Clinical Trial Supply and Logistics Market for Pharma 2018-2028 pharmaceutical clinical trials drug supply
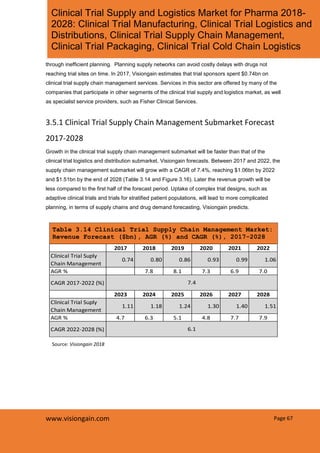
- 1. Page 67www.visiongain.com Clinical Trial Supply and Logistics Market for Pharma 2018- 2028: Clinical Trial Manufacturing, Clinical Trial Logistics and Distributions, Clinical Trial Supply Chain Management, Clinical Trial Packaging, Clinical Trial Cold Chain Logistics through inefficient planning. Planning supply networks can avoid costly delays with drugs not reaching trial sites on time. In 2017, Visiongain estimates that trial sponsors spent $0.74bn on clinical trial supply chain management services. Services in this sector are offered by many of the companies that participate in other segments of the clinical trial supply and logistics market, as well as specialist service providers, such as Fisher Clinical Services. 3.5.1 Clinical Trial Supply Chain Management Submarket Forecast 2017-2028 Growth in the clinical trial supply chain management submarket will be faster than that of the clinical trial logistics and distribution submarket, Visiongain forecasts. Between 2017 and 2022, the supply chain management submarket will grow with a CAGR of 7.4%, reaching $1.06bn by 2022 and $1.51bn by the end of 2028 (Table 3.14 and Figure 3.16). Later the revenue growth will be less compared to the first half of the forecast period. Uptake of complex trial designs, such as adaptive clinical trials and trials for stratified patient populations, will lead to more complicated planning, in terms of supply chains and drug demand forecasting, Visiongain predicts. Table 3.14 Clinical Trial Supply Chain Management Market: Revenue Forecast ($bn), AGR (%) and CAGR (%), 2017-2028 2017 2018 2019 2020 2021 2022 Clinical Trial Suply Chain Management 0.74 0.80 0.86 0.93 0.99 1.06 AGR % 7.8 8.1 7.3 6.9 7.0 CAGR 2017-2022 (%) 2023 2024 2025 2026 2027 2028 Clinical Trial Suply Chain Management 1.11 1.18 1.24 1.30 1.40 1.51 AGR % 4.7 6.3 5.1 4.8 7.7 7.9 CAGR 2022-2028 (%) 7.4 6.1 Source: Visiongain 2018
- 2. Page 92www.visiongain.com Clinical Trial Supply and Logistics Market for Pharma 2018- 2028: Clinical Trial Manufacturing, Clinical Trial Logistics and Distributions, Clinical Trial Supply Chain Management, Clinical Trial Packaging, Clinical Trial Cold Chain Logistics Figure 4.17 Japanese Clinical Trial Supply and Logistics: Drivers and Restraints, 2017-2028 be attracted to South Korea as a trial destination, as it offers lower cost trials but high regulatory standards. 4.6 Clinical Trials Increasing Demand in Emerging National Markets As of now, the four BRIC nations - the leading emerging pharmaceutical markets - accounts for just 8.6% of trials registered on ClinicalTrials.gov. However, these markets are growing in terms of demand for clinical trials, as companies continue to take advantage of access to large, treatment- naïve patient populations, low-cost trial management and access to growing pharmaceutical markets. According to the Biopharma Cold Chain 2012 Sourcebook, growth of 65% growth in clinical research will come from studies conducted in emerging market countries in 2017. Outsourcing to local and global CROs will boost the clinical trial industries in those markets in the coming 10 years. However, challenges remain with clinical trial logistics for these markets. Most drugs for clinical trials will be manufactured outside these countries, meaning that an understanding of local import regulations is required. Misunderstanding of import regulations can lead to costly delays in trials, as well as longer storage times while sponsors wait for drugs to clear customs. Longer storage times are also the result of less established supplier networks, particularly for temperature- The Japanese market is relatively new, compared with the US and EU, and the market is growing faster Leading global players have established logistics networks in Japan since 2007. Expansion in this decade will provide an improved network there Domestic drug developers have long pipelines, with many novel biopharmaceuticals in development Increased off-shoring of clinical trials to emerging Asian markets, particularly China and South Korea, will restrain logistics demand. Outsourcing is less common in Japan than in other developed markets and drug developers may prefer to keep activities in-house in this decade Strict rules regarding quality and control will restrain outsourcing Drivers Restraints Source: Visiongain 2018
- 3. Page 169www.visiongain.com Clinical Trial Supply and Logistics Market for Pharma 2018- 2028: Clinical Trial Manufacturing, Clinical Trial Logistics and Distributions, Clinical Trial Supply Chain Management, Clinical Trial Packaging, Clinical Trial Cold Chain Logistics Figure 8.4 Catalent: Clinical Manufacturing Space by Region (%), 2017 8.2.1.2 Strong Growth for Clinical Manufacturing Revenue In September 2016, Catalent acquired Pharmatek, a clinical manufacturing and contract drug development company that adds discovery-to-clinic drug development capabilities that expands the company’s portfolio with advanced delivery technologies such as spray drying. For the year 2015, Catalent’s DCS division revenue totalled $438.8m (Table 8.6), up 6.5% compared to 2014. Since 2011, Catalent’s revenue is growing with a CAGR of 29.3%. In 2012, Catalent acquired Aptuit which resulted in DCS revenue dramatic increase in 2013 - the acquisition reportedly added $50m revenue in each of the first and second quarters of that year alone. Catalent reports that, following the acquisition of Aptuit’s CTS division; it is the world’s second largest manufacturer of clinical trial materials. There was an addition of Antibody Drug Conjugate business when Catalent completed the acquisition of the Redwood BioScience business in October 2014. Later in the next month, company extended particle engineering capabilities via the acquisition of Micron Technologies. We believe our own internal innovation, supplemented by current and future external partnerships and acquisitions, will continue to strengthen and extend our leadership positions in the delivery and development of drugs, biologics and consumer health America 57.5% Europe 33.8% Asia Pacific 8.7% Source: Catalent 2018, Visiongain 2018
