SDAI Tender joint counts Swollen joints counts Patients’ global assessment (mm) Physicians' global assessment (mm) Mean CRP (mg/dl) Mean ESR (mm/hour) MMP-3 (ng/ml) Health Assessment Questionnaire P value 68.3 33/9 147 51.3 6/7/12/17 5/15/21/1 92.3 52.4 4.7 9.0 46.3 2.5 2.0 2 4.99 27.9 9.0 4.6 60.1 56.0 2.7 57.5 301.8 1.17 65.7 29/7 94 51.8 11/5/13/7 11/20/5/0 88.6 63.9 5.2 8.1 42.9 1.8 1.8 1 3.24 11.8 2.1 2.1 35.8 29.3 1.1 36.2 185.8 0.64 0.677 0.829 0.062 0.644 0.584 0.305 0.601 0.393 0.761 0.395 0.452 <0.0001 <0.0001 <0.0001 0.0049 <0.0001 <0.0001 <0.0001 0.001 0.0049 0.003 Mann-Whitney U-test or Chi-square test was used for statistical analysis. DAS28-CRP SDAI Figure 1: Time course of disease activity (DAS28-CRP & SDAI) Wilcoxon signed-rank test was used for statistical analysis. There were statistically significant difference between two groups at all time point. *: p<0.05 vs. baseline (0w). HG MLG Figure 2: Time course of disease activity criteria using DAS28-CRP Cut-off value was 4.1-2.7-2.3 according IORRA criteria in Japan. HG MLG Figure 3: Time course of disease activity criteria using SDAI Cut-off value was 26-11-3.3. More treatment options other than sufficient MTX and BIO are needed in RA patients with concomitant disease such as lung disease or renal dysfunction. High cost of BIO is another issue to inhibit improvement of signs and symptoms in RA patients. This study suggests that IGU is one of the options in RA patients not only treated with sufficient MTX but also treated with insufficient MTX. Careram® from Eisai co. Ltd. and Kolbet® from Taisho Toyama Pharmaceutical Co. Ltd. First Author’s Disclosure: Speeking fee from Tanabe Mitsubishi pharma, Pfizer, Eisai, Abbie, Chugai pharmaceutical, Bristol-Myers Squibb, Astellas, UCB Japan, Santen pharma Corresponding Author: Yuji Hirano from Department of Rheumatology, Toyohashi Municipal Hospital, Toyohashi, Japan, E-mail: yu-hr@sf.commufa.jp